Clear Sky Science · en
Can evolutionary therapy be applied in non-small cell lung cancer?
Rethinking cancer treatment as an evolutionary contest
Cancer care is often portrayed as a straightforward battle: give the strongest possible drugs and try to wipe out every last cancer cell. Yet in fast, aggressive cancers like advanced non-small cell lung cancer (NSCLC), this “maximum attack” approach frequently backfires. Tumors shrink at first, but almost always grow back, now dominated by drug-resistant cells. This study asks a provocative question: what if, instead of trying to obliterate the tumor, doctors treated it like an evolving ecosystem and used drug schedules that deliberately keep some drug‑sensitive cells alive to hold resistant cells in check?
Why standard high-dose treatment can speed resistance
In current practice, patients with metastatic NSCLC and certain genetic changes often receive daily pills called tyrosine kinase inhibitors (TKIs), such as erlotinib. These drugs can dramatically shrink tumors at first, but nearly all patients eventually relapse because resistant cancer cells survive and take over. When doctors push the dose to the maximum patients can tolerate, they mainly kill the sensitive cells that respond well to the drug. Resistant cells, which can survive even high doses, suddenly face less competition for space and nutrients. The result is an evolutionary “win” for the toughest cancer cells, and the tumor ultimately returns, now much harder to treat. 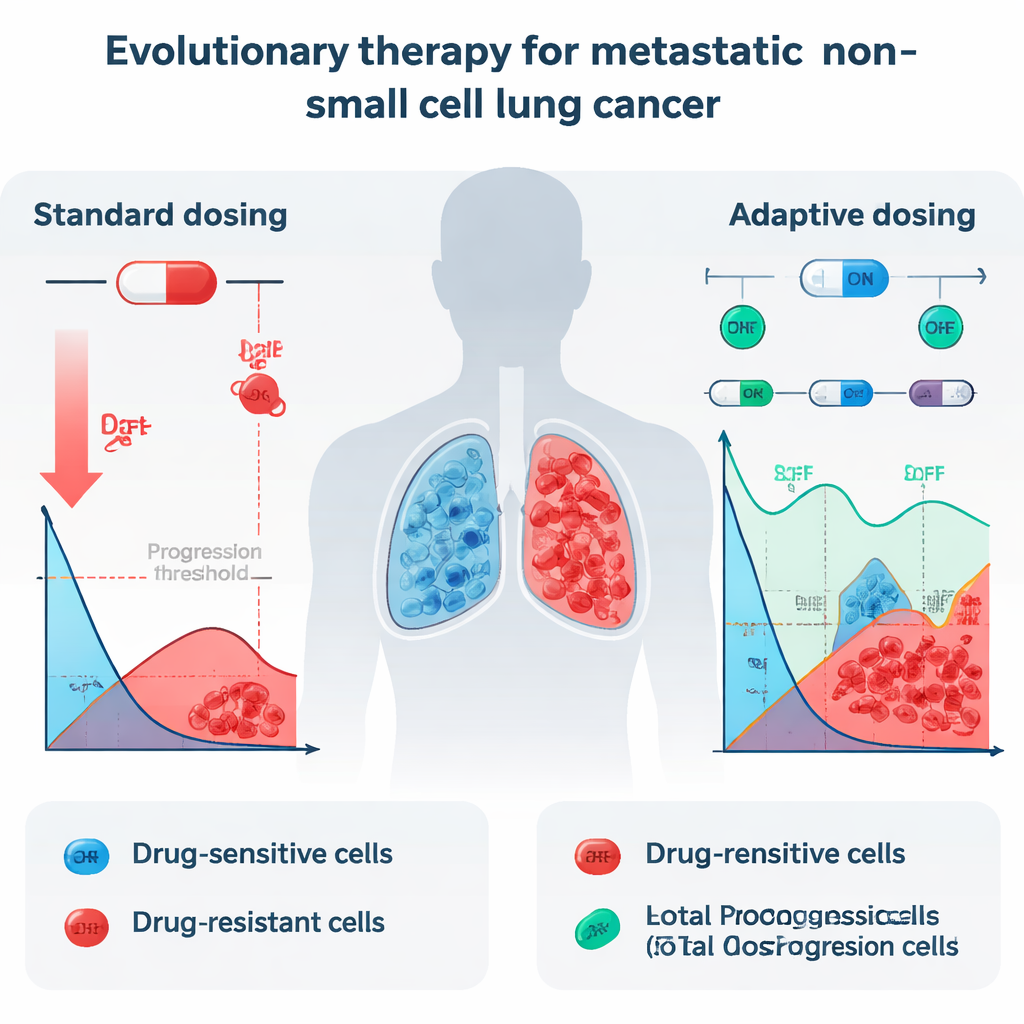
Using math to test a new dosing strategy on real patients
The researchers explored whether a different strategy, called evolutionary therapy, could work in stage IV NSCLC. Evolutionary therapy does not aim to eradicate the tumor outright. Instead, it tries to control it by keeping a stable mix of sensitive and resistant cells, letting the sensitive cells “crowd out” the resistant ones. The team borrowed a specific on–off dosing protocol that had already shown promise in prostate cancer, then asked: would this kind of strategy be theoretically effective for lung cancer as well? To answer this, they analyzed detailed tumor-size measurements from 13 NSCLC patients treated with erlotinib in a clinical trial, converting the scans into total tumor volume over time and feeding these data into a suite of mathematical models.
Finding the model that really captures resistance
The investigators tested 26 different models of tumor growth, all dividing cancer cells into two groups: drug-sensitive and drug-resistant. These models differed in how they described growth limits, how the two cell types compete with each other, how the drug is processed in the body, and how treatment kills cells. Many simpler models fit the patient data reasonably well when tumors only shrank. But when tumors followed a more realistic “U-shaped” path—shrinking with treatment, then regrowing as resistance emerged—most models failed. The best match was a type called the Gompertzian model that included two crucial ideas: first, tumors cannot grow without limit (they feel crowding), and second, sensitive and resistant cells compete in an asymmetric way, so the success of one type depends on how many of the other type are present.
Simulating adaptive therapy versus standard care
Once they identified the best-fitting models, the team used them to simulate two treatment strategies for each patient: the usual constant maximum dose and an adaptive protocol inspired by Zhang and colleagues. In the adaptive approach, erlotinib is given until the tumor shrinks to half its original size, then the drug is paused to allow sensitive cells to regrow; treatment restarts once the tumor returns to its starting size, and this cycle repeats. Across all well-performing models that included competition between cell types, this adaptive strategy consistently delayed the time until the tumor exceeded 110% of its original size—a common benchmark for progression. In the best-fitting Gompertzian model with competition, the median time to progression increased from about 24.8 months under standard dosing to 42.3 months under the adaptive protocol, a gain of roughly a year and a half. 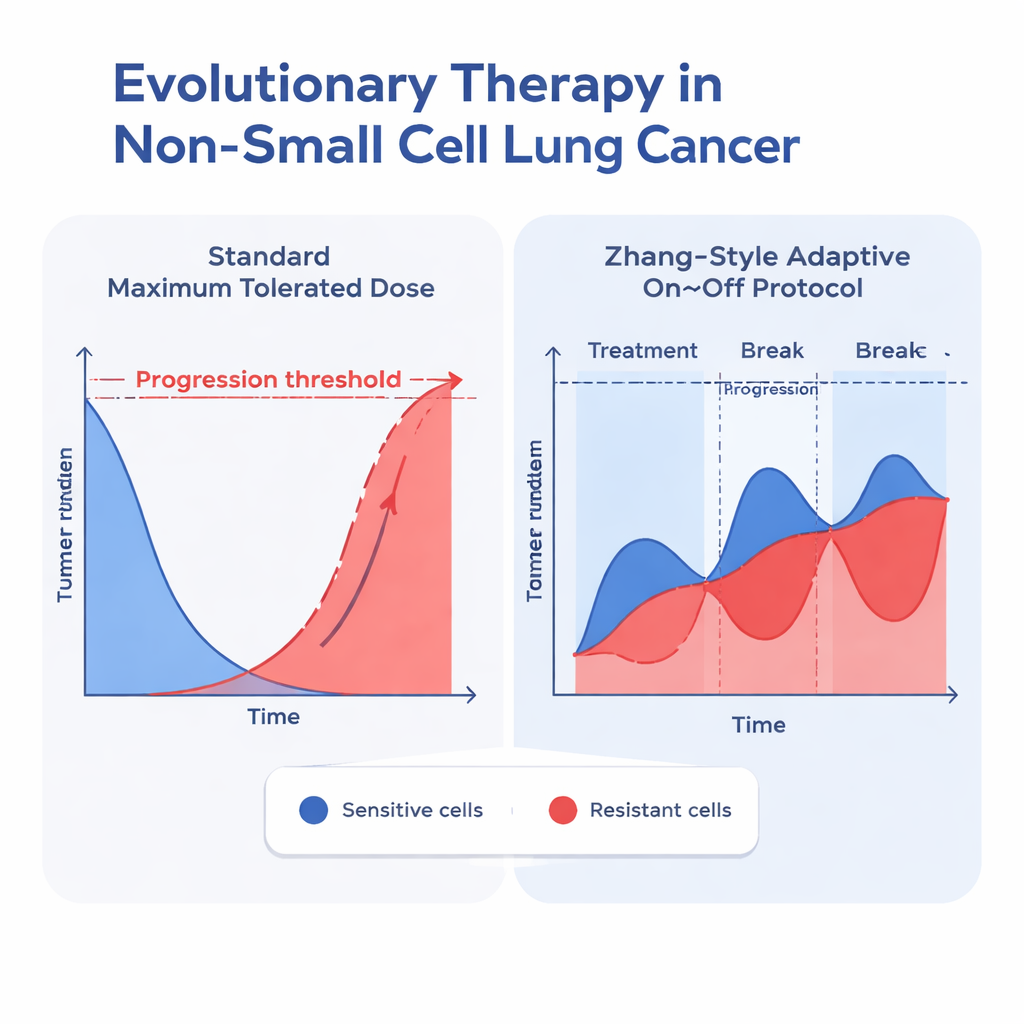
What this could mean for future patients
This work does not yet change how doctors treat NSCLC patients today, but it offers a powerful proof of concept. By grounding their models in real patient data and insisting that the models reproduce not just early shrinkage but also resistance-driven regrowth, the authors show that evolutionary therapy is theoretically viable even in a fast-growing and deadly cancer. Their results suggest that carefully timed treatment breaks might prolong control of the disease by using drug-sensitive cells as allies rather than collateral damage. Turning this idea into practice will require more data, better biomarkers such as blood tests that track tumor DNA, and rigorous clinical trials. Still, the message to lay readers is clear: sometimes, the smartest way to fight cancer is not to hit it as hard as possible, but to steer its evolution so that it remains a manageable, slower-moving foe.
Citation: Jansén-Storbacka, L.R., Honasoge, K.S., Molnárová, E. et al. Can evolutionary therapy be applied in non-small cell lung cancer?. Sci Rep 16, 7442 (2026). https://doi.org/10.1038/s41598-026-36712-x
Keywords: evolutionary therapy, non-small cell lung cancer, drug resistance, adaptive dosing, mathematical oncology