Clear Sky Science · en
Bioinformatic characterization and automated detection of metabolically activated monocyte subpopulations in dyslipidemia
Why hidden immune cells matter for heart health
Cholesterol problems are usually talked about in terms of diet, numbers on a blood test, and the risk of heart attacks. But beneath those numbers, our immune system is quietly changing. This study looks at a special group of white blood cells in the blood of young adults with high LDL (“bad”) cholesterol. These cells seem to become “metabolically activated” and remember past exposure to excess fats, potentially making artery inflammation—and future heart disease—more likely. Understanding these hidden cell types could help doctors detect trouble earlier and design more precise treatments.
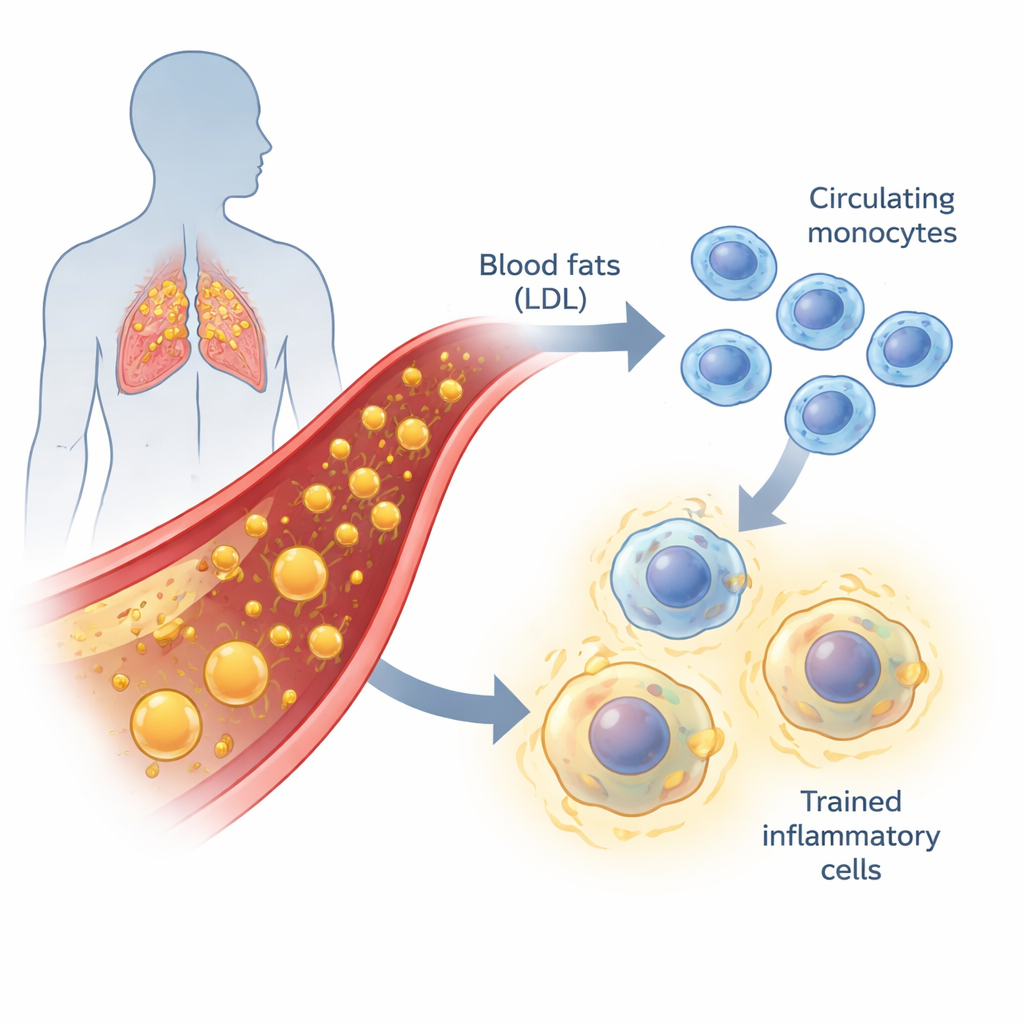
Fat in the blood and slow-burning inflammation
Atherosclerosis, the process that leads to clogged arteries, is not just about fat sticking to vessel walls. It is also a slow, chronic inflammatory disease. When LDL cholesterol is high, especially in its oxidized forms, it irritates the lining of blood vessels. Monocytes—circulating white blood cells—and the macrophages they become are drawn into these areas and start gobbling up fat. If this cleaning process is poorly regulated, the cells turn into “foam cells” and help build fatty plaques. Traditionally, scientists have divided blood monocytes into three groups based on surface markers, each with different roles in patrolling blood vessels, triggering inflammation, or maintaining tissue health.
Beyond simple labels: discovering new monocyte types
The authors suspected that some circulating monocytes in people with dyslipidemia (unhealthy blood fat levels) might already be reprogrammed into a metabolically active state, primed to become harmful macrophages. To explore this, they collected blood from 23 young adults with high LDL cholesterol and 17 healthy participants with normal lipid profiles. None were on cholesterol-lowering drugs or had other major illnesses, helping isolate the impact of dyslipidemia itself. Using a technology called flow cytometry, they measured not only classical surface markers but also proteins involved in fat handling and chemical tags on histones—tiny protein spools that help control which genes are turned on or off in each cell.
Smart algorithms to read complex immune landscapes
Instead of relying only on manual “gating” (drawing borders by eye on two-marker plots), the team built a bioinformatic pipeline that treated each cell as a point in high-dimensional space. They cleaned and standardized the data, then used t-SNE, a mathematical method that turns many-marker measurements into a two-dimensional map where similar cells cluster together. Next, they applied clustering algorithms (X-shift and FlowSOM) to automatically group cells with similar marker patterns. This allowed them to go beyond the three classic monocyte types and identify four major monocyte populations defined by combinations of fat-handling proteins (ABCA1, PLIN2, CD36) and the usual CD14 and CD16 markers.
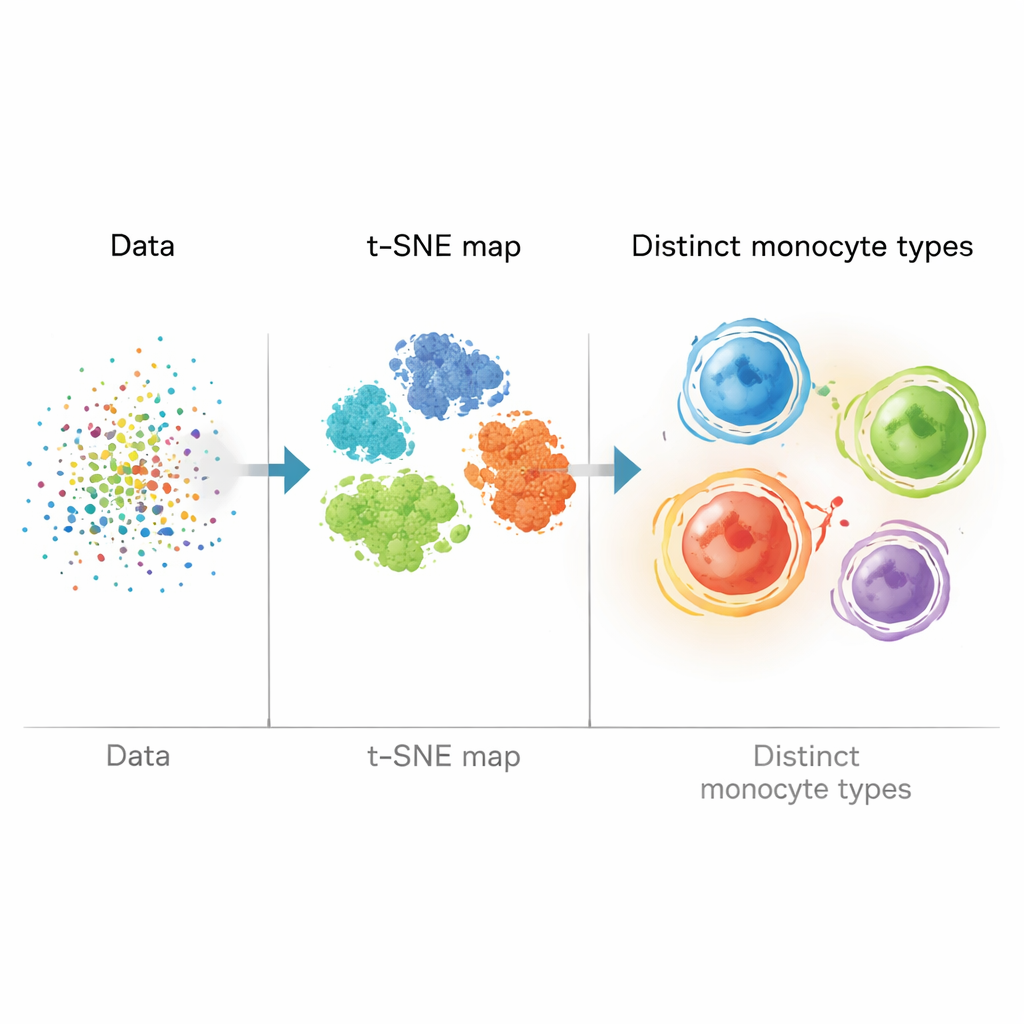
A primed inflammatory population in dyslipidemia
Two of these four monocyte groups stood out. One in particular—characterized by high CD14, lack of CD16, and strong ABCA1 with variable PLIN2 and CD36—matched the profile of “metabolically activated” monocytes (MoMe). In people with dyslipidemia, this MoMe population and another related group were significantly more abundant than in healthy controls. When the researchers examined epigenetic marks—specifically H3K4me3 and H3K27me3, which are chemical tags on histone proteins linked to gene activity—they found that MoMe cells from dyslipidemic individuals carried higher levels of H3K4me3. This mark is associated with genes that promote inflammation, suggesting these cells have a form of innate “memory,” or trained immunity, after repeated exposure to high LDL.
From circulating cells to plaque-building macrophages
To see what these monocytes might become inside tissues, the team cultured purified monocytes in a lab environment that mimicked metabolic syndrome, nudging them to turn into macrophages. They again used advanced analysis to identify macrophage subgroups and measured their epigenetic marks. Interestingly, macrophages derived from healthy donors often showed higher levels of activating histone tags than those from dyslipidemic patients, hinting that the inflammatory training may play out differently once cells leave the bloodstream. Although the sample size was modest, the findings support the idea that long-term exposure to excess blood lipids reshapes monocytes at both the protein and epigenetic levels, predisposing them to stronger inflammatory responses.
What this means for patients and future care
For a layperson, the takeaway is that high LDL cholesterol is not only a problem of “too much fat” but also of “rewired” immune cells. This study shows that even in young adults, dyslipidemia is linked to a distinct population of monocytes that appear metabolically charged and epigenetically primed to fuel artery inflammation. By combining powerful computational tools with detailed cell measurements, researchers can now spot these risky cell types in blood, long before a heart attack happens. In the future, such approaches could help doctors better gauge cardiovascular risk, monitor how well treatments are reversing harmful immune training, and ultimately design therapies that target not just cholesterol levels but the immune cells that respond to them.
Citation: Ramírez-Torres, R., Ramírez-Segovia, S.G., González-Huerta, M.J. et al. Bioinformatic characterization and automated detection of metabolically activated monocyte subpopulations in dyslipidemia. Sci Rep 16, 6170 (2026). https://doi.org/10.1038/s41598-026-36678-w
Keywords: dyslipidemia, monocytes, trained immunity, atherosclerosis, flow cytometry