Clear Sky Science · en
Development and validation of a PSMA-positive triple-negative breast cancer mouse model for preclinical targeted radionuclide therapies
Why this research matters
Triple-negative breast cancer is one of the toughest forms of breast cancer to treat. It tends to spread quickly, offers patients fewer targeted treatment options, and often comes back after standard therapies. At the same time, a new class of “smart” radiation drugs that lock onto a specific molecule called PSMA has transformed care for advanced prostate cancer. This study asks a simple but powerful question: can we build a realistic laboratory model of triple-negative breast cancer that carries the same PSMA target, so we can fairly test whether these smart radiation drugs might also help patients with this aggressive breast cancer?
From prostate target to breast cancer challenge
PSMA, short for prostate-specific membrane antigen, is a small structure that sits on the surface of certain cells. It is abundant in many prostate cancers and can be recognized by radioactive drugs that both reveal tumors on scans and deliver radiation directly to them. Researchers have recently discovered that PSMA also appears, to varying degrees, in the blood vessels and cells of several other cancers, including triple-negative breast cancer. However, in these non-prostate tumors PSMA tends to be patchy and weaker, making it hard to know whether PSMA-targeted therapies will work. Before trying new treatments in patients, scientists need animal models that faithfully copy this PSMA pattern in breast tumors—something that has been surprisingly hard to achieve.
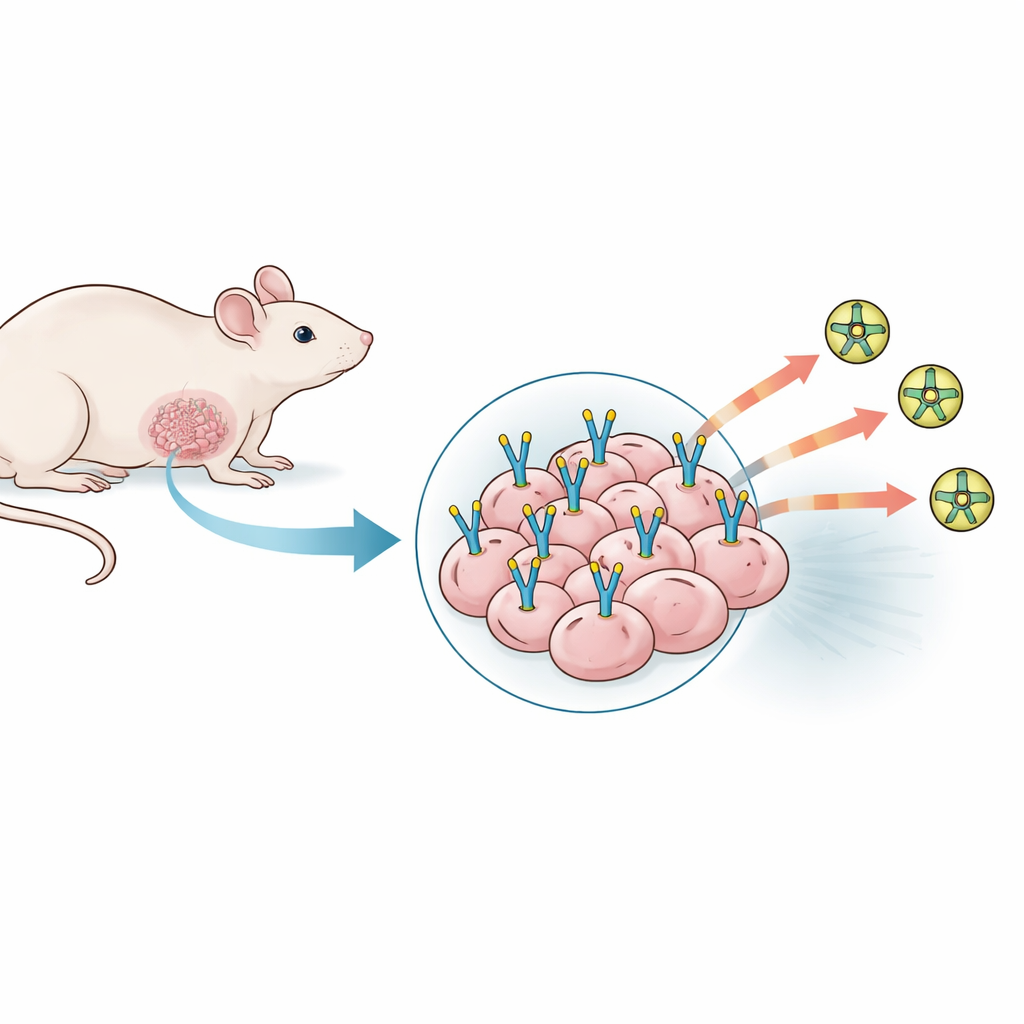
Testing many tumor models that refused to cooperate
The team first explored a wide range of existing mouse models of triple-negative breast cancer, using both mouse and human cancer cell lines implanted in different locations and under different conditions. They altered the number of cancer cells injected, added supportive gels to promote blood vessel growth, and even mixed in human blood vessel cells in hopes of boosting PSMA levels. For each model, they measured how reliably tumors took hold and grew, inspected tumor slices under the microscope, and used a PSMA-seeking PET tracer to see whether the tumors lit up on whole-body scans. Despite robust tumor growth and rich networks of blood vessels, none of these 23 models showed meaningful PSMA presence, either on tissue staining or on PET imaging. The tumors looked active and well supplied with blood, but the specific PSMA “docking site” the researchers needed was essentially absent.
Engineering a tumor that displays the right target
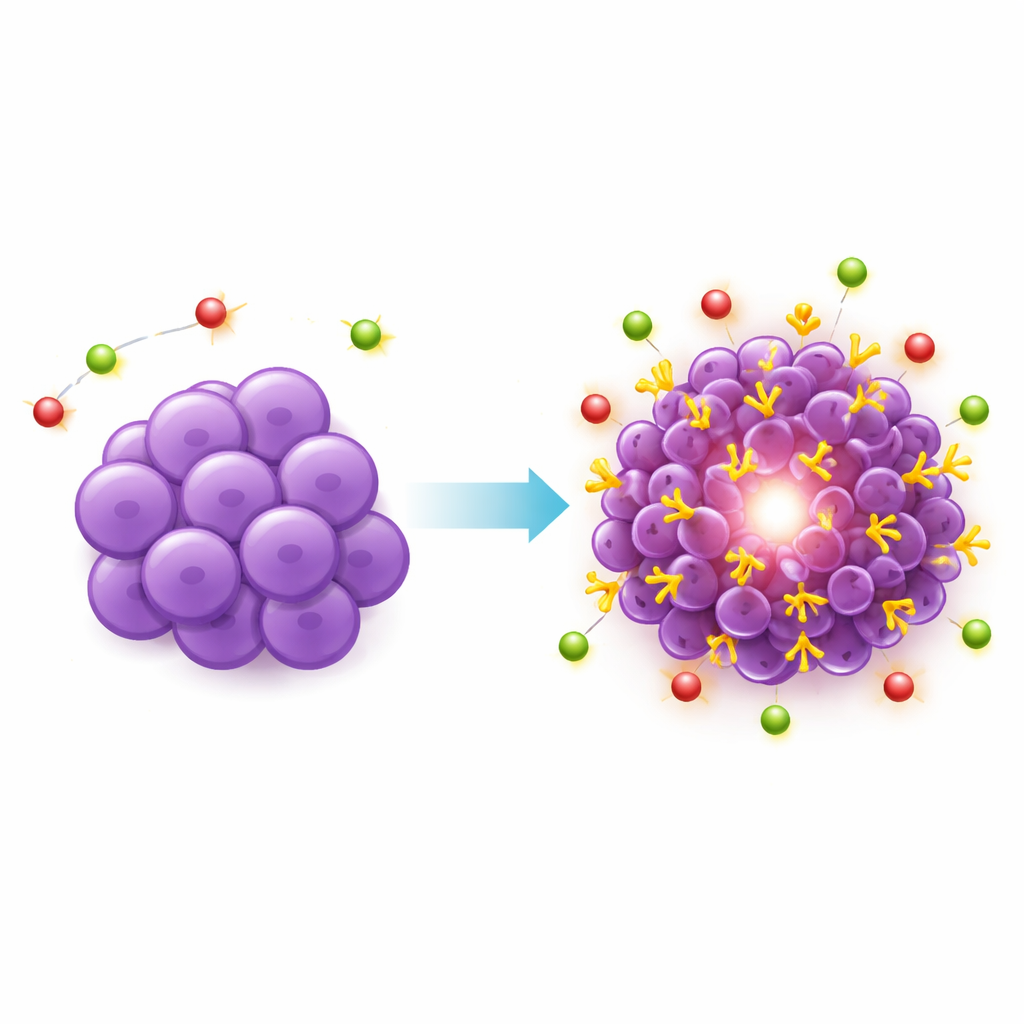
Faced with this roadblock, the researchers took a more direct route. They genetically modified a widely used human triple-negative breast cancer cell line, MDA-MB-231, to force it to produce PSMA on its surface. These altered cells were then implanted into the mammary fat pad of immune-deficient mice, a site that better mimics the natural breast environment. The scientists created two versions of the model: one where all tumor cells expressed PSMA, and another “mixed” tumor containing equal parts PSMA-positive and ordinary cancer cells, to imitate the patchy patterns seen in patients. Both models formed tumors just as reliably and grew at similar speeds as tumors made from unmodified cells, showing that adding PSMA did not make the cancer itself behave oddly.
Seeing the new target on scans and under the microscope
When the PSMA-hunting PET tracer was injected into these mice, the engineered tumors shone brightly on the scans, with tumor uptake roughly ten times higher than in the liver or muscle. This strong and selective signal confirmed that PSMA was now present in abundance on the cancer cells and could be reached by a circulating drug. Microscopic analysis of tumor tissue agreed: PSMA staining was high but uneven, with patches of stronger and weaker expression, resembling the heterogeneous pattern observed in human triple-negative breast cancers. Importantly, the added PSMA was confined to tumor cells, not to surrounding blood vessels, keeping the model focused on cell-targeted therapies. Areas of dead tissue inside larger tumors did not take up the tracer, matching what is seen clinically.
What this means for future treatments
By systematically showing that standard breast cancer mouse models do not reliably express PSMA and then building a new model that does, this work provides a crucial testing ground for next-generation targeted radiation treatments. The PSMA-positive triple-negative breast cancer model is stable, grows predictably, and shows a realistic mix of high and low PSMA areas, making it well suited to evaluate how PSMA-directed drugs behave before they are tried in patients. While this model cannot capture every aspect of human disease—such as the influence of a fully functioning immune system—it offers a powerful tool to explore whether the success of PSMA-based therapies in prostate cancer might one day be extended to people living with this particularly aggressive form of breast cancer.
Citation: Chaussin, B., Sanchez, L., Levesque, S. et al. Development and validation of a PSMA-positive triple-negative breast cancer mouse model for preclinical targeted radionuclide therapies. Sci Rep 16, 9348 (2026). https://doi.org/10.1038/s41598-026-36724-7
Keywords: triple-negative breast cancer, PSMA, targeted radionuclide therapy, preclinical mouse model, PET imaging