Clear Sky Science · en
Prophage-encoded RexAB-type phage defense system in Pseudomonas putida
Hidden bodyguards living inside bacteria
Bacteria are constantly hunted by viruses called phages, which can wipe out whole microbial populations. This study looks at how “sleeping” viruses already hidden inside a bacterium’s DNA can secretly act as bodyguards, sacrificing infected cells to save the rest. Understanding these natural defense tricks matters for phage therapies, biotechnology, and even for keeping industrial microbes running reliably.
Viruses that turn from enemies into allies
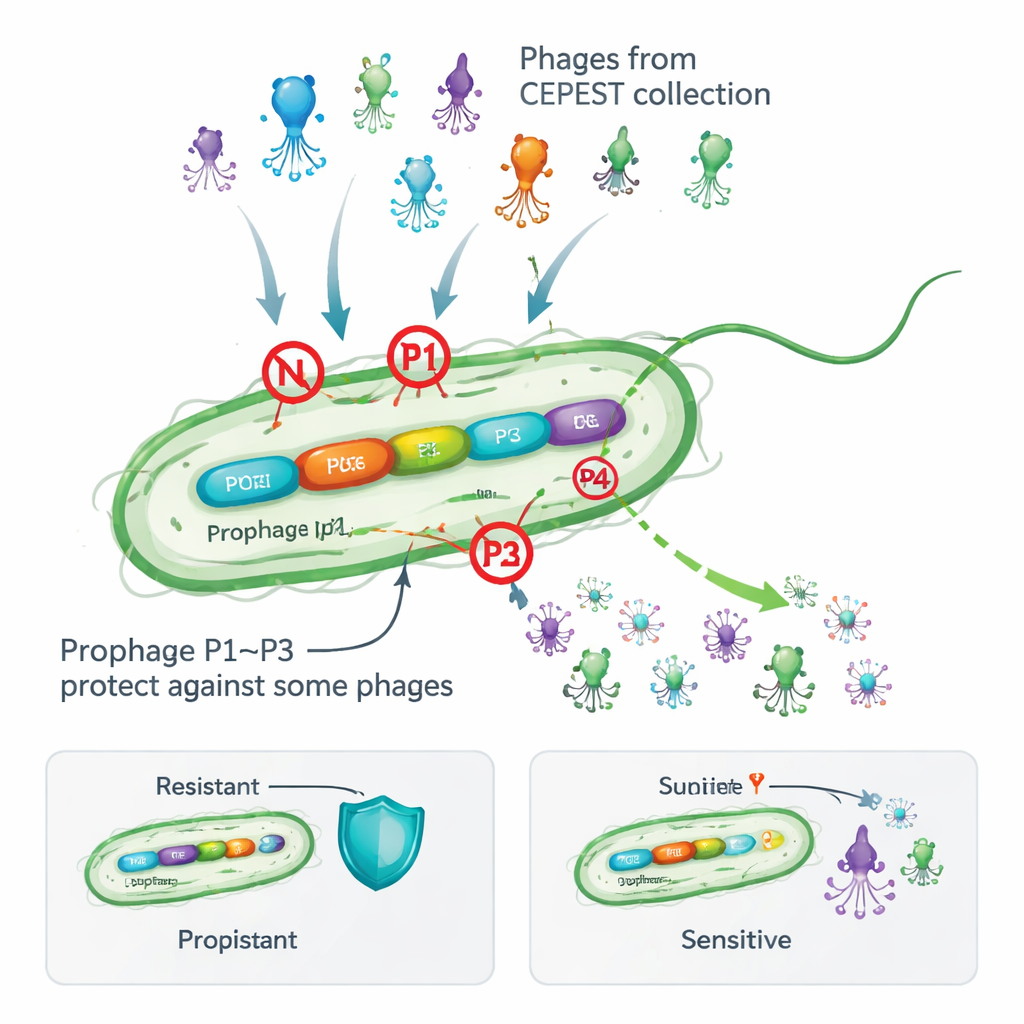
Many bacteria carry temperate phages in a dormant form called prophages, stitched into their chromosomes. At first glance this looks risky: these viral hitchhikers can wake up and damage the host cell, and they make the genome bulkier. Yet surveys of thousands of bacterial genomes show that most species keep such prophages, suggesting they offer advantages. Earlier work on the soil bacterium Pseudomonas putida showed that removing its four cryptic (non‑productive) prophages made the cells more vulnerable to attack by an experimental collection of phages called CEPEST. That hinted that the prophages carry anti‑phage weapons, but the exact genes and mechanisms were unknown.
Three resident viruses give targeted protection
The authors dissected the contribution of each prophage by deleting them one at a time and testing how well 26 different CEPEST phages could form plaques on bacterial lawns. They found that prophages named P1, P2, and P3 each provide protection, but only against particular groups of phages, while P4 appears neutral. P1 alone can cut infection by some phages roughly a thousand‑fold, P2 offers more modest protection, and P3 is spectacularly effective—but only against certain giant “jumbo” phages. In most cases, a single prophage explains the observed resistance pattern, though for a few phages multiple prophages seem to cooperate. These results show that even “silent” prophages can act as a customized shield tuned to specific viral threats.
Tracking down a two‑piece molecular alarm
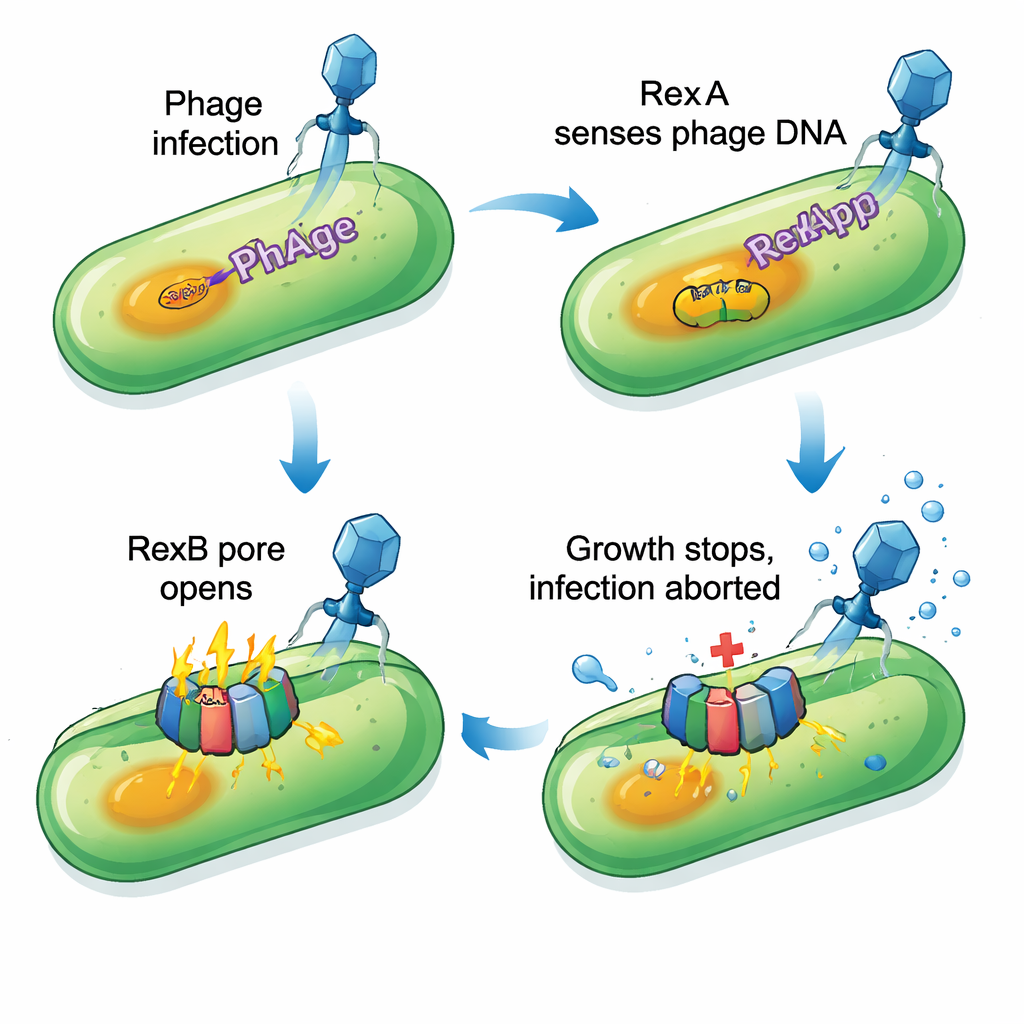
Focusing on the strong and relatively broad protection provided by P1, the team systematically deleted blocks of P1 genes until the defense vanished. Narrowing the search, they discovered that losing just two neighboring genes, PP_5643 and PP_5644, made the cells as sensitive as if the entire P1 prophage were gone. Bioinformatic tools revealed that PP_5643 resembles RexA, a DNA‑binding protein from classic lambda phage studies, while PP_5644 looks like a membrane protein with several helices that could form a pore, similar to RexB. Together, this pair matches a well‑known type of “abortive infection” system: once a specific phage is detected, the infected cell deliberately halts its own growth so the virus cannot multiply and spread to its neighbors.
How the defense sabotages infected cells
Experiments confirmed that the P1 version of RexA (called RexApp) binds DNA without caring much about the exact sequence, making it well suited to sense unusual DNA structures created during viral replication. The partner protein RexBpp embeds in the cell membrane. When RexApp is artificially overproduced, cells that still carry the rexBpp gene show severe growth arrest and signs of membrane damage: dyes that normally cannot cross intact membranes suddenly enter, and small amounts of a cytoplasmic enzyme leak out. If rexBpp is missing or mutated, RexApp overproduction no longer harms the cells. This indicates that RexApp acts as a trigger, while RexBpp is the effector that damages the cell envelope. Interestingly, most affected cells do not fully burst; instead they enter a deeply slowed, almost stationary state that appears incompatible with phage multiplication.
Why giving up a few cells can save the population
The picture that emerges is of an altruistic defense: when a sensitive phage infects P. putida, RexApp likely detects viral DNA–protein complexes and switches on RexBpp, which compromises the membrane and abruptly stops growth. The infected cell pays a price, but the phage’s life cycle is aborted and the wider bacterial community is protected. Systems of this RexAB type have now been found in only a handful of different phages, yet they share the same core logic—an intracellular sensor linked to a membrane effector that can quickly turn a productive cell into a dead‑end for the virus. For bacteria, keeping cryptic prophages that carry such defense modules can offset the costs of harboring viral DNA, helping them survive in a world where phages are everywhere.
Citation: Rosendahl, S., Kängsep, A., Ainelo, A. et al. Prophage-encoded RexAB-type phage defense system in Pseudomonas putida. Sci Rep 16, 5898 (2026). https://doi.org/10.1038/s41598-026-36734-5
Keywords: bacteriophage defense, prophage, Pseudomonas putida, abortive infection, RexAB system