Clear Sky Science · en
Optimized thermal response of Au nanoframes in NIR-II window: a numerical study
Gentle Heat as a Cancer-Fighting Tool
Doctors are increasingly turning to tiny particles of gold to fight cancer by warming tumors from the inside. The challenge is to heat cancer cells enough to damage them, without burning surrounding healthy tissue or breaking the particles themselves. This study uses advanced computer simulations to design a new type of hollow gold particle, called a double-torus nanoframe, that can safely and efficiently heat tumors deep inside the body using a special kind of invisible light.
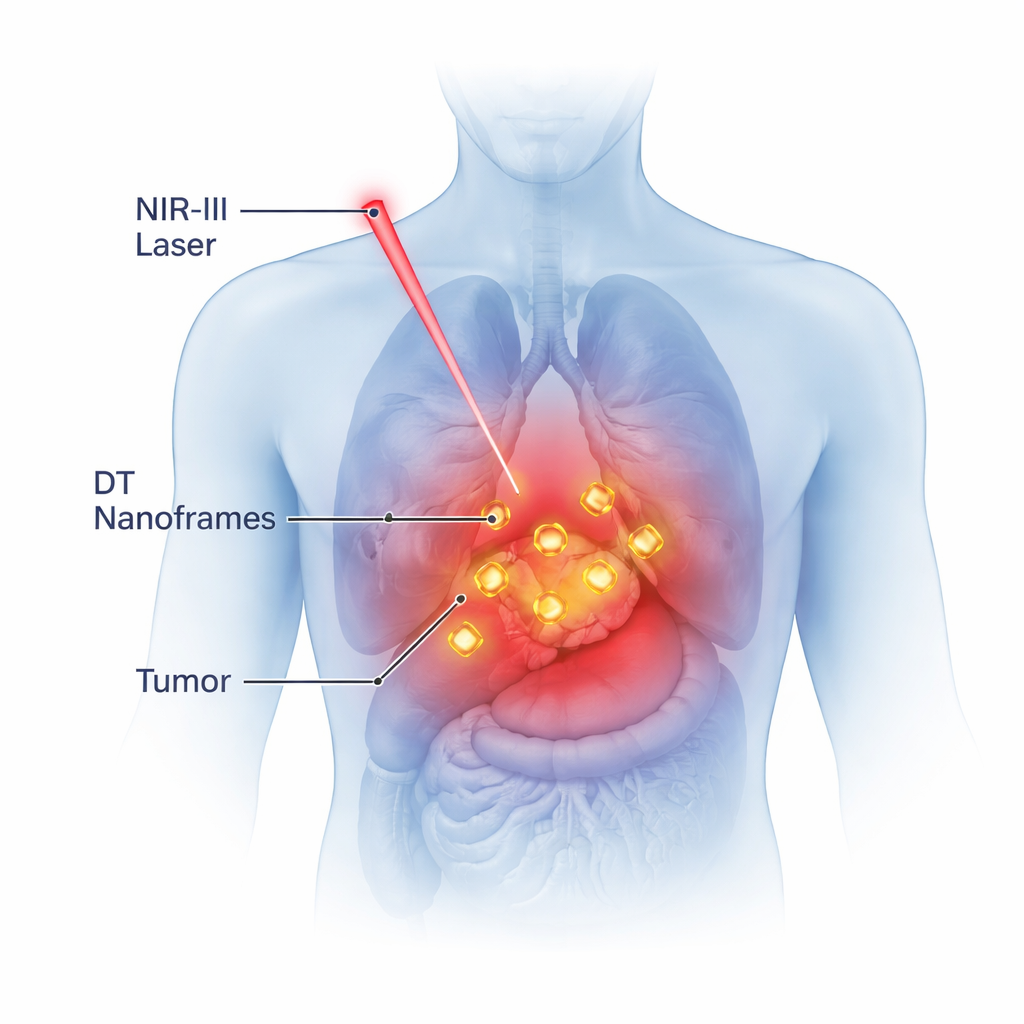
Why Invisible Light Matters
Our bodies block or scatter most visible light, which limits how deeply doctors can shine light into tissue. However, there is a “sweet spot” in the near-infrared spectrum, known as the NIR-II window (1000–1400 nanometers), where light can travel several centimeters into the body with less scattering and damage. Gold nanoparticles can be tuned so that their electrons vibrate strongly at specific wavelengths of light, a phenomenon called resonance. When this happens in the NIR-II window, the particles can efficiently absorb laser light and turn it into heat exactly where it is needed, deep inside a tumor.
Limits of Today’s Gold Nanoparticles
Many gold shapes have been tried for cancer heating: solid spheres, cubes, rods, ring-like structures, and thin “frame” shells. Each has drawbacks. Solid particles often cannot be tuned far enough into the NIR-II window. Gold nanorods heat very efficiently, but they can overheat, deform into spheres, and lose their special optical behavior. Cubic and spherical gold nanoframes can focus heat at sharp corners, which is useful, but those same sharp features make them vulnerable to rounding and shape change when exposed to strong heating. Ring-shaped nanotori can be tuned into the right wavelength range but absorb less heat and their performance depends strongly on how they are oriented relative to the laser light, a problem for particles freely floating in blood.
A New Double-Ring Gold Frame
To overcome these issues, the researchers propose a new design: a double-torus nanoframe made of two hollow gold rings arranged perpendicular to each other, like a three-dimensional figure-eight. Using computer models, they compared this design with standard nanorods, cubic and spherical frames, and single nanotori. All the particles were adjusted so that their resonant wavelength fell within the NIR-II window. Then, using a combination of optical and heat-transfer simulations, they tracked how much heat each particle produced over time, and how that heating changed when particles were randomly oriented in water, as they would be in the bloodstream.
Balancing Heat, Stability, and Size
The study focused on achieving a precise temperature range: about 40–49 °C, hot enough to stress or kill cancer cells (hyperthermia) but not so hot that tissue is burned or the particles melt or deform. The simulations showed that some shapes, like cubic frames and nanorods, can heat very quickly but risk overshooting this safe window or changing shape under prolonged heating. Single nanotori, on the other hand, often failed to reach therapeutic temperatures, especially when their orientation to the laser was unfavorable. The spherical and cubic frames also turned out to be highly sensitive to small changes in their thickness or porosity, which can easily happen during manufacturing or under heat, shifting their behavior out of the desired range.
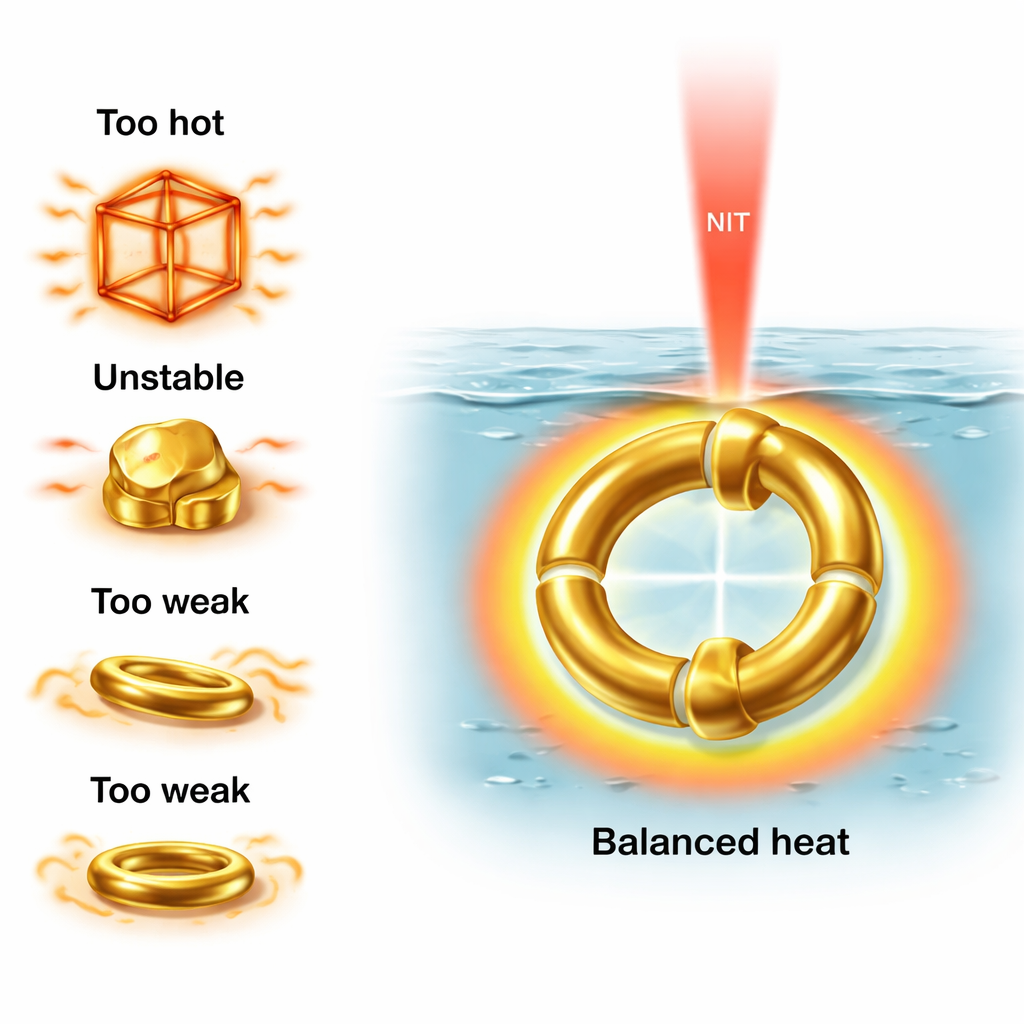
Why the Double-Torus Stands Out
The double-torus design combines several advantages. Its high symmetry means it absorbs light and generates heat in a stable way, even when randomly oriented; it does not rely on being aligned with the laser’s polarization. Its curved, rounded shape offers more resistance to heat-induced deformation than sharp-edged frames. Because it contains more gold than a single torus, it can generate sufficient heat while still staying within the safe hyperthermia window over a wide range of sizes and volumes. This extra metal volume also makes it promising for dual roles: not only heating tumors, but also scattering light strongly enough to help with imaging and local temperature sensing.
Implications for Future Cancer Treatments
For non-specialists, the key takeaway is that the exact shape of a gold nanoparticle can make or break its usefulness as a cancer-heating tool. This work suggests that double-torus nanoframes provide a sweet balance between strong, controllable heating and structural stability under realistic conditions. While challenges remain in fabricating such smooth, curved gold frames reliably, the simulations point to them as a compelling blueprint for future nanoparticles that can precisely warm tumors deep inside the body, improving both the safety and effectiveness of light-based cancer therapies.
Citation: Alali, F.A. Optimized thermal response of Au nanoframes in NIR-II window: a numerical study. Sci Rep 16, 5658 (2026). https://doi.org/10.1038/s41598-026-36727-4
Keywords: gold nanoparticles, photothermal therapy, cancer treatment, near-infrared light, nanomedicine