Clear Sky Science · en
Pepsin-driven corrosion of orthodontic titanium alloys in candidiasis-simulated saliva: electrochemical and statistical insights
Why this matters for braces wearers
Millions of people rely on metal brackets, wires, and mini-implants to straighten their teeth. These devices are usually made from titanium alloys that are considered safe and long‑lasting. But inside the mouth, they sit in a constantly changing mix of saliva, food acids, and microbes. This study asks a practical question with real consequences for comfort and safety: what happens to orthodontic titanium when stomach acid and enzymes reach the mouth in reflux disease, and when a common oral fungus, Candida albicans, joins the mix?
The metal used to straighten teeth
The researchers focused on a popular orthodontic metal called Ti‑6Al‑4V, a titanium alloy valued for its strength and compatibility with the body. Normally, titanium protects itself with an extremely thin but tough oxide skin that slows corrosion and limits the release of metal ions. However, the oral cavity is far from stable. Saliva contains salts, acids, enzymes, and a diverse community of microbes, and its acidity can shift dramatically from person to person. In people with gastroesophageal reflux disease (GERD), acidic stomach contents and the enzyme pepsin often reach the mouth, lowering salivary pH and potentially changing how both microbes and metals behave.
Simulating a sick mouth in the lab
To probe these conditions, the team created artificial saliva and then modified it to mimic a GERD‑affected mouth. They added pepsin, adjusted the acidity to about pH 4.9 (similar to GERD saliva), and introduced Candida albicans, a fungus responsible for a large share of oral yeast infections, especially in people wearing orthodontic devices. Tiny cylinders of Ti‑6Al‑4V were immersed at body temperature for up to 10 days in four solutions: saliva alone, saliva with pepsin, saliva with Candida, and saliva with both pepsin and Candida. Using sensitive electrochemical methods, the scientists tracked how easily current passed through the metal–solution interface, which reflects how strongly the alloy resists corrosion over time.
When a digestive enzyme protects metal
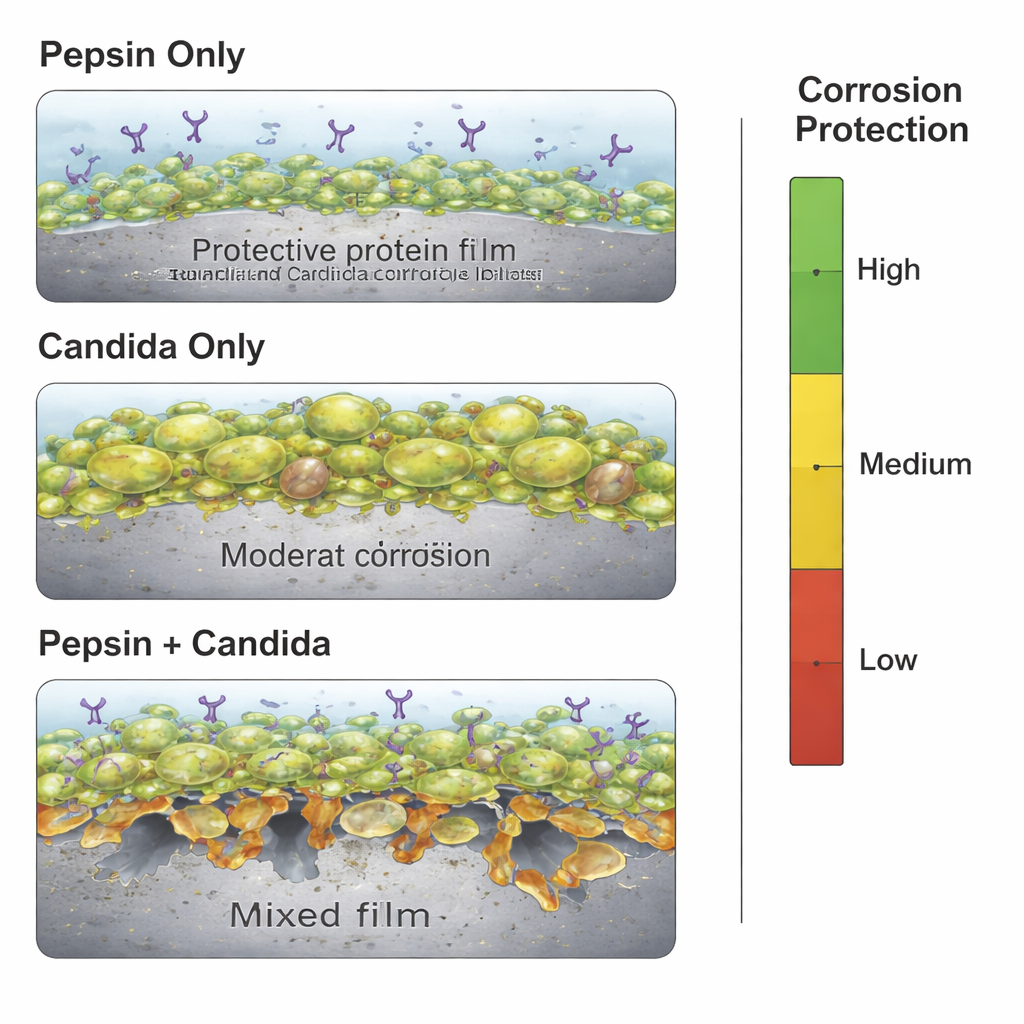
Surprisingly, pepsin by itself turned out to be a strong protector. Measurements showed that in saliva containing only pepsin, the corrosion rate of the titanium alloy dropped markedly, with protection climbing to nearly 87% after 240 hours. The data and microscopic images suggest that pepsin molecules stick to the metal surface and form a protein film, acting like a temporary clear coat that slows the arrival of aggressive ions and water. This protein layer made the metal’s electrochemical behavior more stable and shifted the corrosion potential in a safer direction, confirming that, under these simulated conditions, the digestive enzyme behaves more like a shield than an attacker.
When fungus builds and then loses a shield
Candida albicans alone also offered a degree of protection, at least at first. The fungus adhered to the titanium and secreted a sticky mix of sugars and proteins that formed a biofilm. This coating physically covered much of the surface and initially slowed corrosion, with protection exceeding 80% at certain times. But as exposure continued, that benefit faded. The fungal layer became patchy and less uniform, and corrosion resistance gradually declined to around 72%. The study shows that while microbial films can sometimes act as a barrier, their long‑term stability is uncertain, and they may eventually contribute to surface damage and metal ion release.
When allies turn into enemies
The most striking result emerged when pepsin and Candida were present together. Instead of adding their protective powers, the combination undermined them. The fungus still formed a biofilm, and pepsin still adsorbed to surfaces, but pepsin’s protein‑cutting activity began to break down the fungal matrix, creating gaps and exposing bare metal. At the same time, acidic by‑products from the microbes and fragments from the digested film intensified the chemical attack on the alloy. Electrochemical tests showed that overall protection dropped to about 56%, significantly worse than with either pepsin or Candida alone. Statistical modeling confirmed that this interaction between components—not time by itself—was the dominant factor controlling corrosion behavior.
What this means for patients and dentists
For people with braces or titanium mini‑implants who also suffer from GERD or oral yeast infections, this work delivers a clear message. Individual factors like a digestive enzyme or a fungal film can sometimes help shield dental metals, but when they interact in a GERD‑affected mouth, they may instead speed up damage. The study suggests that controlling reflux and managing oral Candida are not just comfort issues; they may be important for keeping orthodontic titanium stable and reducing the risk of early implant failure or increased metal ion release. In short, a healthy, well‑managed mouth is also a better environment for the metals that help straighten teeth.
Citation: El-Kamel, R.S., Fekry, A.M. Pepsin-driven corrosion of orthodontic titanium alloys in candidiasis-simulated saliva: electrochemical and statistical insights. Sci Rep 16, 5937 (2026). https://doi.org/10.1038/s41598-026-36707-8
Keywords: orthodontic titanium, GERD and saliva, Candida albicans, pepsin corrosion, dental implants