Clear Sky Science · en
Knock-down of the long isoform of the WNK1 kinase mitigates the anti-glomerular basement membrane glomerulonephritis in mice
Why this matters for kidney health
Rapidly progressive kidney diseases can destroy the organ's tiny filters in a matter of weeks, often leading to dialysis or a transplant. This study explores whether dialing down a single protein, called L-WNK1, can slow that destruction in mice. Because drugs against this protein are already being developed for blood pressure problems, the findings hint at a possible new way to protect kidneys under attack.
A traffic controller inside kidney filters
Each kidney contains hundreds of thousands of microscopic filters, or glomeruli, that clean the blood. Within these filters, two key cell types work together: podocytes, which wrap around blood vessels like interlocking fingers, and parietal epithelial cells (PECs), which line the outer shell. In a dangerous condition called rapidly progressive glomerulonephritis (RPGN), podocytes are injured and PECs become overactive, piling up into “crescents” that block filtration. The protein L-WNK1, a signaling switch best known for helping the body balance salt and blood pressure, is actually made in high amounts in these glomerular cells. The authors asked whether too much L-WNK1 worsens RPGN, and whether reducing it could blunt the disease.
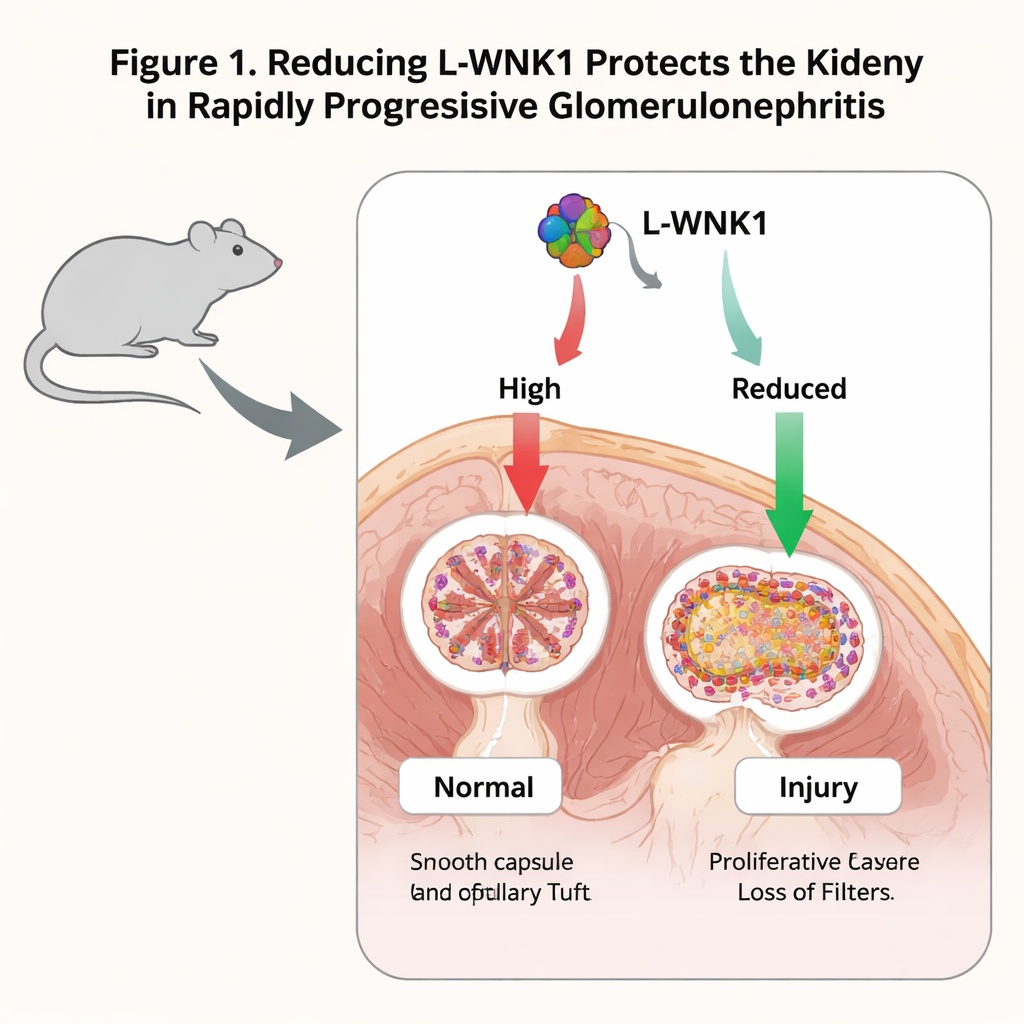
Turning down L-WNK1 eases kidney damage in mice
The researchers first used a classic mouse model of RPGN triggered by antibodies that attack the glomerular basement membrane, a key filter component. They found that levels of L-WNK1 rose in glomeruli after injury, not only in mice but also in kidney biopsies from patients with related diseases. To test cause and effect, they studied mice born with only one working copy of the L-WNK1 gene. These animals developed kidney failure after antibody injury, but their blood waste levels were lower, and their kidney tissue showed fewer crescents and less scarring than in normal mice. Markers of inflammation and scarring, such as the signaling molecules MCP-1, TGF-β1 and collagen genes, were also reduced. Together, this suggested that partial loss of L-WNK1 makes the disease milder rather than worse.
Zooming in on podocytes and their neighbors
Because podocytes are central to glomerular health and strongly express L-WNK1, the team next removed WNK1 specifically from these cells using a genetic trick. Under normal conditions these mice had healthy kidneys, showing that podocytes do not need L-WNK1 to develop or function in everyday life. When RPGN was induced, podocyte-specific knockout mice again fared better than controls: their blood tests were improved and they tended to have fewer crescents and fewer PECs expressing CD44, a marker of activation. However, the protection was weaker than in mice with the broader, bodywide reduction of L-WNK1, and levels of some inflammatory markers did not fall. This pointed to a role for L-WNK1 not only in podocytes but also in other glomerular or immune cells that participate in the disease.
Slowing harmful cell movement
To explore how L-WNK1 might drive crescent formation, the investigators turned to cell cultures. Using a small-molecule drug that blocks WNK kinases, they treated human podocytes grown in dishes and watched how quickly they crawled into an artificial “wound” gap. Blocking L-WNK1 activity markedly slowed their movement. They observed a similar effect in cultured PECs: when a growth factor was added, PECs normally spread to fill the gap, but this response was blunted when the WNK inhibitor was present. Interestingly, the drug did not noticeably disturb the internal scaffolding of the podocytes in their system, despite earlier work in mouse cells suggesting major structural problems. This supports the idea that, at least in living animals, L-WNK1’s main role here may be to control how glomerular cells and immune cells move and respond during injury rather than to maintain basic filter architecture.
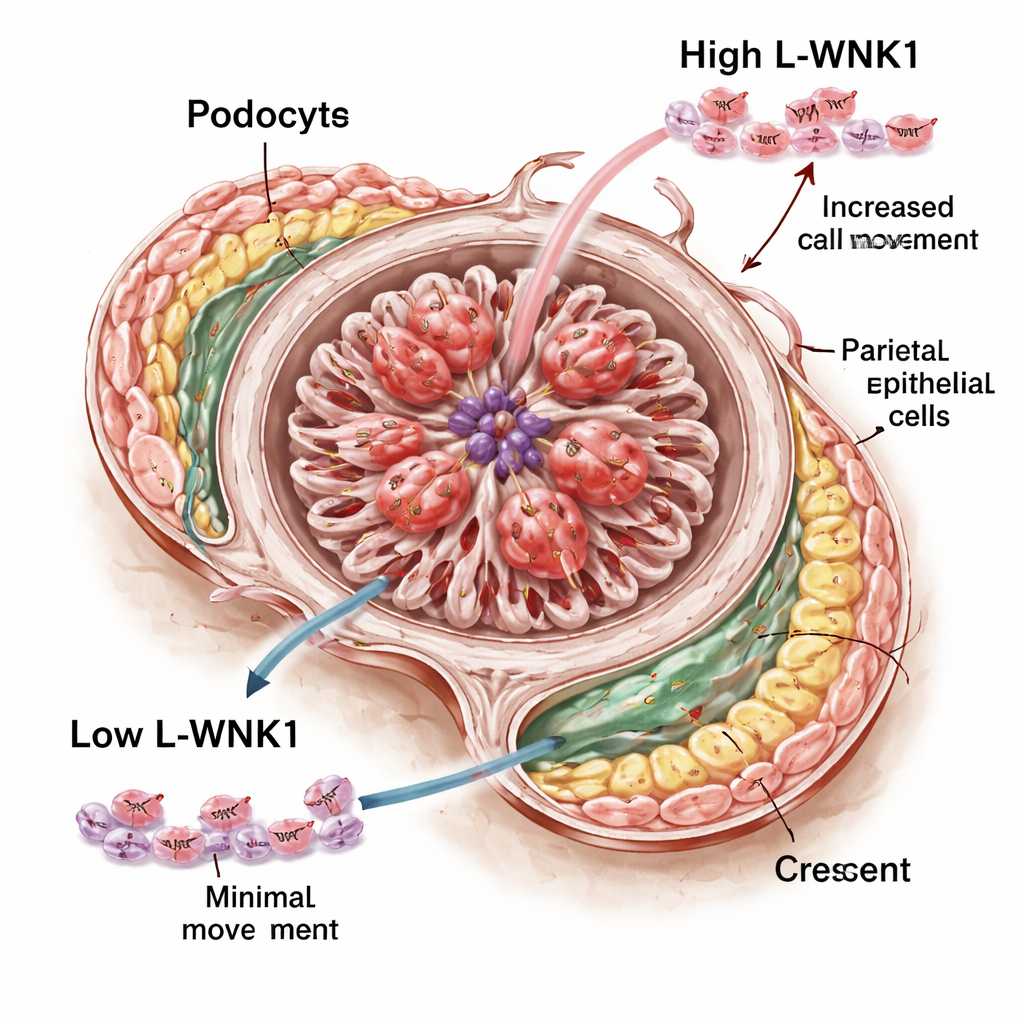
What this means for future treatments
Overall, the study reveals L-WNK1 as a harmful accelerator in a violent form of kidney inflammation: when this protein is turned up, key cells in the glomerulus move and multiply in ways that clog and scar the filters; when its activity is reduced, the damage is softened. Because L-WNK1 is already being studied as a drug target for blood pressure, these findings raise the possibility that carefully tuned inhibitors might one day help protect patients with rapidly progressive kidney diseases. Much work remains to translate mouse and cell-culture results into safe therapies, but L-WNK1 now stands out as a promising new handle on an otherwise devastating condition.
Citation: Mousseaux, C., Migeon, T., Frère, P. et al. Knock-down of the long isoform of the WNK1 kinase mitigates the anti-glomerular basement membrane glomerulonephritis in mice. Sci Rep 16, 7335 (2026). https://doi.org/10.1038/s41598-026-36715-8
Keywords: rapidly progressive glomerulonephritis, kidney inflammation, podocytes, glomerular crescents, WNK1 inhibition