Clear Sky Science · en
Long-term in vivo pharmacokinetics of dexamethasone-loaded cochlear implant electrode carrier dummies with optimized release profiles
Making High-Tech Hearing Implants Gentler on the Ear
Cochlear implants can restore hearing to people with severe hearing loss, but placing the tiny electrode inside the delicate inner ear still causes injury and inflammation. That early damage can erase any remaining natural hearing and may reduce how well the implant works over time. This study explores a smarter way to build drug-releasing dummy electrodes—stand-ins for real implant arrays—that slowly leak a steroid medicine into the inner ear, aiming to calm inflammation right where it starts while using as little drug as possible.
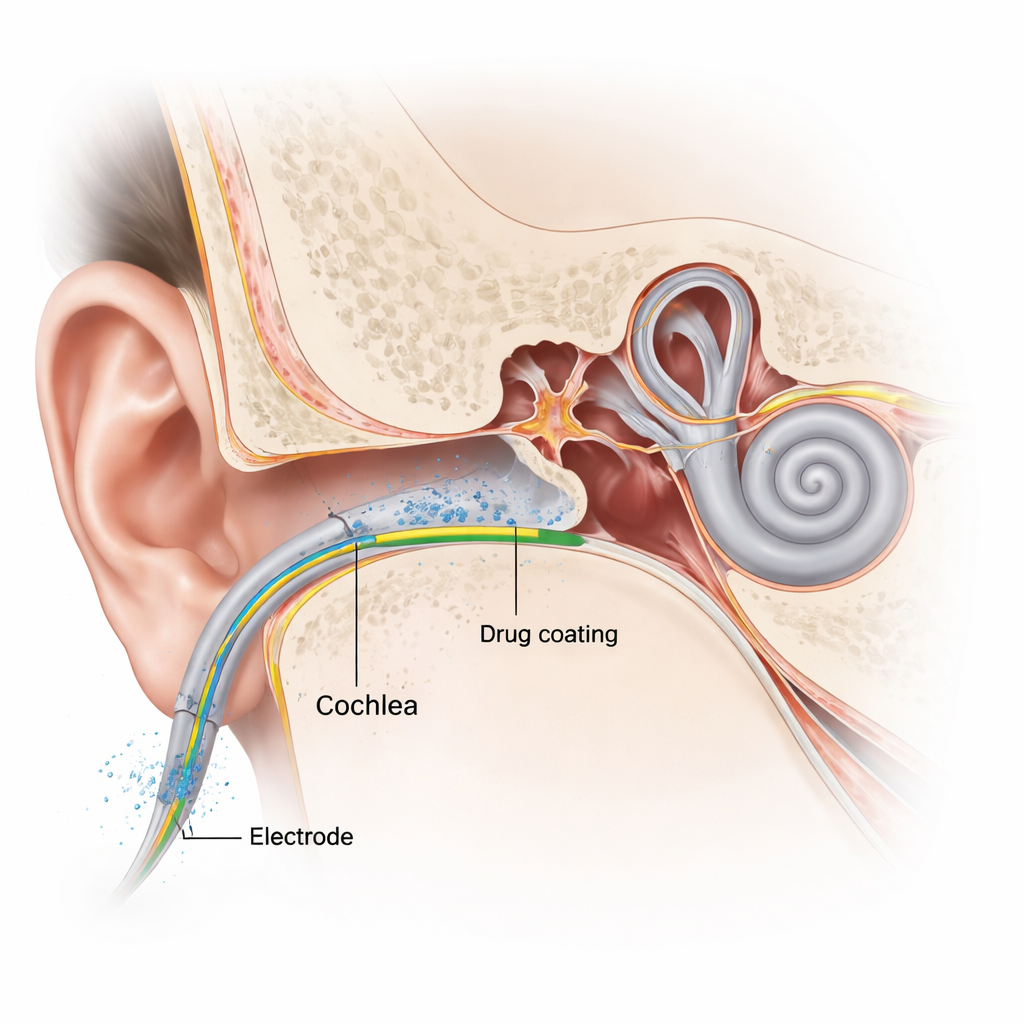
Why Cochlear Implants Can Still Harm Hearing
Even though cochlear implants have become more refined, inserting the electrode into the spiral-shaped cochlea is still an invasive procedure. The mechanical push of the electrode can injure sound-sensing hair cells and nerve structures and trigger an inflammatory response. In the days and weeks that follow, the body reacts to the implant as a foreign object, building up fibrous tissue around it. This scar-like tissue can raise the electrical resistance of the electrode, forcing the device to use stronger currents and making pitch signals less precise. Over time, this process may erode any remaining natural hearing and limit the full benefit of the implant.
Using a Familiar Drug in a Smarter Way
One promising way to protect the inner ear is to deliver anti-inflammatory drugs right at the site of injury. The steroid dexamethasone is already used to treat inner-ear problems because it can protect fragile cells and reduce swelling. But when given through the bloodstream or middle ear, the drug quickly washes out of the inner-ear fluid, making it hard to keep helpful levels for long. Earlier work showed that mixing dexamethasone evenly into silicone rods—similar to the flexible material of an implant—can give a steady trickle of drug for weeks. However, this approach required relatively large total amounts of steroid and produced only a short initial “burst” of higher levels, which may not be ideal for the intense early phase of inflammation right after surgery.
A Strip of Medicine Along the Electrode
In the new study, researchers tested a different idea: instead of filling the whole silicone rod with drug, they carved a shallow groove along its first few millimeters and filled that groove with dexamethasone-mixed silicone. These slender “strips” created weaponized edges of drug along the portion that sits closest to the main injury zone. Three versions were made, carrying 1.3, 2.6, or 5.2 micrograms of dexamethasone. The team implanted these coated rods into the inner ears of guinea pigs and, over 12 weeks, repeatedly sampled the inner-ear fluid to track how much drug was present and how it spread along the cochlea. Concentrations were measured with highly sensitive chemical analysis.
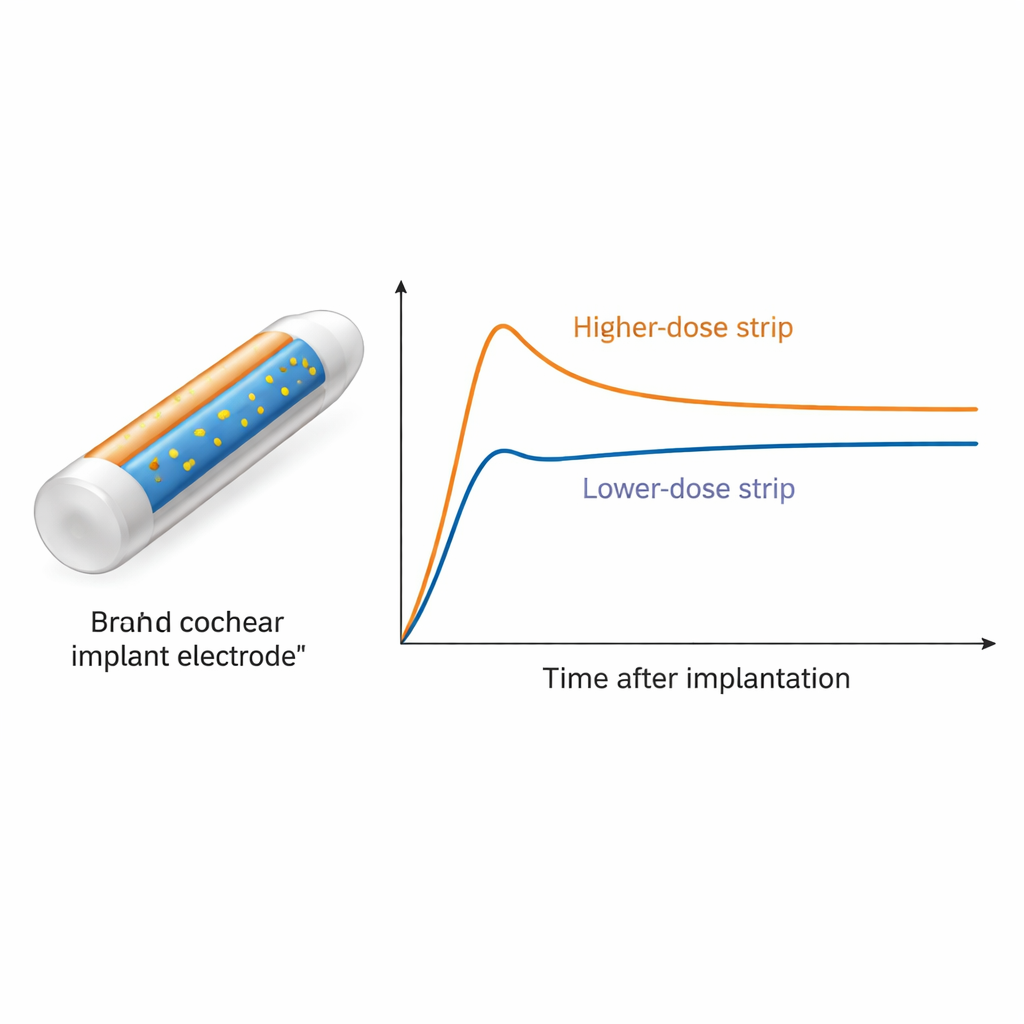
Longer Protection with Less Medicine
The strip-coated rods produced the desired two-phase pattern: an early surge of drug followed by a long, gentle plateau. The highest-dose rods (5.2 micrograms) reached peak levels around 450 nanograms per milliliter one day after surgery, then slowly declined over about a month into a stable zone between roughly 50 and 60 nanograms per milliliter that lasted at least 84 days. The medium dose (2.6 micrograms) showed the same shape but at lower levels, ending near 10 nanograms per milliliter by week twelve. The smallest dose (1.3 micrograms) never reached the roughly 50-nanogram range thought to offer strong protection, so it was dropped from later sampling. When the team drew a series of tiny samples along the length of the cochlea at day 42, they found dexamethasone everywhere in the inner-ear fluid, with the highest levels near the implanted rod and lower but still detectable amounts toward the far tip of the cochlea.
Doing More with Less for Future Patients
When the new strip-coated rods were compared with earlier, fully loaded rods, the coated design was more efficient. The 5.2-microgram coated rods achieved burst peaks similar to rods that contained about ten times more drug and maintained useful levels for a comparable or longer period. Concentrating the drug close to the surface and using smaller drug particles helped speed the early release and smooth the transition into the steady, lower phase. This means a real cochlear implant could potentially deliver strong local protection against early inflammation while limiting the total steroid dose the inner ear sees, easing safety concerns.
What This Could Mean for People with Implants
For patients, the ultimate goal is gentler surgery and better long-term hearing. A drug-releasing electrode that delivers a powerful early burst of anti-inflammatory medicine, followed by months of lower, stable levels, could help preserve the ear’s remaining natural hearing and keep scar tissue from degrading implant performance. Although this work was done in animals and with dummy rods, it shows that careful control over how and where the drug is built into the electrode can fine-tune the timing and amount of drug reaching the inner ear. This kind of engineering may pave the way for cochlear implants that both stimulate hearing and actively protect the fragile structures they rely on.
Citation: Liebau, A., Kammerer, B., Kather, M. et al. Long-term in vivo pharmacokinetics of dexamethasone-loaded cochlear implant electrode carrier dummies with optimized release profiles. Sci Rep 16, 5424 (2026). https://doi.org/10.1038/s41598-026-36620-0
Keywords: cochlear implant, inner ear drug delivery, dexamethasone, hearing preservation, controlled release