Clear Sky Science · en
The morpho-kinetic landscape of macrophage modes during wound healing in zebrafish
Why tiny wound healers matter
Every cut or scrape on our skin sets off a complicated repair job that our bodies usually handle without us noticing. Central to this process are macrophages—roaming immune cells that clear debris, fight germs, and help tissues rebuild. This study uses transparent zebrafish larvae and advanced imaging to watch thousands of individual macrophages in action, revealing how their shapes and movement patterns shift over time as a wound heals. Understanding these subtle behaviors could one day help doctors fine-tune inflammation and improve healing in chronic wounds and other diseases.
Immune cells on the move
Macrophages are often described in two broad modes. Early after injury, “M1-like” cells spark inflammation, kill microbes, and devour debris. Later, “M2-like” cells help calm inflammation and support tissue repair. But in living tissue this switch is not a simple on–off change. Using zebrafish tail fin injuries, the researchers filmed macrophages in real time and used computer tools to follow each cell’s path and outline. From these time-lapse movies they extracted 63 different measurements describing how round or elongated each cell was, how fast it moved, how straight or winding its path became, and how it moved relative to the wound.
Different jobs, different motion styles
When the team compared early M1-like macrophages with later M2-like ones, they found clear behavioral differences. M1-like cells were generally rounder and moved in straighter, more directed paths that steered them toward the wound and kept them close by. M2-like cells, in contrast, were more elongated, wandered in looped or meandering paths, and tended to stay farther from the injury. The researchers quantified “random-like” motion by looking for paths that crossed over themselves; M2-like cells spent more of their time in these looping patterns, consistent with a more exploratory or less tightly guided role once the main inflammatory phase has passed.
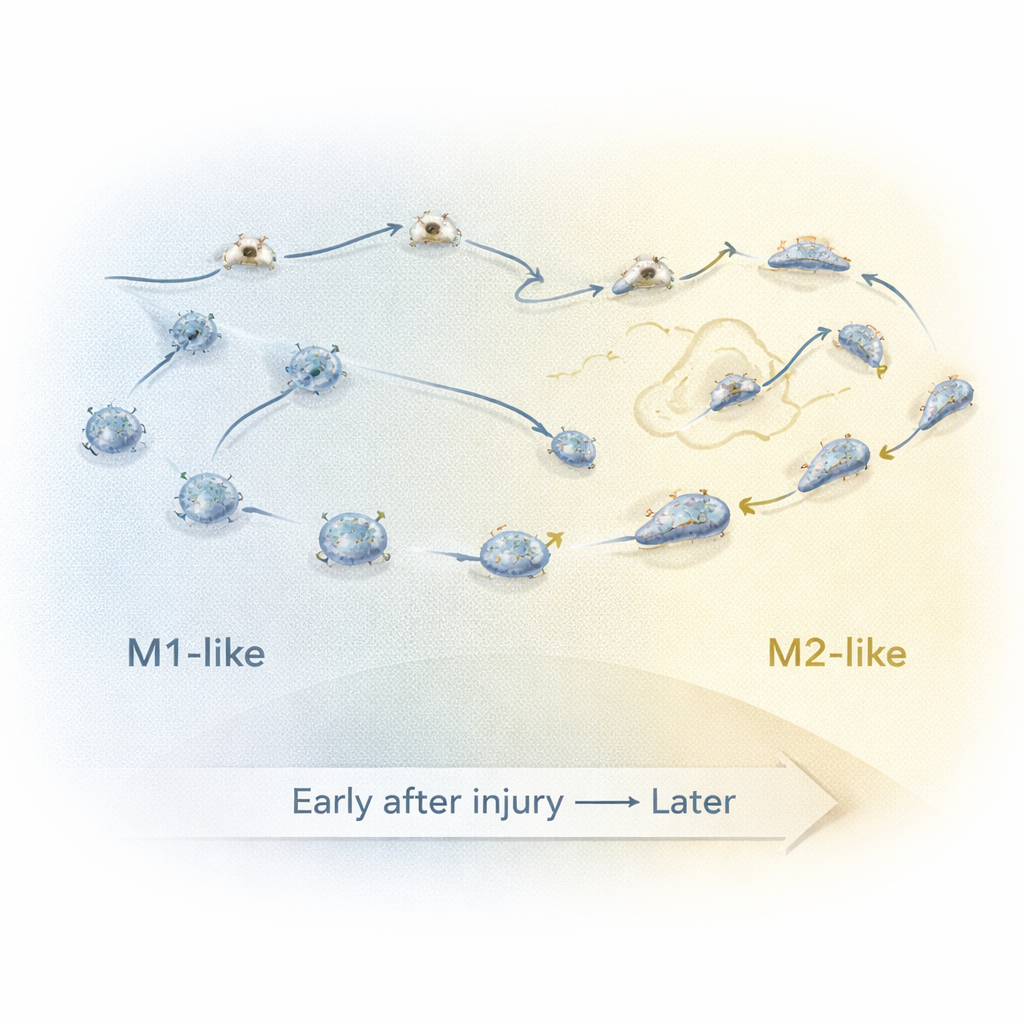
Catching cells in mid-switch
To understand what happens between the early and late phases, the researchers focused on a “transition period” several hours after injury. They trained a computer classifier on the detailed movement and shape features of clearly early (M1-like) and late (M2-like) macrophages, then used it to label cells recorded during this middle window as either “classified M1-like” (cM1-like) or “classified M2-like” (cM2-like). By grouping the data into 20-minute slices, they could track how key features changed over time. Around 7.5 to 9 hours after injury, cells with M2-like behavior began to lose their directed movement toward the wound, then actively migrated away while becoming more elongated and briefly speeding up. This time window likely marks when many macrophages switch from inflammatory fighters to repair supporters.
Macrophages without a clear alarm
The study also examined two additional groups: cells that did not turn on a key inflammatory signal (TNF) near a wound, and macrophages in completely unwounded fish. The non-TNF cells near an injury looked and moved more like early M1-like cells, but they stayed farther from the wound and slowed down over time, suggesting that strong inflammatory activation is tied to being physically close to the damaged tissue. In unwounded fish, macrophages drifted slowly with no strong sense of direction, often following curved, random paths. Their shapes were rounder and their speeds lower than any wounded state, reflecting a quiet, “patrolling” mode rather than an emergency response.
What this means for healing
By turning movies of living immune cells into numbers, this work maps out a “behavioral landscape” for macrophages as a wound progresses from early inflammation to resolution. It shows that how these cells look and how they move are tightly linked to what they are doing—racing straight to a wound, looping as they remodel tissue, or quietly wandering in healthy tissue. Such quantitative fingerprints could help scientists spot when healing goes off track, design therapies that nudge macrophages into helpful modes, and better understand inflammatory diseases where this balance is disturbed.
Citation: Park, S.A., Lupi, G., Ozbilgic, R. et al. The morpho-kinetic landscape of macrophage modes during wound healing in zebrafish. Sci Rep 16, 6506 (2026). https://doi.org/10.1038/s41598-026-36624-w
Keywords: wound healing, macrophages, zebrafish, cell migration, inflammation