Clear Sky Science · en
First-principles study of X2TlAgCl6 (X = K, Rb, Cs) double perovskites for high-performance optoelectronic and thermoelectric devices
New Materials for Turning Heat and Light into Power
As the world searches for cleaner ways to power our homes and devices, scientists are hunting for materials that can efficiently turn sunlight and waste heat into electricity without relying on toxic elements. This study explores a new family of crystalline compounds, called X2TlAgCl6 (where X can be potassium, rubidium, or cesium), to see whether they could serve in next-generation solar cells and thermoelectric generators that harvest otherwise wasted heat.
The Promise of Safer Crystal Semiconductors
Many of today’s most efficient perovskite solar cells contain lead, which raises concerns about toxicity and long-term stability. The researchers focused on a group of “double perovskites,” where the crystal structure can be tuned by swapping different atoms into precise lattice positions. By replacing lead with a combination of elements including thallium, silver, and common alkali metals (K, Rb, Cs), they aimed to retain strong light- and heat-conversion performance while reducing environmental impact. Using advanced computer simulations based on quantum mechanics, they screened these materials without needing to grow them in the lab first.
Building and Stress-Testing the Crystal Framework
The first question was whether these crystals are actually stable in the forms needed for devices. The team modeled the atomic arrangement in a cubic double perovskite lattice and checked several measures of stability, including how the atoms fit together (so-called tolerance and octahedral factors), how much energy is needed to form the compound, and how the lattice vibrates. They calculated phonon spectra—essentially the allowed vibration patterns in the solid—and found that the cesium version is fully dynamically stable, while the potassium and rubidium versions show minor instabilities that are mitigated when realistic temperature effects are included. Additional molecular dynamics simulations at room temperature showed that all three compositions maintain their structure over time, suggesting they should be robust in practical conditions. Mechanical tests based on elastic constants further indicated that these crystals are not brittle, but ductile, meaning they are less likely to crack during processing.
Handling Light: Narrow-Gap Semiconductors for Near-Infrared Use
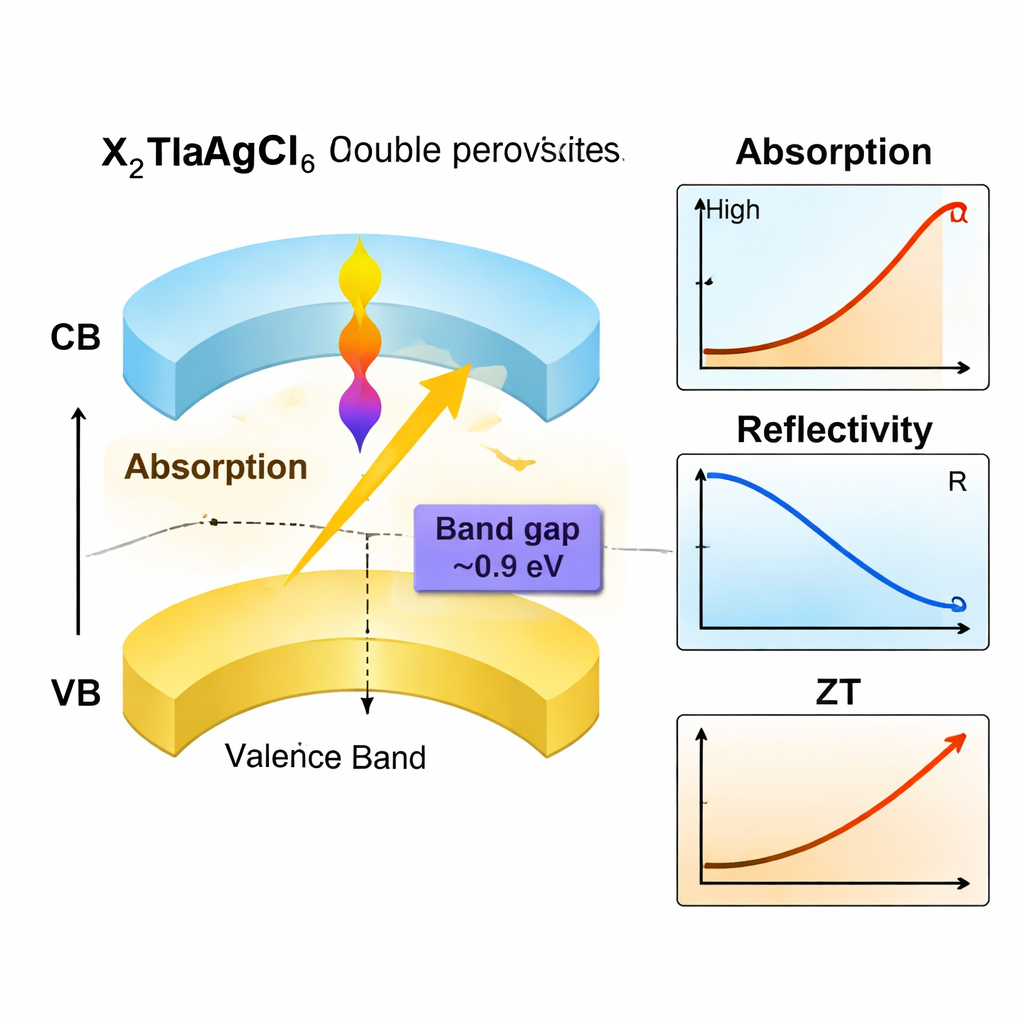
To work well in solar cells and light detectors, a material must have an energy gap that allows it to absorb light efficiently. The authors calculated the electronic band structure using several high-level methods and found that all three X2TlAgCl6 compounds are direct band gap semiconductors, an especially favorable trait for converting light into electricity. Their gaps fall around 0.9 electron volts in the most reliable scheme—significantly narrower than many other lead-free perovskites—placing them in the near-infrared range. This means they can capture lower-energy photons that standard visible-light absorbers miss. The simulations show strong optical absorption, low reflectivity, and moderate refractive index values across the visible and near-infrared spectrum, implying that thin films of these materials could efficiently soak up light while minimizing losses due to reflection.
Moving Charge and Heat: Clues from Electrical and Thermal Transport
Beyond simply absorbing light, a good energy material must move electrical charges and manage heat effectively. By examining how electrons and holes respond to electric fields, the team found that charge carriers in these crystals have relatively low effective masses—especially electrons—suggesting they can move quickly through the material. Transport calculations indicate that holes are the majority carriers, placing these compounds firmly in the p-type semiconductor category. The researchers then simulated how the materials perform as thermoelectrics, which convert heat differences directly into electric power. They found sizable Seebeck coefficients (a measure of voltage generated per degree of temperature difference), rising electrical conductivity with temperature, and a thermal conductivity that remains modest even at high temperatures. Together, these lead to a respectable thermoelectric figure of merit, ZT, approaching about 0.73 at 800 K, high enough to be technologically interesting.
From Theory to Future Devices
In everyday terms, this work identifies a new family of crystals that appear, on paper, to be both sturdy and efficient at turning light and heat into electricity, without relying on highly toxic lead. Their ability to strongly absorb near-infrared light, carry electric charge well, and maintain decent thermoelectric performance at elevated temperatures suggests they could play a role in tandem solar cells, infrared detectors, and waste-heat recovery modules. Although these predictions are based on first-principles calculations rather than finished devices, they provide a roadmap for experimental teams to synthesize X2TlAgCl6 materials and test them in real-world energy technologies.
Citation: Shah, S.H., Alomar, M., Al Huwayz, M. et al. First-principles study of X2TlAgCl6 (X = K, Rb, Cs) double perovskites for high-performance optoelectronic and thermoelectric devices. Sci Rep 16, 6324 (2026). https://doi.org/10.1038/s41598-026-36650-8
Keywords: lead-free perovskites, thermoelectric materials, optoelectronics, solar energy conversion, waste heat harvesting