Clear Sky Science · en
Integrated economic and adsorption performance study of CMC/MMT nano-composite for cationic dye removal from industrial wastewater
Why cleaning colorful wastewater matters
From the clothes we wear to the paints on our walls, bright colors often come from synthetic dyes that eventually wash down the drain. Many of these dyes are stubborn pollutants that slip through ordinary treatment plants and can harm aquatic life and human health. This study explores a new, low-cost material made from natural biopolymers and clay that can pull a common blue dye out of water with remarkable efficiency, while also examining whether the process makes financial sense on an industrial scale.
A new sponge made from shells and clay
The researchers built their dye-catching material by combining two ingredients: chitosan, a substance derived from crustacean shells, and montmorillonite, a naturally occurring clay. By chemically modifying chitosan to add negatively charged groups and then blending it with the layered clay, they created a “nanocomposite” called CMC/MMT. At the microscopic scale, this composite presents a highly porous structure with many tiny channels and a large surface area, giving dye molecules plenty of places to stick. Energy-dispersive X-ray measurements before and after treatment showed that elements from the dye, such as nitrogen, sulfur, and chlorine, appeared on the composite surface, confirming that the material was actually capturing the pollutant rather than simply filtering it mechanically.
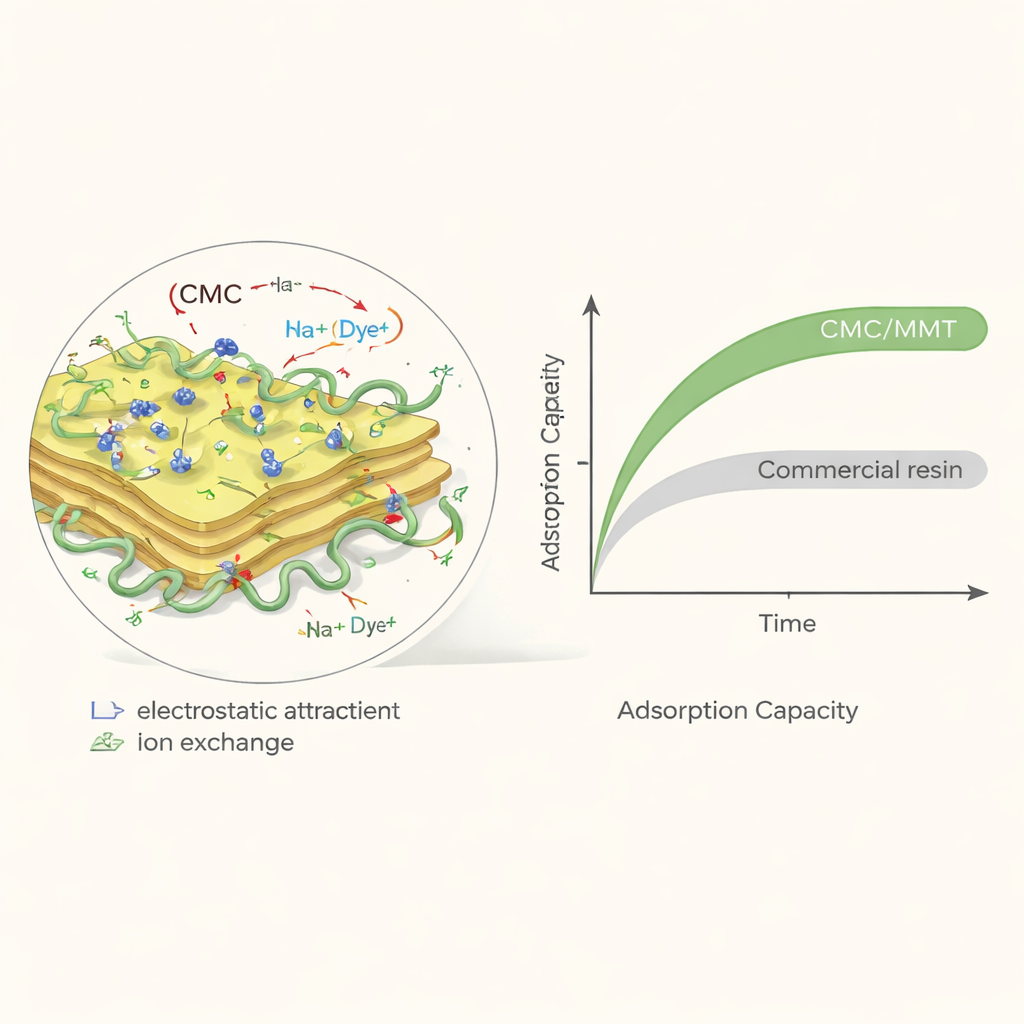
How the material grabs and holds dye
The team focused on methylene blue, a widely used cationic (positively charged) dye known to irritate eyes and cause breathing and blood problems at high doses. They tested how well CMC/MMT removes this dye under different conditions, including water acidity (pH), starting dye concentration, temperature, and contact time. The composite worked efficiently across a broad pH range and was especially effective at a mildly basic pH of about 8.5 and a temperature of 30 °C, both typical of many real wastewater streams. Under these conditions, the surface of the composite carries a net negative charge, which attracts the positively charged dye molecules. Detailed infrared spectroscopy indicated that the dye attaches through a combination of electrostatic attraction, ion exchange with metal ions in the clay layers, and hydrogen bonding to the polymer’s functional groups.
Fast and powerful dye removal
When methylene blue solutions were mixed with small amounts of the composite, most of the dye disappeared from the water within the first half hour, and the system reached near-complete equilibrium in about two hours. Mathematical modeling of this time dependence showed that the process follows what scientists call “pseudo-second-order” kinetics, which is consistent with a chemically driven binding step rather than simple diffusion alone. By analyzing how much dye the material could hold at different concentrations, the authors found that the behavior was best described by the Langmuir model, meaning the dye forms an orderly single layer on the composite surface. Under optimized conditions, the maximum amount of dye that could be taken up reached about 435 milligrams per gram of composite—substantially higher than many other biopolymer and clay-based adsorbents reported in the literature.
Beating a commercial standard at lower cost
To judge whether this new material is truly practical, the team compared CMC/MMT with a widely used commercial ion-exchange resin known as Amberlite IR 120. In head-to-head tests, the nanocomposite removed more methylene blue per unit mass, outperforming the commercial product by roughly 27 percent in capacity. The researchers then designed a hypothetical production line capable of making two tons of the composite per day and carried out a detailed techno-economic analysis, including equipment costs, energy use, labor, and maintenance. They estimated a production cost of about 21 US dollars per kilogram of composite. Because CMC/MMT is so effective at binding dye, less material is needed to clean a given amount of wastewater, and the calculated cost to remove one kilogram of dye was much lower than for the commercial resin.
What this means for cleaner, cheaper water
In simple terms, this study shows that a sponge-like material made from natural polymers and clay can clean stubborn blue dye from industrial wastewater more effectively and more cheaply than a common commercial alternative. The composite works quickly, holds a large amount of dye, and can be regenerated and reused several times with only a gradual loss of performance. By combining laboratory measurements with a full economic evaluation, the work suggests that scaling up such biobased nanocomposites could be a realistic path toward cleaner rivers and safer drinking water in dye-intensive industries.
Citation: Khedr, M., Waly, A.I., Hafez, A.I. et al. Integrated economic and adsorption performance study of CMC/MMT nano-composite for cationic dye removal from industrial wastewater. Sci Rep 16, 7726 (2026). https://doi.org/10.1038/s41598-026-36615-x
Keywords: wastewater treatment, dye adsorption, chitosan clay composite, methylene blue removal, techno-economic analysis