Clear Sky Science · en
The structural, mechanical, electrical, and radiation-shielding properties of newly yttrium and neodymium-doped lithium-zinc-phosphate glasses
Protective glass for a high‑radiation world
Modern hospitals, research labs, and nuclear facilities all need materials that can safely block harmful radiation without sacrificing transparency or strength. This study explores a new kind of specialty glass, tweaked at the atomic level with rare‑earth elements, to see whether it can better absorb radiation while also becoming tougher and more electrically responsive. The work shows how small changes in recipe—swapping in a heavier element called yttrium—subtly reorganize the glass structure and improve several useful properties at once.
Building a better glass recipe
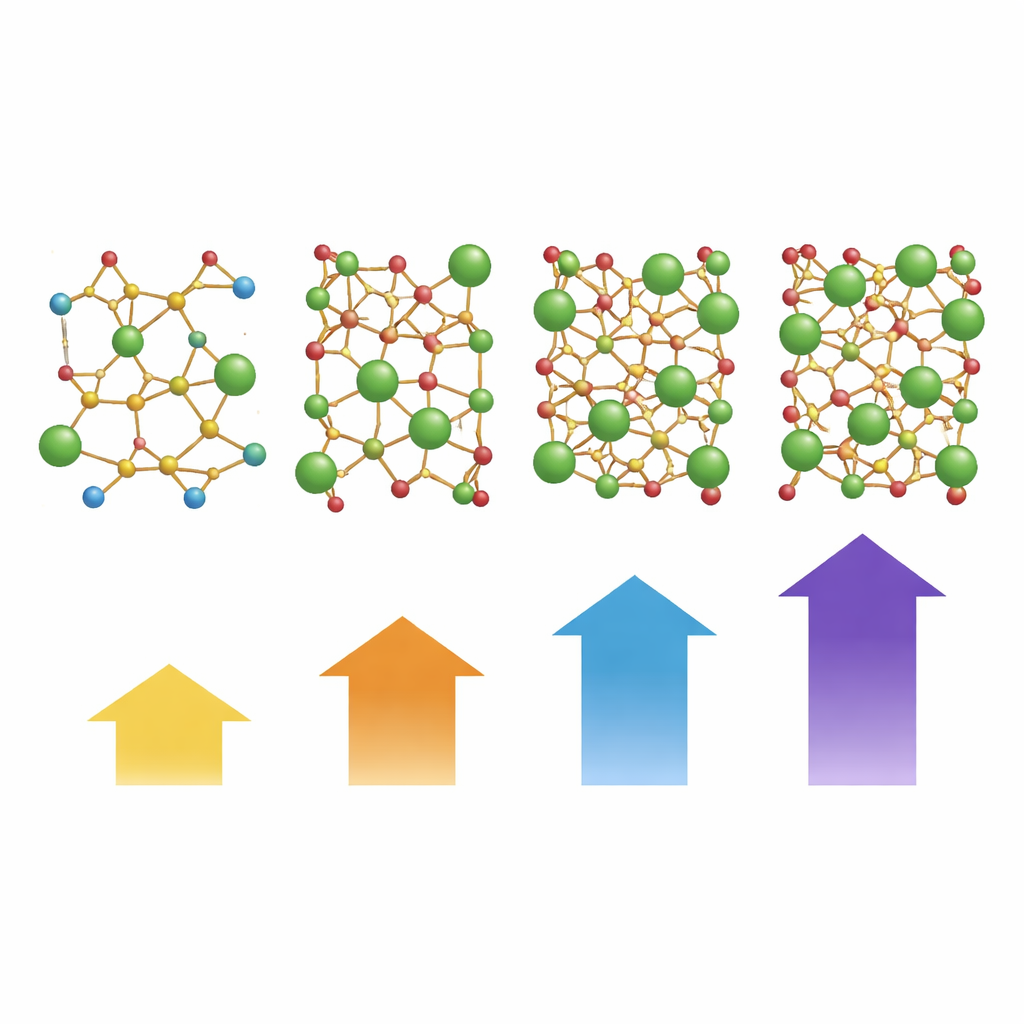
The researchers started with a base glass made mainly of phosphorus and oxygen (a phosphate glass), combined with lithium, zinc, bismuth, and a small dose of neodymium, a light‑emitting rare‑earth ion already used in lasers. To this mix they gradually added increasing amounts of yttrium oxide. Each batch was melted in a very hot furnace and then rapidly cooled, or “quenched,” to lock in a solid glass before crystals could form. By comparing glasses with four different yttrium levels, the team could watch how this single change affected structure, density, strength, electrical behavior, and ability to stop high‑energy radiation.
What happens inside the glass
At the microscopic level, ordinary phosphate glass is made of corner‑linked tetrahedral units—little pyramid‑like building blocks connected into chains and networks. Using infrared spectroscopy, the authors tracked how these building blocks shift as yttrium is added. They found that yttrium breaks some of the original links and forms new yttrium–oxygen bonds, creating more “loose ends” in the network. These non‑bridging oxygen sites and the new bonds increase structural disorder but also pull the network closer together. Measurements confirmed that density rises steadily as the lighter phosphorus‑rich units are replaced by heavier yttrium oxide, leading to a more compact, cohesive glass.
Electric behavior and mechanical strength
The changed internal network also alters how the glass responds to electric fields. When an alternating voltage is applied over a wide range of frequencies, the glass’s ability to store electric energy—its relative permittivity—starts high at low frequency and drops off as the field oscillates faster. With more yttrium, both permittivity and electrical conductivity increase overall, suggesting that the newly created oxygen “loose ends” and the rearranged network offer easier pathways for mobile ions such as lithium to move. At the same time, calculated mechanical parameters show that the glass becomes stiffer: Young’s modulus, bulk modulus, and shear modulus all climb with yttrium content. In practical terms, the glass resists compression, stretching, and shearing more effectively, even though its hardness changes only slightly.
Stopping X‑rays and neutrons
Because yttrium atoms are heavier than phosphorus, their presence also influences how the glass interacts with high‑energy photons and fast neutrons. The team computed an effective atomic number, a measure linked to how strongly a material absorbs radiation, across photon energies from medical X‑ray levels up to energies relevant for nuclear technology. This value is highest at very low photon energies, dips in the mid‑range where scattering dominates, and rises again at the highest energies. Adding yttrium nudges the effective atomic number upward at all energies and produces a small but consistent improvement in both photon and neutron shielding. In some cases, the glass performs as well as or better than common building materials like concrete and approaches the performance of commercial shielding glasses.
Why this glass matters
Altogether, the study shows that carefully introducing yttrium into lithium‑zinc‑phosphate glass creates a denser, mechanically stronger, and more electrically responsive material that also absorbs radiation a bit more effectively. For a layperson, the takeaway is that “designer” glass can be tuned like an alloy: by switching in specific elements, scientists can trade a relatively open, lightweight network for a heavier, more connected structure that both blocks radiation and stands up well to mechanical and electrical demands. Such glasses could one day help improve windows, viewports, and components in environments where people and instruments must be protected from intense radiation without losing visibility or durability.
Citation: Alharshan, G.A., Shaaban, S.M., Elsad, R. et al. The structural, mechanical, electrical, and radiation-shielding properties of newly yttrium and neodymium-doped lithium-zinc-phosphate glasses. Sci Rep 16, 7971 (2026). https://doi.org/10.1038/s41598-026-36616-w
Keywords: radiation shielding glass, yttrium-doped phosphate, rare-earth materials, dielectric properties, mechanical strength