Clear Sky Science · en
Interaction mechanisms between liquid organic matter and solid bitumen
Why this buried carbon matters
Deep beneath our feet, in the tight pores and cracks of shale rocks, a solid, tar‑like material called bitumen quietly holds and releases oil and gas. This hidden substance does more than just sit in the rock: it can soak up certain liquid hydrocarbons and let others go, helping to decide how much oil and gas ultimately reach a well. Understanding exactly how this solid bitumen grabs onto different liquid molecules could improve how we explore for hydrocarbons and how efficiently we produce them.
A hidden sponge in the rock
Solid bitumen forms when once‑fluid organic matter—ancient kerogen and crude oil—is cooked and altered over millions of years. As it becomes more concentrated and carbon‑rich, it ends up filling tiny pores and micro‑fractures in source rocks. In these cramped spaces, bitumen can block pathways for fluids, but it can also act like a molecular sponge, selectively binding and storing different components of oil. Because of this dual role, bitumen affects both the quality of a reservoir and the types of hydrocarbons that can be produced from it, making it a key but often overlooked player in subsurface energy systems.
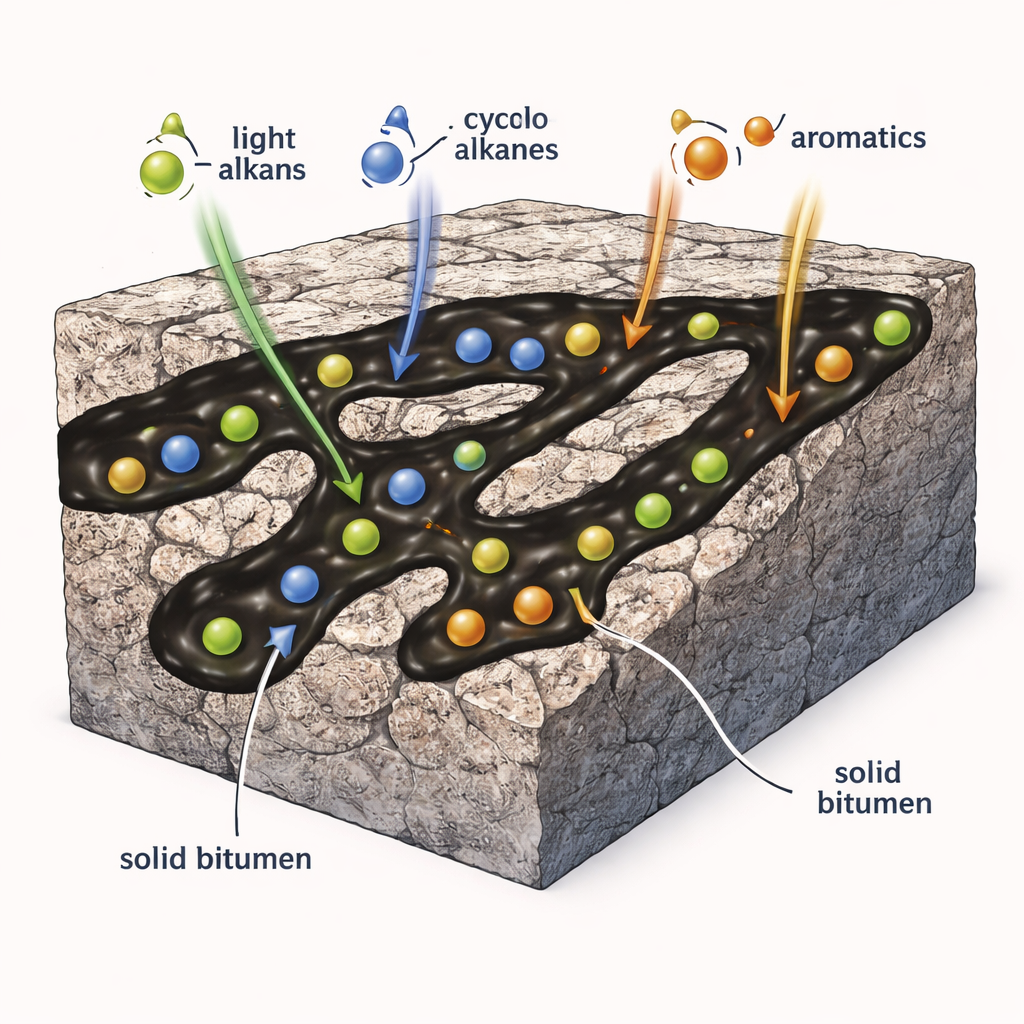
Using virtual experiments to watch molecules stick
Directly watching individual oil molecules interact with solid bitumen is nearly impossible in the lab, so the authors turn to computer simulations. They started with a well‑characterized sample of natural solid bitumen from the Sichuan Basin in southwest China. Laboratory heating experiments mimicked the rock’s geological “cooking” history, creating a series of samples from low to very high thermal maturity. Detailed chemical analyses and carbon‑13 NMR measurements were then used to build realistic three‑dimensional molecular models of the bitumen at each stage. With these models in hand, the team applied a tool borrowed from drug design—molecular docking—to calculate how strongly different liquid hydrocarbons would bind to bitumen surfaces, using changes in Gibbs free energy as a measure of how favorable each interaction is.
Which liquid molecules does bitumen like best?
The researchers tested a wide range of hydrocarbon types, including straight‑chain alkanes, branched alkanes, ring‑shaped cycloalkanes, simple aromatics like benzene, larger polycyclic aromatic hydrocarbons (PAHs), and PAHs with extra methyl side groups. Across this chemical spectrum, all classes showed some tendency to stick to solid bitumen, but with large differences in strength. Aromatic molecules generally bound more strongly than saturated ones, and ring‑containing cycloalkanes outperformed straight‑chain alkanes of similar size. Within each family, heavier molecules—those with more carbon atoms—tended to be held more tightly. In many cases, extra methyl groups increased binding even further, suggesting that small changes in molecular “decoration” can noticeably shift how hydrocarbons partition between rock‑bound and mobile phases.
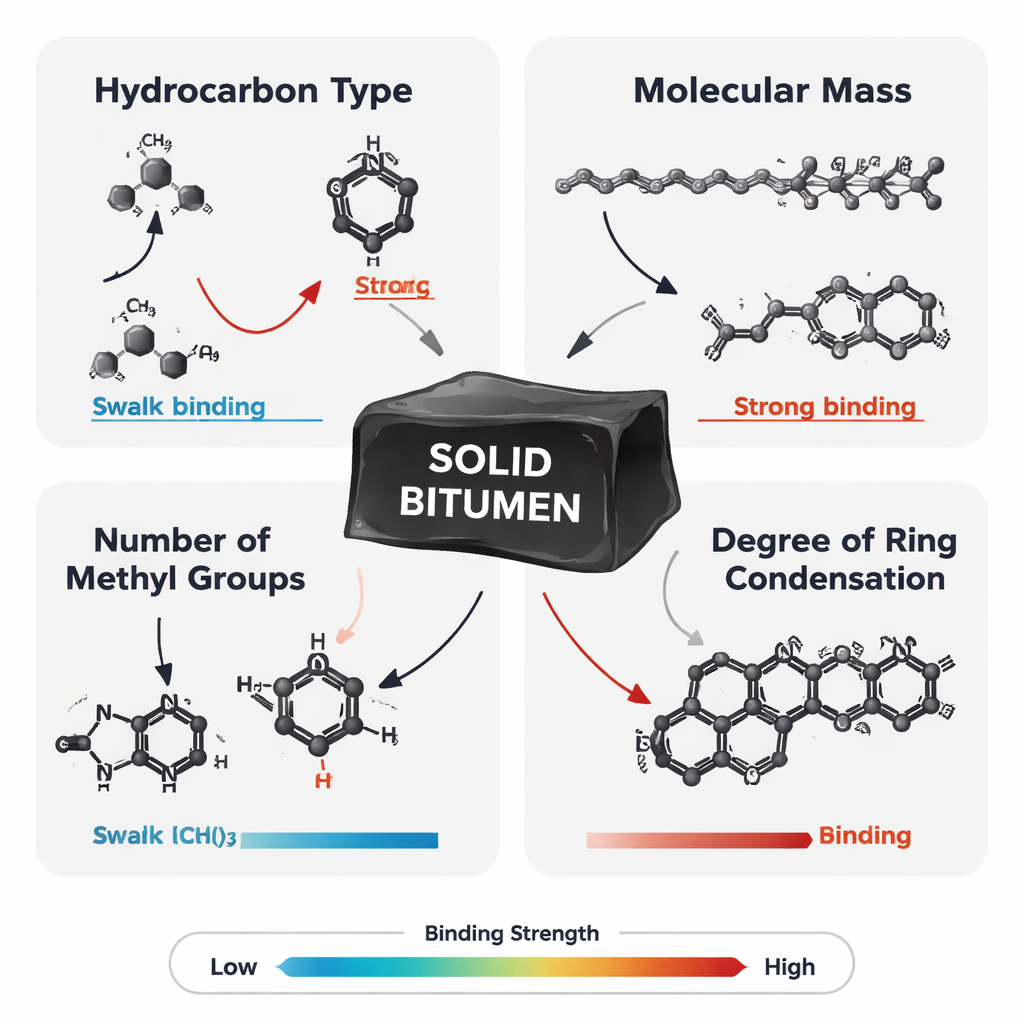
When structure matters more than size
Beyond simple size, the shape and compactness of aromatic molecules emerged as a crucial control. The study used a parameter called condensation degree to describe how tightly fused the aromatic rings are. Comparing molecules with the same number of rings but different connection patterns, the team found that linearly fused PAHs often adhered more strongly than highly condensed or polymer‑linked versions, even when their masses were similar. This means that not all “heavy aromatics” behave alike: subtle differences in ring arrangement can tilt the balance between staying locked in solid bitumen and being released into flowing oil. Surprisingly, the simulations did not show a consistent decrease in adsorption as the bitumen itself became more mature and more aromatic, contradicting the authors’ initial hypothesis and highlighting the complex interplay of molecular structure on both sides of the interface.
Implications for oil generation and recovery
Taken together, the results suggest that four simple factors—hydrocarbon type, molecular mass, methyl content, and ring condensation—jointly control how strongly liquid hydrocarbons interact with solid bitumen. During the earliest stages of oil generation, this selectivity means that lighter, smaller molecules and modestly sized aromatics are more likely to escape first, enriching early oils in mobile, light components. At later stages, heavier and more aromatic molecules, especially those with multiple rings and methyl groups, tend to remain trapped in or near the bitumen. For petroleum engineers, these insights hint at new strategies: for example, designing injection fluids rich in tailored aromatic polymers that can compete for binding sites and help dislodge tightly held aromatic hydrocarbons. In simple terms, this work shows that the microscopic handshake between bitumen and oil molecules is far from random—and that learning its rules could help us coax more usable energy out of rocks while better predicting what kinds of fluids they can yield.
Citation: Lin, X., Liang, T., Zou, Y. et al. Interaction mechanisms between liquid organic matter and solid bitumen. Sci Rep 16, 5839 (2026). https://doi.org/10.1038/s41598-026-36636-6
Keywords: solid bitumen, hydrocarbon adsorption, molecular docking, shale reservoirs, aromatic hydrocarbons