Clear Sky Science · en
Cholangiocarcinoma derived exosomes attenuate the anti-tumor functions of NK cells
How a Hidden Cancer Outsmarts the Immune System
Cholangiocarcinoma, a cancer of the bile ducts inside the liver, is notoriously hard to detect early and difficult to treat. This study explores a subtle tactic that these tumors use to slip past the body’s natural defenses: they send out tiny packages called exosomes that quietly weaken natural killer (NK) cells, a front-line squad of immune cells specialized in hunting and killing cancer. Understanding this stealth strategy could open new paths to therapies that restore the immune system’s ability to fight back.
The Body’s First-Responder Cancer Killers
NK cells belong to the innate immune system, the body’s rapid-response force. Unlike many immune cells that need time to recognize a specific threat, NK cells can quickly attack abnormal cells, including virus-infected and cancerous cells. They do this by latching onto targets and releasing toxic molecules such as granzyme B and signaling proteins like interferon-gamma (IFN-γ) that both kill tumor cells and rally other immune defenders. In many blood cancers and some solid tumors, boosting NK cell activity has shown promise as a treatment strategy. But in solid tumors like bile duct cancer, NK cells often appear strangely scarce and sluggish right where they are most needed.
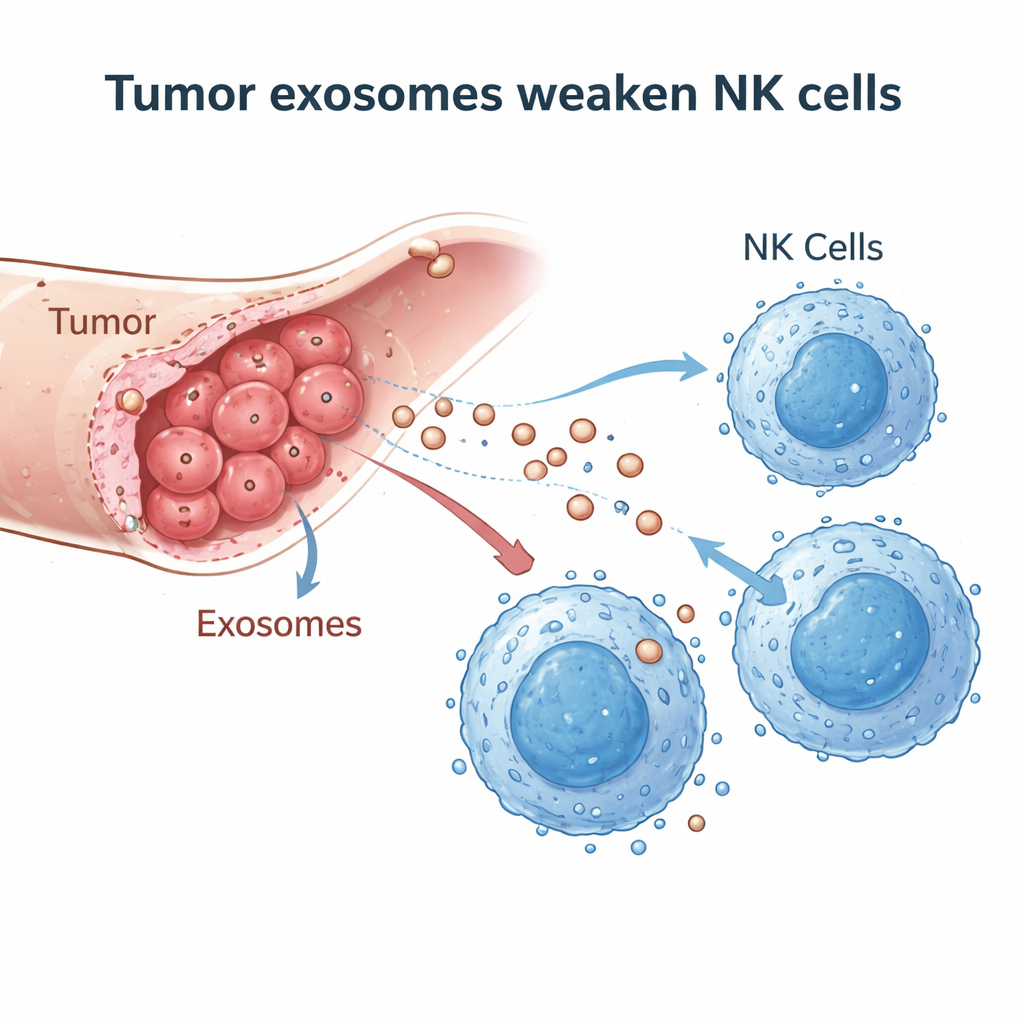
Tiny Vesicles with a Big Impact
The researchers suspected that cholangiocarcinoma cells might be sabotaging NK cells using exosomes—nano-sized membrane bubbles loaded with proteins and genetic material that cells release to communicate. They collected exosomes from a human bile duct cancer cell line and from the blood of cholangiocarcinoma patients, then confirmed their identity and size using electron microscopy, particle tracking, and standard exosome markers. In tumor tissue samples from 20 patients, they compared cancerous areas with nearby non-cancerous liver tissue and found far fewer NK cells infiltrating the tumors, suggesting that something in the tumor environment was keeping NK cells away or disabling them.
Turning Off Survival and Killing Signals
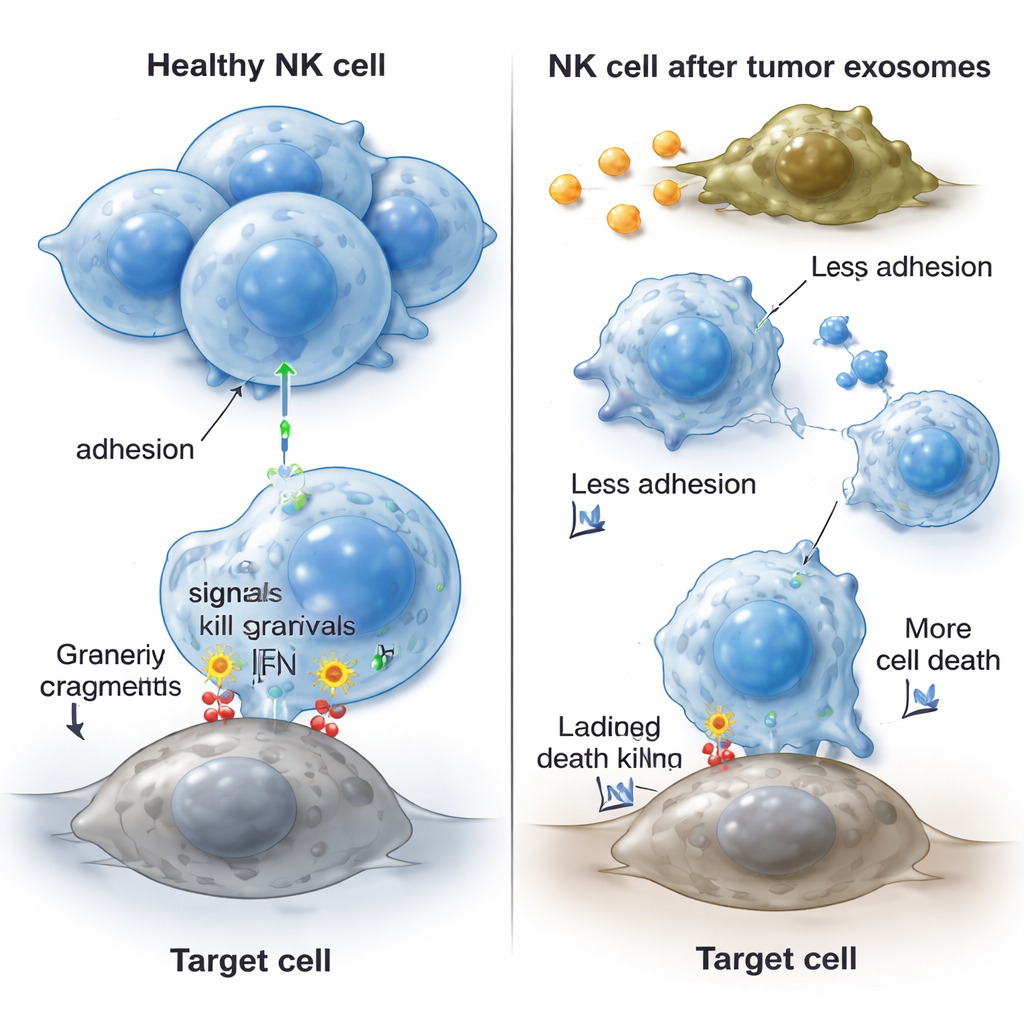
To see how these tumor exosomes affect NK cells, the team exposed an NK cell line (NK-92) to exosomes from cancer cells and from patient blood. The NK cells began to undergo higher levels of programmed cell death, especially when treated with exosomes from the tumor cell line. At the molecular level, the NK cells reduced production of key “stay alive” proteins (BCL-2 and BCL-XL), tipping the balance toward cell death. Interestingly, their overall growth and division did not change, meaning the exosomes mainly made NK cells more likely to die rather than slowing their proliferation. The exosome-treated NK cells also released less IFN-γ and less granzyme B, weakening both their ability to kill targets directly and to coordinate wider immune responses. Lab tests measuring how well NK cells could kill standard target cells showed that this killing power dropped after exposure to cholangiocarcinoma exosomes.
Breaking Up the Immune Cell Teamwork
NK cells need to stick to each other and to tumor cells to work effectively, relying on surface “adhesion molecules” that act like molecular Velcro. After treatment with cancer-derived exosomes, NK cells lost their normal clustered growth pattern and appeared more scattered. The researchers found that the levels of several important adhesion molecules (CD11a, CD18, and CD54) dropped at both the gene and protein level, while another adhesion marker, CD2, was unchanged. This loss of adhesion likely makes it harder for NK cells to reach tumors, to form stable contact with cancer cells, and to deliver lethal blows. Crucially, when the team studied NK cells directly from the blood of cholangiocarcinoma patients, those cells showed the same hallmarks: weaker killing activity and lower levels of the same adhesion molecules compared with NK cells from healthy volunteers.
What This Means for Patients and Future Treatments
Taken together, the findings suggest that cholangiocarcinoma uses exosomes as a form of molecular sabotage to evade immune attack. These tiny vesicles help push NK cells toward death, reduce their production of key killing factors, and strip away the adhesive proteins that allow them to cluster and engage tumors. The result is fewer functional NK cells in and around the tumor and a blunted immune response that lets the cancer grow more freely. While this work was largely done in controlled laboratory settings and with a limited number of patient samples, it points to new strategies: blocking tumor exosomes, restoring NK survival signals, or boosting adhesion molecules might help reawaken the immune system’s natural defenses against this deadly bile duct cancer.
Citation: Wang, X., Zhang, F., Cao, Z. et al. Cholangiocarcinoma derived exosomes attenuate the anti-tumor functions of NK cells. Sci Rep 16, 5810 (2026). https://doi.org/10.1038/s41598-026-36706-9
Keywords: cholangiocarcinoma, natural killer cells, exosomes, tumor immune escape, tumor microenvironment