Clear Sky Science · en
Surface markers on supermeres outperform extracellular vesicles in colorectal cancer diagnosis
Why tiny blood particles could transform cancer checks
Colorectal cancer is one of the most common cancers worldwide, and doctors urgently need better blood tests that can catch it early and track whether treatment is working. This study looks at a newly recognized class of ultra-small particles in the blood, called “supermeres,” and shows that they may carry clearer cancer signals than better-known particles. The authors also unveil a rapid sensor that can measure these supermeres directly from a tiny drop of blood in under half an hour, potentially opening the door to faster, more precise cancer diagnostics.
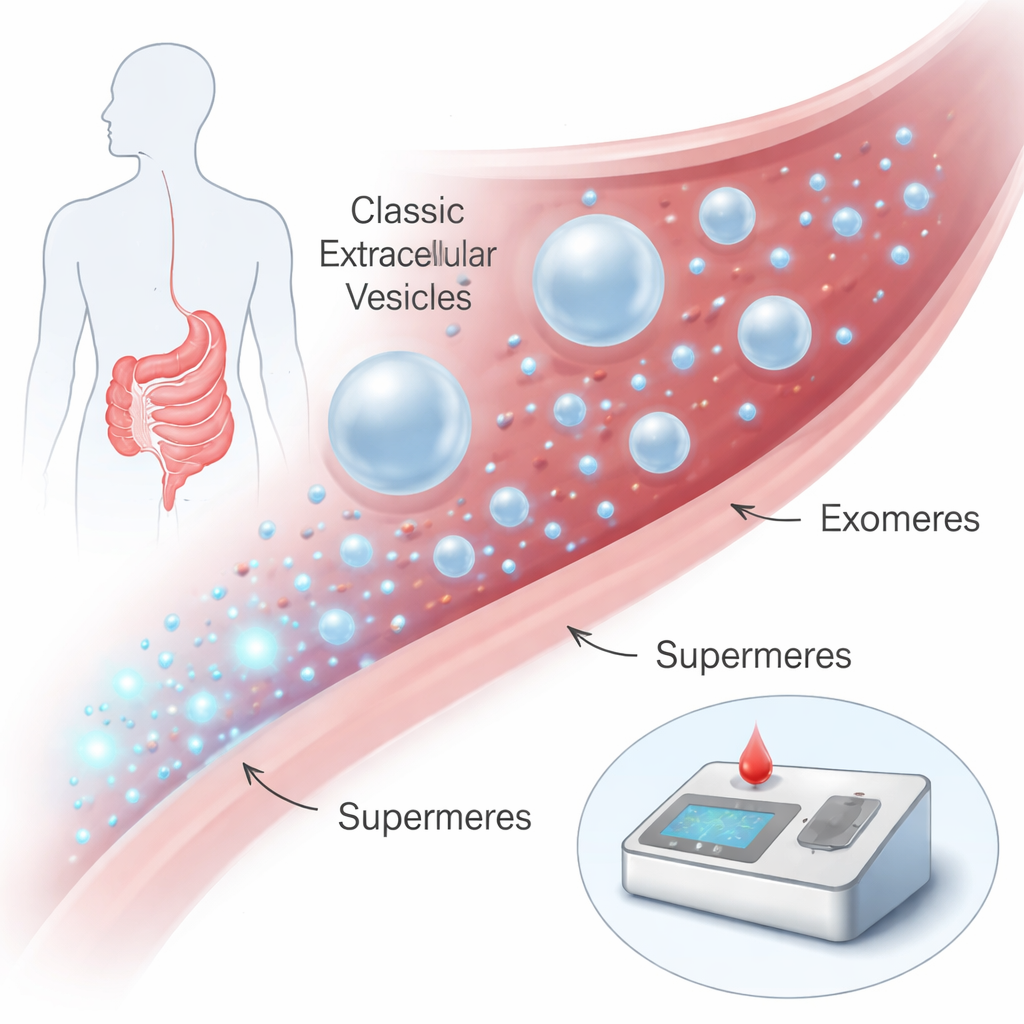
Tiny messengers in the bloodstream
Our cells constantly release nanosized packages into the fluids of the body, such as blood. For years, most attention has focused on membrane-bound particles like exosomes and other extracellular vesicles, which can ferry proteins and genetic material between cells and have been explored as disease biomarkers. More recently, researchers discovered even smaller, membrane-free particles called exomeres and, smaller still, supermeres. Supermeres are only about 15–25 nanometers across—thousands would fit across the width of a human hair—yet they are packed with disease-related molecules. Earlier work hinted that supermeres might be especially rich in cancer signals, but they were very hard to isolate and study, requiring days of ultracentrifugation with specialized equipment.
What makes supermeres stand out
The team carefully separated three major types of nanoparticles from cancer cell cultures and human blood: small extracellular vesicles, exomeres, and supermeres. They confirmed that each group has a characteristic size range, with supermeres the smallest. The key difference, however, was electrical. Supermeres carried a much stronger negative surface charge than the other particles—about twice the level at which random thermal motion becomes important—due mostly to strands of RNA clinging to their surface. When the researchers digested these RNAs with enzymes, the charge dropped sharply, showing that surface RNA is central to supermeres’ electrostatic signature. Protein mapping revealed that supermeres also have their own “ID tags”: proteins such as HSPA13, ENO2, and DDR1 were highly enriched on supermeres but largely absent from the other particle types, much like classic markers that identify ordinary extracellular vesicles.
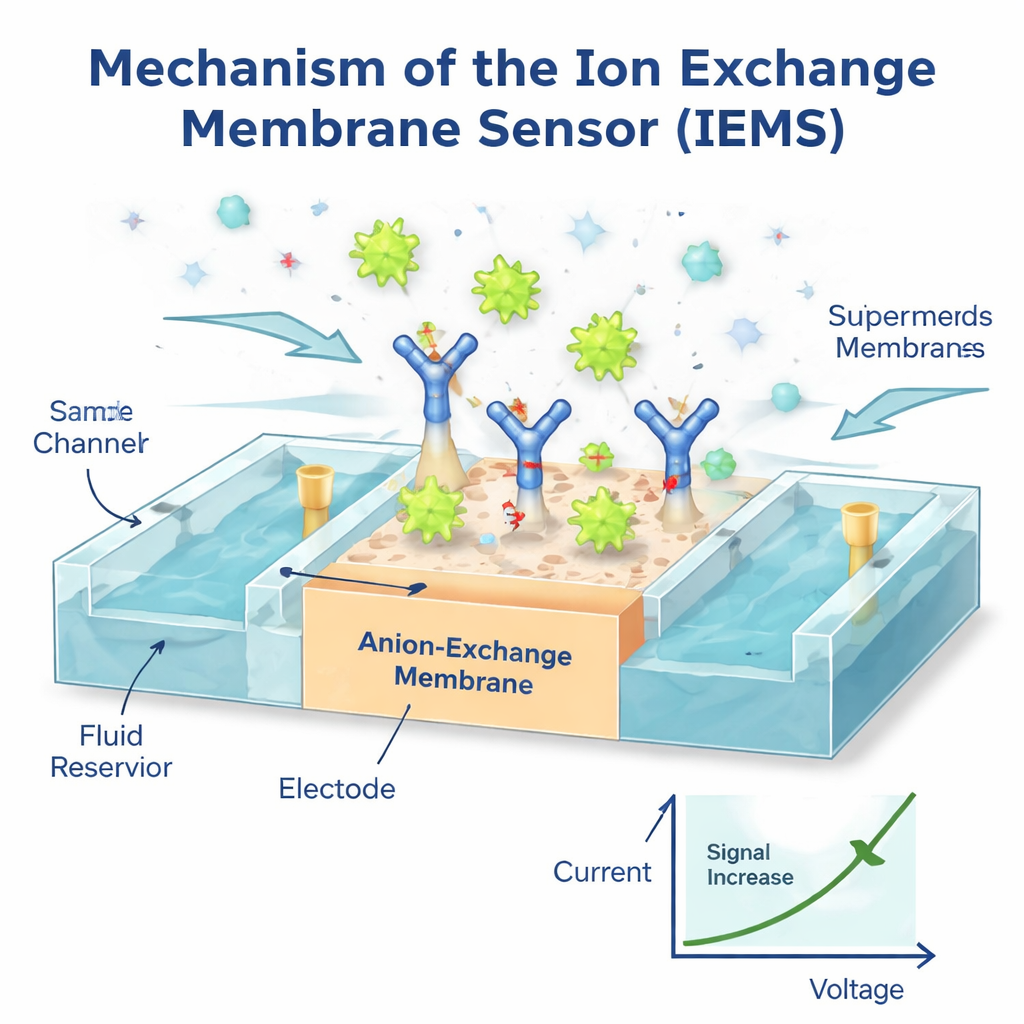
A fast sensor that listens to charge
To exploit these unique properties, the authors built an Ion Exchange Membrane Sensor (IEMS), a tiny device that senses changes in electric current as charged particles bind to its surface. They coated a special membrane with antibodies that grab a chosen protein, such as a cancer-related marker, from a flowing blood sample. Because supermeres are so highly charged while most other particles and free proteins are weakly charged, only captured supermeres noticeably alter the current–voltage behavior of the membrane. The result is a strong, selective electrical signal that reflects the number of supermeres carrying that marker. Using this setup, the team could detect as few as one million supermeres per milliliter over a range of a thousand-fold in concentration, all within about 30 minutes and using only 50 microliters of sample—no lengthy purification required. Tests where known amounts of supermeres were added to healthy plasma showed nearly complete recovery, and measurements matched those from slower, high-end optical instruments.
Outperforming existing nanoparticle markers
Armed with both traditional and rapid methods, the researchers compared supermeres head-to-head with exomeres and small extracellular vesicles in blood from people with colorectal cancer and healthy volunteers. They focused on several proteins that appear on all three particle types, including well-known tumor markers like CEA and GPC1. For nearly every shared protein, the signal carried by supermeres did a better or equal job of distinguishing cancer patients from healthy individuals, while exomeres often showed little diagnostic value. Supermere-specific proteins such as HSPA13, ENO2, and DDR1 were especially telling: these were barely detectable in healthy plasma but strongly elevated in colorectal cancer, providing excellent separation between the two groups. Strikingly, in two patients studied before and after tumor surgery, levels of supermeres dropped sharply after tumor removal, whereas signals from traditional vesicles changed very little, suggesting that supermeres closely track tumor burden.
What this could mean for future cancer tests
To put their new sensor to the test under realistic conditions, the team used IEMS to measure supermeres carrying different markers in plasma from colorectal cancer, pancreatic cancer, glioblastoma, and healthy donors. The rapid, isolation-free readings closely matched those from the much slower ultracentrifugation plus optical analysis, confirming that speed did not come at the cost of accuracy. In colorectal cancer, supermere-based measurements outperformed total blood levels of CEA, a long-standing clinical marker. Together, the findings suggest that supermeres are not just another microscopic curiosity but a powerful and accessible source of cancer information. If validated in larger and more diverse patient groups, measuring marker-bearing supermeres with compact, charge-based sensors could become a practical way to screen for colorectal cancer, monitor treatment, and possibly tailor therapies with far less time, sample volume, and complexity than today’s methods.
Citation: Kumar, S., Sinclair, J.A., Shi, T. et al. Surface markers on supermeres outperform extracellular vesicles in colorectal cancer diagnosis. Sci Rep 16, 5989 (2026). https://doi.org/10.1038/s41598-026-36626-8
Keywords: colorectal cancer, blood biomarkers, extracellular nanoparticles, supermeres, biosensor technology