Clear Sky Science · en
Application of group A streptococcal collagen-like protein 1-expressing Lactococcus as a novel immunotherapeutic against pancreatic ductal adenocarcinoma
Turning Friendly Bacteria into Cancer Fighters
Pancreatic cancer is one of the deadliest cancers, in part because it hides behind powerful defenses that shut down the body’s immune system. This study explores an unexpected ally in the fight against this disease: a common “good” bacterium from the dairy world, re‑engineered to home in on pancreatic tumors and help the immune system attack them more effectively. By borrowing a sticky surface protein from a dangerous strep germ, the researchers transformed a harmless microbe into a targeted, anti‑cancer tool in mice.
A Tough Cancer That Outsmarts the Immune System
Pancreatic ductal adenocarcinoma is notoriously lethal, with only about one in eight patients alive five years after diagnosis. One major reason is its tumor microenvironment: a dense, scar‑like shell packed with supportive cells and molecules that blunt immune attacks and block many drugs. Among the culprits are neutrophil extracellular traps, or NETs—webs of DNA and proteins that white blood cells cast into the surroundings. In pancreatic cancer, these sticky nets do more harm than good: they help tumors grow, keep killer T cells at bay, and are linked to worse outcomes. Shutting down NETs, while waking up anti‑tumor immunity, has become an appealing strategy.
Borrowing a Sticky Protein, but Not the Dangerous Germ
Earlier work showed that a surface protein from group A Streptococcus, called Scl1, can both latch onto tumor‑associated fibers and dampen NET formation, slowing pancreatic tumor growth in mice. But group A strep is also the bacterium that causes strep throat and serious invasive infections, making it unsuitable as a live therapy. To keep the helpful protein while avoiding the harmful germ, the team engineered Lactococcus lactis—a probiotic widely used in food and considered safe—to display Scl1 on its surface. This new strain, dubbed Lactococcus::620, was tested in mice bearing pancreatic tumors, using both simple flank tumors and more realistic tumors growing in the pancreas itself.
Guided Bacteria Cut Tumor Growth and Extend Survival
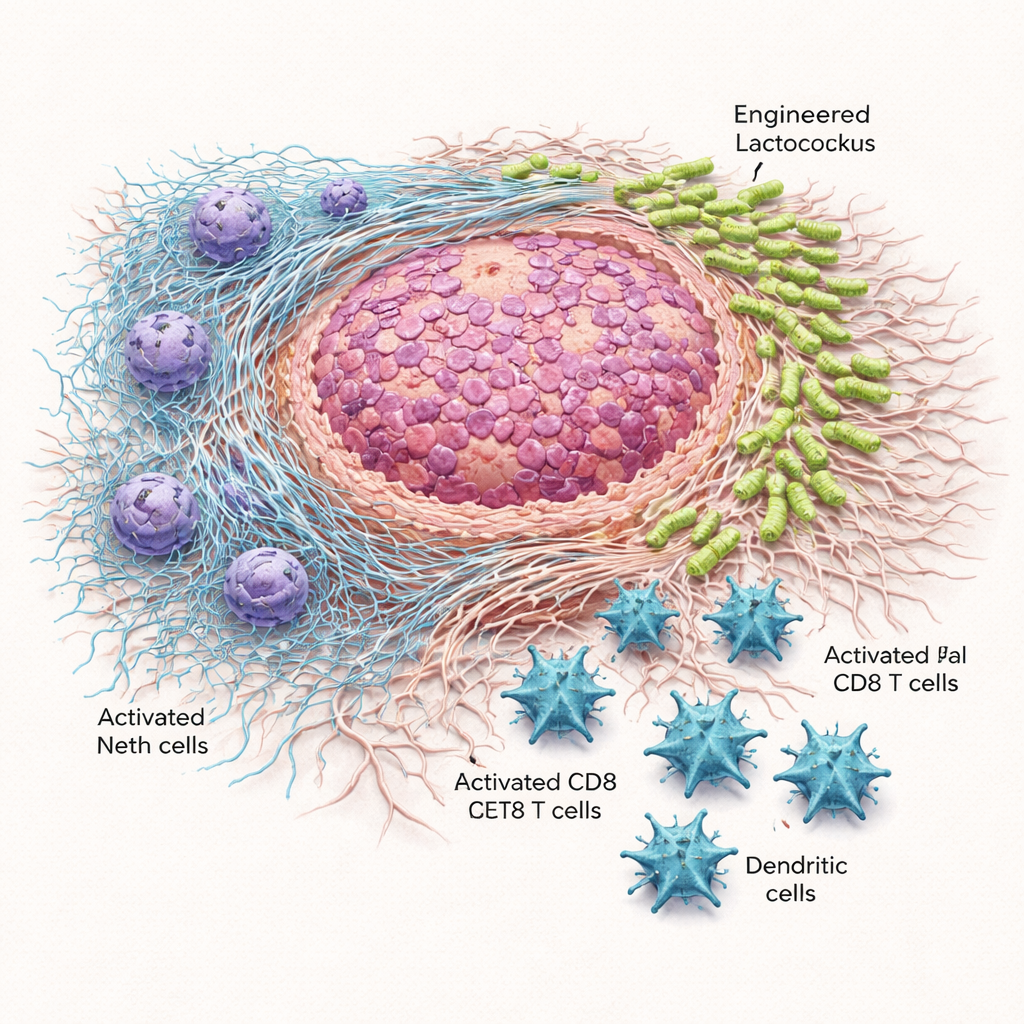
When a single dose of the engineered bacteria was injected directly into flank tumors, or delivered into the abdomen, tumors grew more slowly and weighed less than in mice given ordinary Lactococcus or a salt‑water control. In the more demanding pancreatic model, multiple doses every three days were needed, but the benefits were striking. Lactococcus::620 shrank tumors, reduced their weight, and lengthened the animals’ survival without causing treatment‑related deaths. Microscopy and culture of tumor tissue showed that the Scl1‑bearing bacteria reliably accumulated in the tumor area, especially in the fibrous matrix laid down by cancer‑associated fibroblasts, while largely sparing the spleen. In contrast, unmodified Lactococcus spread more widely in the body and was sometimes toxic when given systemically.
Disarming Harmful Nets and Letting Immune Cells In
The engineered bacteria also reshaped the immune landscape around the tumors. Tumors from Lactococcus::620‑treated mice contained more CD8 “killer” T cells and dendritic cells—key players in recognizing and destroying cancer cells—and those T cells showed fewer exhaustion markers and more activity‑related molecules. At the same time, measures of NET activity fell. In laboratory tests, neutrophils exposed to Lactococcus::620 released less free DNA and showed lower activity of myeloperoxidase, an enzyme needed to build NETs. In treated mice, circulating DNA linked to NETs was reduced. Crucially, when the same experiments were done in mice genetically unable to form NETs, the engineered bacteria no longer slowed tumor growth. That loss of benefit strongly suggests that blocking NETs is central to the therapy’s power.
What This Could Mean for Future Cancer Care
Taken together, the findings outline a new concept: using a safe, food‑grade bacterium as a programmable delivery vehicle to bring a NET‑blocking, tumor‑homing protein right into the hostile environment of pancreatic cancer. In mice, this approach lessened tumor burden, improved survival, and opened the door for immune cells to do their jobs more effectively. While much work remains before such a strategy could be tried in people—including testing in models of metastatic disease and combining it with modern immunotherapies—the study shows that carefully engineered “good” bacteria might one day help tip the balance against one of our most resistant cancers.
Citation: Godfrey, E.A., Choi, S.J., Sestito, M. et al. Application of group A streptococcal collagen-like protein 1-expressing Lactococcus as a novel immunotherapeutic against pancreatic ductal adenocarcinoma. Sci Rep 16, 5911 (2026). https://doi.org/10.1038/s41598-026-36657-1
Keywords: pancreatic cancer, probiotic therapy, tumor microenvironment, immune modulation, neutrophil extracellular traps