Clear Sky Science · en
Antibacterial, antibiofilm, and metabolomic profiling of the novel freshwater fungi Longipedicellata megafusiformis and Wicklowia fusiformispora
Hidden allies in the fight against superbugs
As doctors struggle with infections that no longer respond to our usual antibiotics, scientists are searching for new weapons in unexpected places. This study turns to decaying wood lying in tropical rivers and discovers two little-known freshwater fungi that make a rich cocktail of natural chemicals able to slow dangerous bacteria and block the slimy films that help them survive on medical devices and tissues.
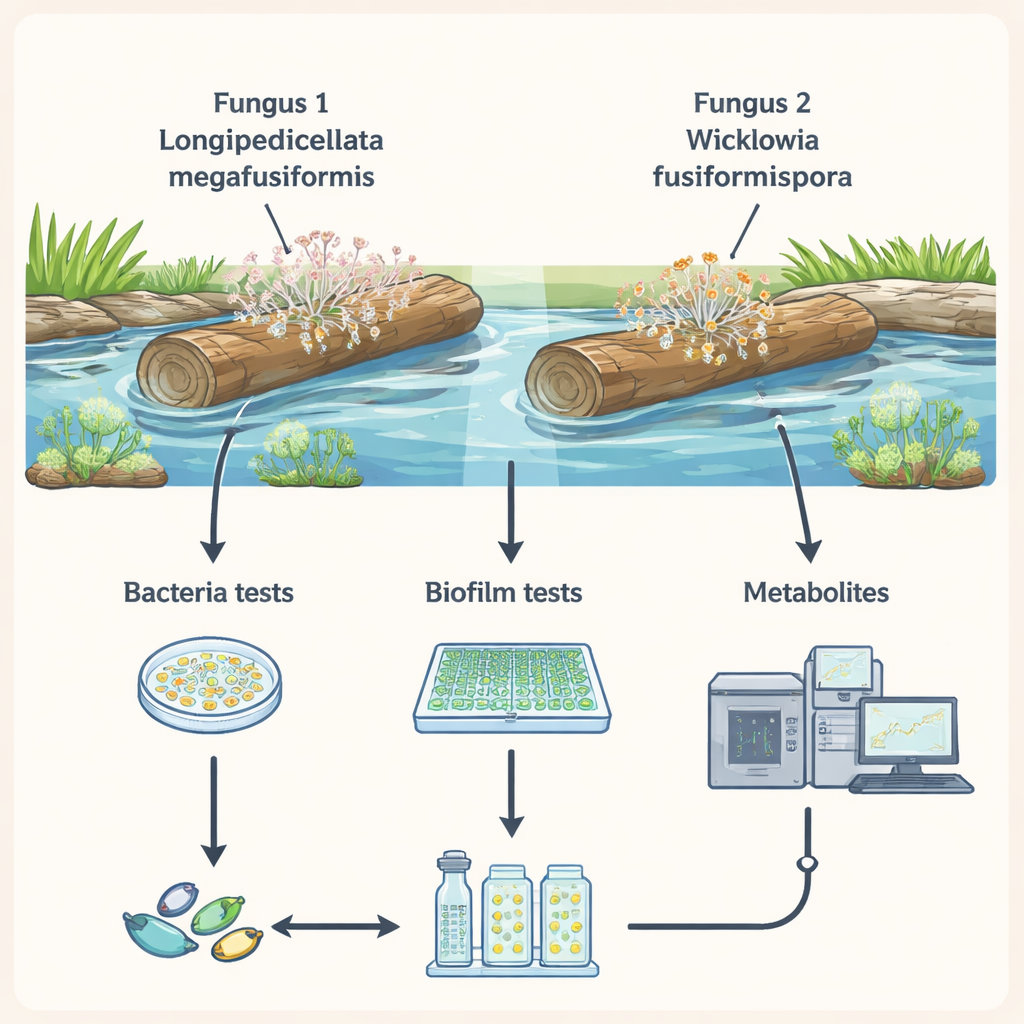
River wood as a pharmacy
In warm streams of northern Thailand, fungi quietly break down fallen branches, recycling nutrients back into the ecosystem. The researchers isolated two newly described fungi from submerged wood and grew them in the lab. They were interested in the “secondary metabolites” these fungi release—small molecules not needed for basic survival, but used as chemical defenses and tools in their microscopic battles with neighboring microbes. Because freshwater fungi have been far less studied than their land or marine cousins, they may hold untapped collections of useful antibiotic-like substances.
Putting fungal brews to the test
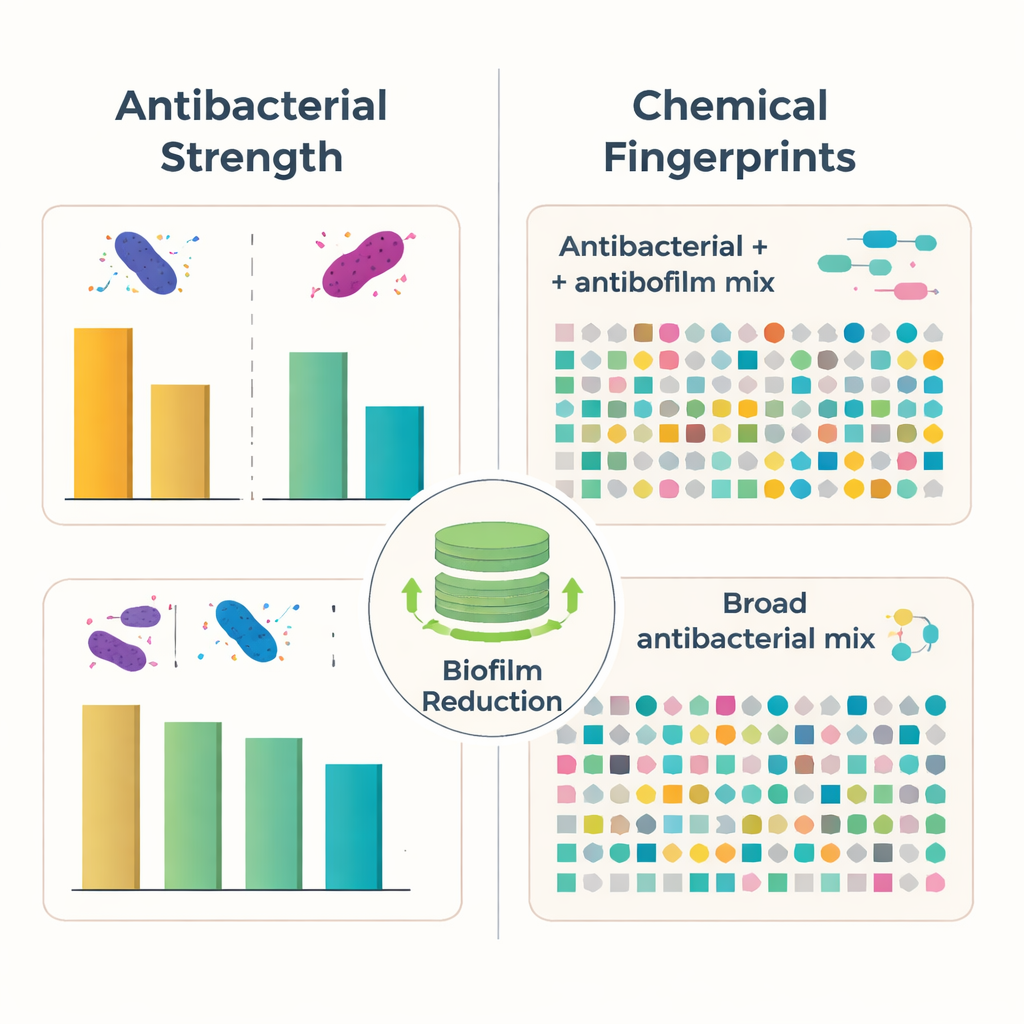
The team extracted mixtures of chemicals from the fungal growth using a common solvent and tested them against four well-known bacterial troublemakers, including Staphylococcus aureus and Escherichia coli. On petri dishes, all fungal extracts produced clear, bacteria-free zones, showing that the mixtures could halt growth of both “gram-positive” and “gram-negative” bacteria. When the scientists measured the lowest dose needed to stop growth in liquid culture, both fungi showed activity in the microgram-per-milliliter range—strong enough to be scientifically interesting, even if not yet ready to rival standard drugs like ampicillin.
Breaking up bacterial strongholds
Because many persistent infections depend on biofilms—sticky, layered communities that protect bacteria from antibiotics—the researchers also asked whether the fungal mixtures could interfere with this shield. In a plate-based test, they allowed bacteria to start forming biofilms in the presence of the fungal extracts and then stained the remaining slime. Both fungi reduced biofilm build-up by roughly half or more at the tested dose, with some combinations cutting it by over 70%. These results suggest that the natural mixtures can weaken the early stages of biofilm formation, a promising trait for preventing infections on catheters, implants, and other medical surfaces, even though mature, well-established biofilms were not examined here.
Peeking into the chemical toolbox
To understand what might be behind these effects, the team used high-resolution liquid chromatography–mass spectrometry, a technique that separates and weighs molecules with great precision, to sketch a “chemical fingerprint” for each fungus. One species, Longipedicellata megafusiformis, carried at least 27 distinct compounds, including alkaloids, peptides, pigment-like molecules, and other structures already known from the scientific literature to harm bacteria or disrupt biofilms. The other, Wicklowia fusiformispora, produced at least 33 different compounds, forming a broad antibacterial blend, but with no individual molecules yet proven to act directly on biofilms. In both fungi, the chemical diversity points to a complex strategy: rather than relying on a single silver bullet, they deploy many related and unrelated molecules at once.
What this means for future medicines
For non-specialists, the key message is that ordinary-looking river fungi can manufacture sophisticated chemical mixtures capable of slowing harmful bacteria and undermining their protective slime layers. The study does not claim to have found a ready-made drug; instead, it maps where the most promising molecules may be hiding and shows that whole extracts already have meaningful antibacterial and anti-biofilm effects. By combining classic lab tests with modern chemical profiling, the work lays a roadmap for future efforts to isolate, refine, and understand the most potent ingredients—steps that could ultimately turn these quiet river dwellers into valuable allies against antibiotic-resistant infections.
Citation: Khruengsai, S., Sripahco, T., Kittakoop, P. et al. Antibacterial, antibiofilm, and metabolomic profiling of the novel freshwater fungi Longipedicellata megafusiformis and Wicklowia fusiformispora. Sci Rep 16, 6083 (2026). https://doi.org/10.1038/s41598-026-36637-5
Keywords: freshwater fungi, antibiotic resistance, biofilms, natural products, antibacterial compounds