Clear Sky Science · en
Enhancing structural and optical properties of hybrid perovskite layers with polymer modification
A New Way to Make Solar Materials Last Longer
Modern solar panels are getting better at turning sunlight into electricity, but many promising next-generation materials wear out too quickly in everyday air and humidity. This study explores how a pinch of common plastics—polymers already used in products from shampoos to contact lenses—can make a fragile but highly efficient solar material more robust, potentially helping future solar cells last long enough for real-world use.
Why These New Solar Crystals Matter
At the heart of this work are “perovskites,” a family of crystals that absorb light remarkably well and can be made using low-cost, solution-based methods rather than high‑temperature processing. The researchers focus on a hybrid perovskite that mixes an organic molecule (methylammonium), an inorganic ion (cesium), and lead and iodine atoms in a carefully tuned recipe. This particular blend is attractive because it combines strong light absorption with a band gap around 1.58 electron volts—close to ideal for solar energy conversion. The big drawback, however, is that such perovskites tend to fall apart when they meet moisture and oxygen, losing their dark, light-harvesting color and turning yellow as their crystal structure degrades.
Helping Crystals with a Little Plastic
To tackle this weakness, the team mixed tiny amounts of three water‑soluble polymers—polyethylene glycol (PEG), polyvinylpyrrolidone (PVP), and polyvinyl alcohol (PVA)—directly into the liquid perovskite solution before coating it onto glass. Using a one‑step spin‑coating process, they spread the solution across transparent, conductive glass and then gently heated it to form thin, dark perovskite films. In this recipe, the polymers behave a bit like molecular scaffolds or glue: their chemical groups can latch onto the perovskite’s building blocks, guide how crystals grow, and help seal up tiny defects at the boundaries between grains. The researchers systematically varied polymer type and concentration to see which combination best improved film quality and durability.
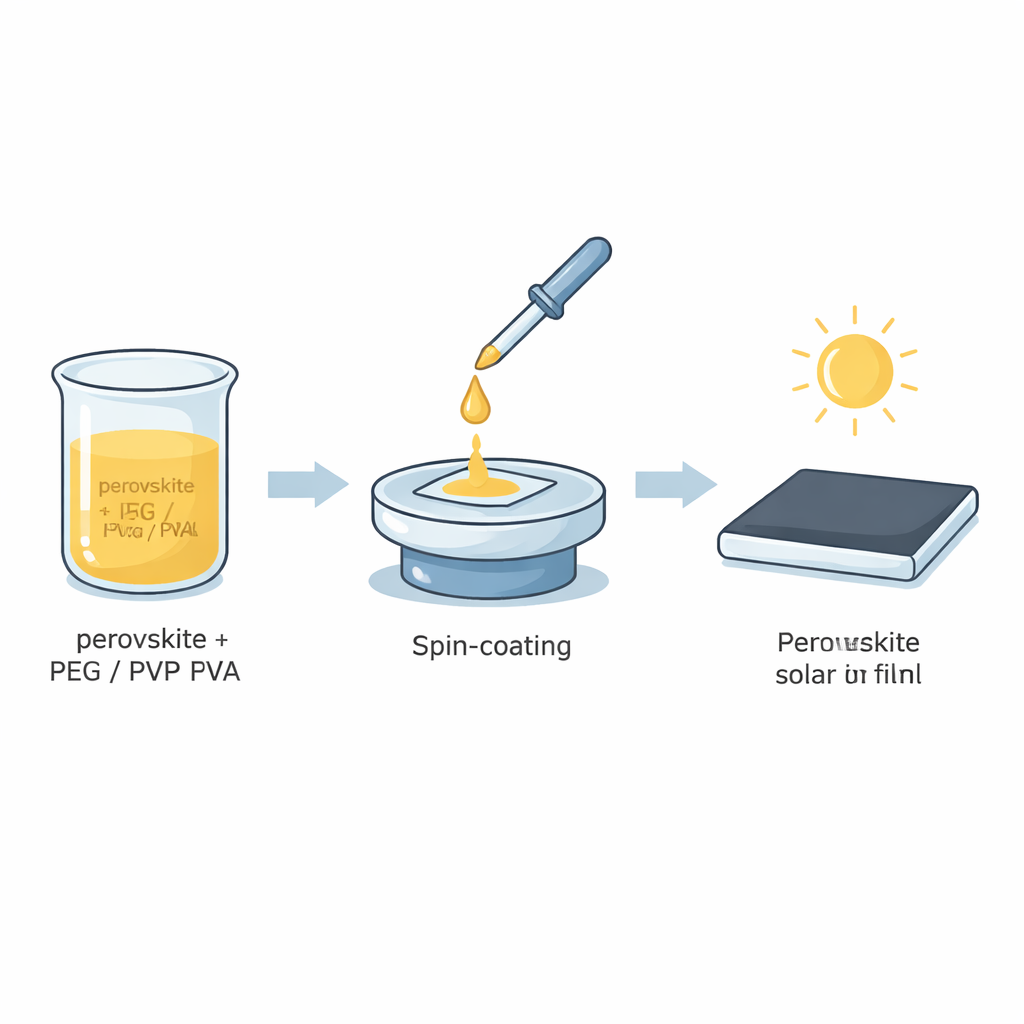
Sharper Films, Better Light Capture
Detailed tests showed that adding polymers made the perovskite films more orderly and more effective at catching light. X‑ray measurements confirmed that the underlying crystal structure stayed intact, while microscopy revealed that the grains grew larger and surfaces became smoother, especially when PEG was used at a moderate concentration (0.3 milligrams per milliliter). Optical measurements found that all polymer‑modified films absorbed more light than the unmodified perovskite, without altering the band gap—meaning they harvest more sunlight but still match the energy range ideal for solar cells. Other measurements that trace how the material glows after being excited by light, as well as how electric current flows through it, indicated that the right amount of polymer can reduce harmful defects and help charges move more easily rather than being trapped and lost as heat.
How One Polymer Stood Out
Among the different additives, PEG at 0.3 mg/mL emerged as the clear winner. Structural probes such as Raman spectroscopy showed that this formulation produced crystals with less internal stress and fewer imperfections. Electrical tests that track how easily charges move across the film found that this PEG level gave the lowest resistance to charge transfer, a sign that electrons and holes can travel more freely. Atomic‑scale surface scans demonstrated that PEG‑treated films were smoother and more uniform, while chemical mapping confirmed that the elements were evenly distributed. Most compellingly, when the films were simply left in air at room temperature and about 30% relative humidity, the PEG‑optimized samples largely retained their dark color and structure for up to 1,000 hours, whereas unmodified films and those with less suitable polymer loadings degraded much more quickly.
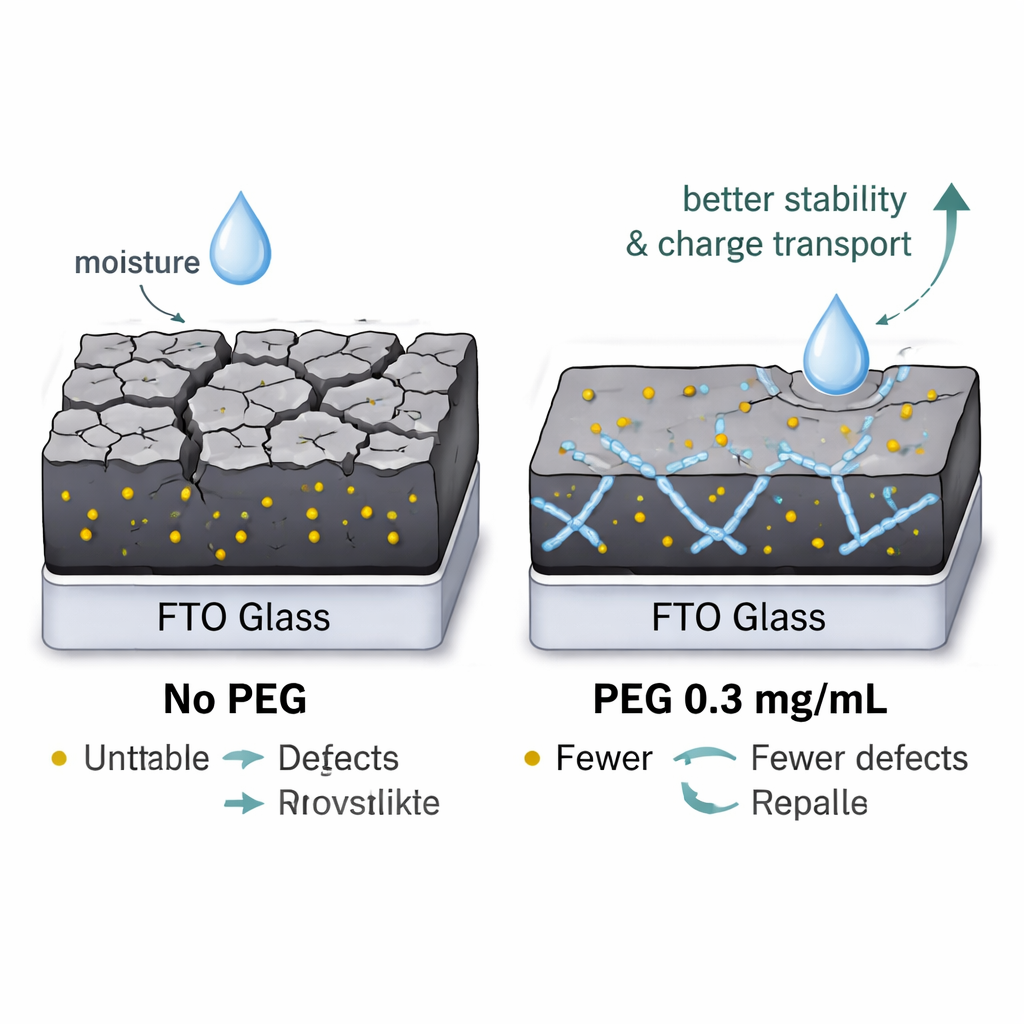
What This Means for Everyday Solar Power
To someone outside the lab, the message is straightforward: by stirring a small, carefully chosen amount of familiar plastic into a cutting‑edge solar material, the researchers significantly strengthened it without sacrificing performance. PEG at the right concentration acts like a microscopic stabilizer and repair kit, tightening the crystal network, blocking the pathways where water and oxygen would normally sneak in, and making it easier for electric charges to travel. While these experiments were done on individual layers rather than full commercial panels, they point toward a practical way to make high‑efficiency perovskite solar cells more reliable and closer to surviving years of outdoor use.
Citation: Bahramgour, M., Niaei, A., Asghari, E. et al. Enhancing structural and optical properties of hybrid perovskite layers with polymer modification. Sci Rep 16, 6210 (2026). https://doi.org/10.1038/s41598-026-36719-4
Keywords: perovskite solar cells, polymer additives, material stability, thin film photovoltaics, hybrid perovskites