Clear Sky Science · en
The tumor microenvironment in esophageal cancer and its association with clinical features and neoadjuvant treatment response
Why the Tumor’s Neighborhood Matters
Esophageal cancer is one of the deadliest cancers worldwide, and even with modern chemotherapy and radiotherapy many patients do not respond as well as hoped. This study asks a deceptively simple question with big implications: what is happening in the “neighborhood” around the tumor—the tumor microenvironment—and can its makeup help explain why some patients respond to treatment while others do not? By looking closely at immune cells and related markers before and after therapy, the researchers begin to map how the body and the tumor interact, and how this hidden dialogue might guide future, more personalized treatments.
The Hidden Ecosystem Around a Tumor
Solid tumors do not grow in isolation. They sit in a complex ecosystem of immune cells, support cells, and signaling molecules known as the tumor microenvironment. In this study, doctors and pathologists in Lausanne examined tumor samples from 68 patients who had surgery for esophageal cancer between 2009 and 2021. Most had received powerful treatment before surgery—either chemotherapy, chemoradiation, or both—to shrink the tumor. Using special stains on tissue slides, the team counted several key immune cell types (including general T cells, killer T cells, and different kinds of macrophages) and measured markers that can predict response to immunotherapy, such as PD-L1 and HER2.
Who the Patients Were and What Was Measured
The patients in this series were mostly men in their early sixties, and the majority had adenocarcinoma, the type of esophageal cancer often linked to chronic acid reflux and Barrett’s esophagus. A smaller group had squamous cell cancer, a different form that arises from the flat lining cells of the esophagus. The researchers carefully linked what they saw under the microscope to clinical details such as stage of disease, smoking status, weight loss, and the kind of pre-surgery treatment received. They also tracked how completely each tumor responded to therapy, using a grading system that distinguishes complete disappearance of cancer cells from little or no response.
Lifestyle, Tumor Type, and a Quieter Immune Presence
Several everyday and disease-related factors turned out to be associated with a weaker immune presence around the tumor. Patients who were active smokers had fewer macrophages overall and fewer of the so‑called M2-like macrophages, which are often linked to tumor support and immune suppression. People with more advanced tumors, greater preoperative weight loss, or squamous cell cancers tended to have fewer T cells and macrophages infiltrating their tumors. In contrast, tumors with high PD-L1 scores—an indicator often used to select patients for immunotherapy—were “hotter,” with more T cells and macrophages, but this pattern has previously been tied to worse outcomes, likely because the tumor is actively using PD-L1 to switch off immune attack.
How Treatment Reshapes the Tumor’s Neighborhood
Neoadjuvant (pre-surgery) treatment substantially reshaped the tumor microenvironment. Both chemotherapy and chemoradiation increased the number of macrophages, including M2-like cells, while sharply reducing regulatory T cells, a group that normally dampens immune responses. Overall T cell and killer T cell numbers changed less consistently. Interestingly, chemoradiation tended to lower PD-L1 levels on tumor and surrounding cells, reducing the share of patients with high PD-L1 scores after treatment. This suggests that standard treatment can make tumors look less suitable for PD-1/PD-L1–blocking immunotherapies if judged only by a single snapshot after chemoradiation.
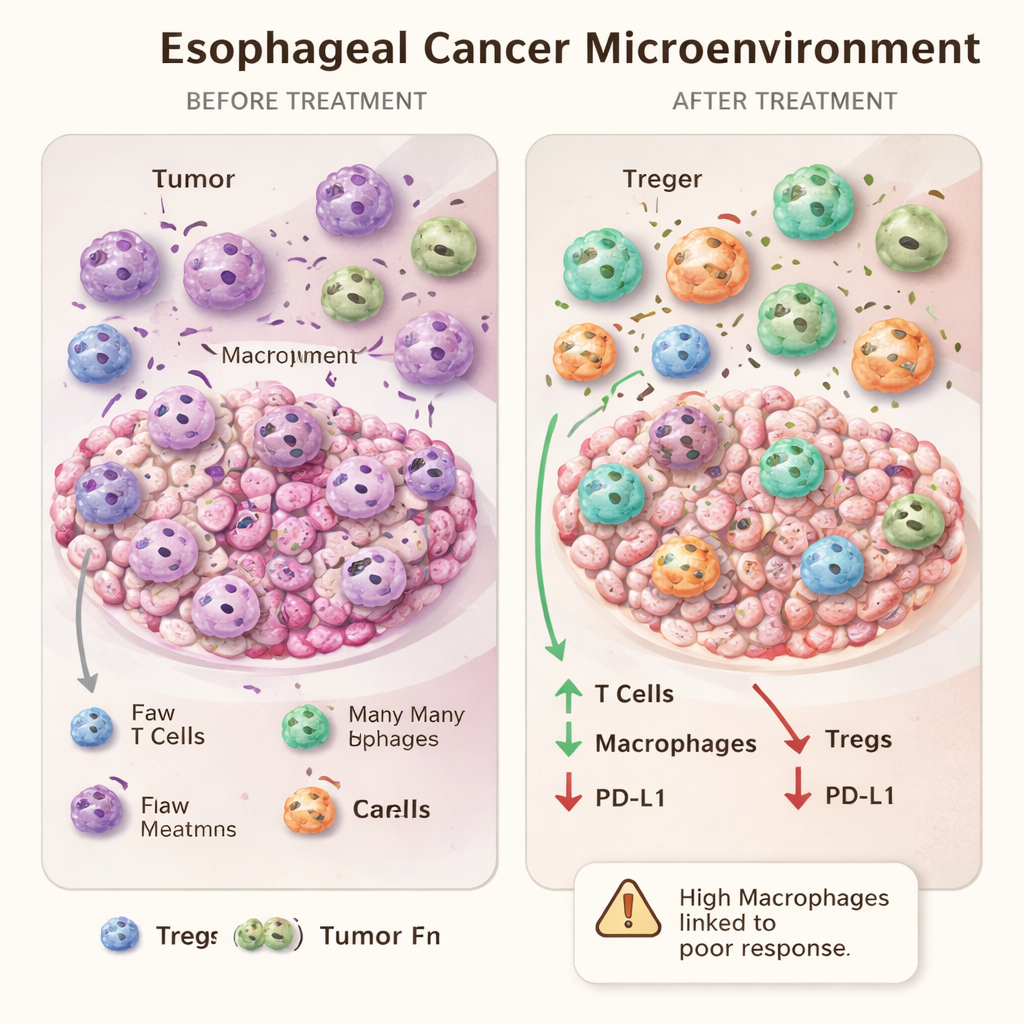
When More Immune Cells Might Signal Resistance
One of the most intriguing findings came from comparing the extremes of treatment response. Patients whose tumors disappeared completely under the microscope did not differ, at baseline, from poor responders in terms of the immune cells surrounding their cancer. However, after treatment, those with poor responses had higher numbers of macrophages in their tumor areas than complete responders. Rather than being a simple sign of strong immunity, an abundance of macrophages after therapy may mark an exhausted or skewed response that the tumor has learned to exploit. This adds weight to the idea that some macrophages, particularly of the M2-like type, may actually promote tumor survival and resistance.
What This Means for Future Care
For patients and clinicians, the take‑home message is that the biology around an esophageal tumor—its microenvironment—is shaped by smoking, tumor type, disease severity, and by the treatments themselves. Standard chemo and chemoradiation do not just shrink tumors; they also recruit and reshape immune cells in ways that may help or hinder long‑term control of the disease. The study suggests that counting certain immune cells, especially macrophages, before and after therapy could one day help identify who is likely to resist treatment and who might benefit from additional drugs that target these cells or the PD-1/PD-L1 pathway. While more, larger studies are needed, this work moves us closer to tailoring esophageal cancer treatment not only to the tumor’s genetics but also to the dynamic ecosystem that surrounds it.
Citation: Fasquelle, F., Teixeira Farinha, H., Sempoux, C. et al. The tumor microenvironment in esophageal cancer and its association with clinical features and neoadjuvant treatment response. Sci Rep 16, 5664 (2026). https://doi.org/10.1038/s41598-026-36537-8
Keywords: esophageal cancer, tumor microenvironment, immune cells, chemoradiation, immunotherapy