Clear Sky Science · en
Combined effects of nitrate and antimicrobial compounds on in vitro subgingival biofilms
Why your gums and gut medicine are connected
Bleeding gums and gum disease are usually blamed on “bad” bacteria. To fight them, dentists often turn to strong mouthwashes and antibiotics. This study asks a bigger question: what if some of these treatments quietly damage the helpful bacteria we need, while a simple nutrient found in leafy vegetables actually nudges our mouth microbes toward health?
Gum disease as a community problem
Periodontitis is a long‑lasting infection around the teeth that destroys the tissues holding them in place and is linked to heart disease, diabetes and even brain conditions. Rather than being caused by a single germ, it arises when the whole community of mouth bacteria shifts into a harmful state, called dysbiosis. Modern treatment still leans heavily on broad‑acting drugs: antibiotics such as amoxicillin and metronidazole, and antiseptic rinses like chlorhexidine. These products can kill troublemakers—but they can also wipe out friendly bacteria and may encourage drug resistance.
A closer look at biofilms from real patients
To see what these treatments actually do to gum bacteria, the researchers collected plaque from deep pockets around the teeth of 12 people with periodontitis. In the lab, they grew these samples as thin layers of bacteria, or biofilms, for eight hours under eight different conditions: no treatment, nitrate alone, each drug alone, and each drug combined with nitrate. Nitrate, a natural compound abundant in spinach, lettuce and beetroot, can be turned by certain mouth bacteria into nitric oxide, a gas that helps blood vessels relax and can slow the growth of disease‑related microbes. The team measured how much biofilm formed, how well it converted nitrate, and which bacterial species thrived under each treatment.
Drugs that shrink growth but may worsen balance
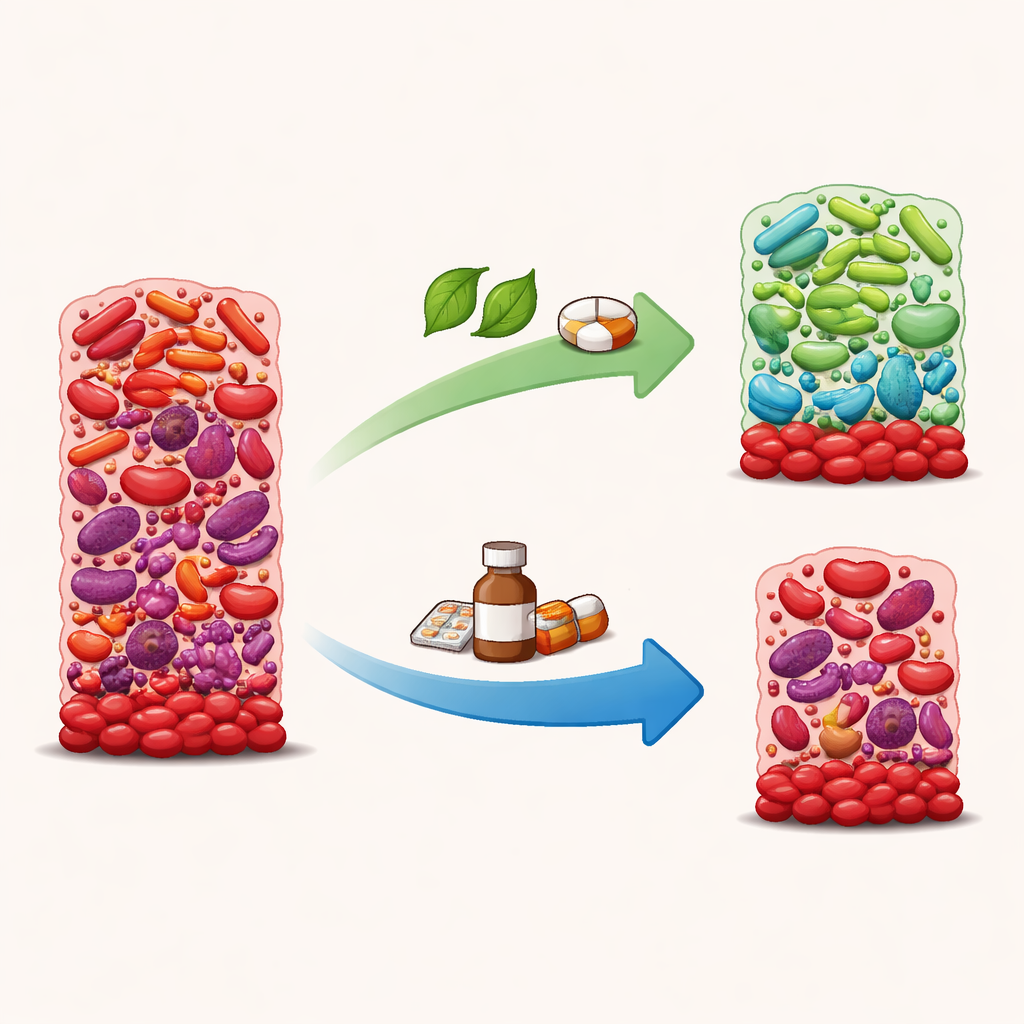
Low doses of chlorhexidine and amoxicillin cut biofilm growth roughly in half and strongly reduced the bacteria’s ability to process nitrate. But this was not a selective “good clean‑up.” DNA sequencing showed that, in these conditions, health‑associated bacteria such as Rothia, Gemella and Kingella tended to decline, while several species linked with gum disease—including Fusobacterium, Treponema and Eubacterium—either stayed put or became more prominent. An index that scores how “diseased” a community looks under the microscope was higher with chlorhexidine and amoxicillin than with nitrate, suggesting these drugs can push the biofilm toward a more harmful, imbalanced state even as they suppress overall growth.
Leafy‑green fuel and a gentler antibiotic
Nitrate told a different story. When plaque was grown with nitrate alone, bacteria that specialize in turning nitrate into useful products, such as Neisseria and some Aggregatibacter species, increased. The dysbiosis score dropped compared with chlorhexidine or amoxicillin conditions, pointing toward a healthier community. Metronidazole, at a level similar to what actually reaches the gum pockets in patients, did not strongly reduce growth or nitrate use on its own. But when combined with nitrate, it appeared to tip the balance in a helpful way: disease‑associated species like Fusobacterium and Treponema fell, while nitrate‑using Neisseria and Kingella rose. This pairing seemed to prune back the worst offenders without stripping away key functions.
What this means for everyday care
For people with gum disease, these findings hint that “strongest” is not always “best” when it comes to antimicrobials. In this lab model, low‑dose chlorhexidine and amoxicillin slowed bacterial growth but also weakened a natural, nitrate‑based pathway that supports both oral and whole‑body health, and they were linked with more disease‑like microbial patterns. Nitrate, especially when combined with metronidazole, favored bacteria tied to healthier gums and preserved nitrate metabolism. While real mouths are more complex than lab dishes, this work supports the idea of gentler, microbiome‑friendly strategies—such as nitrate‑rich diets and carefully chosen antibiotics—to manage gum disease without undermining the very bacteria that help keep us well.
Citation: Moran, S.P., Nadal-Ruiz, M., Mira, A. et al. Combined effects of nitrate and antimicrobial compounds on in vitro subgingival biofilms. Sci Rep 16, 6686 (2026). https://doi.org/10.1038/s41598-026-36588-x
Keywords: gum disease, oral microbiome, nitrate, mouthwash, antibiotics