Clear Sky Science · en
Construction and initial validation of key gene network for progesterone resistance in endometrial cancer based on genome-wide CRISPR screening
Why some uterine cancers stop listening to hormones
For many people with early endometrial (uterine lining) cancer who still hope to have children, hormone pills that mimic the natural hormone progesterone can shrink tumors while preserving the uterus. Yet up to a third of these cancers either never respond or quickly become resistant, forcing patients toward surgery or harsher treatments. This study uses a powerful gene-editing tool to map which genes make endometrial cancer cells ignore progesterone, pointing toward future tests and drugs that could keep this therapy working longer.
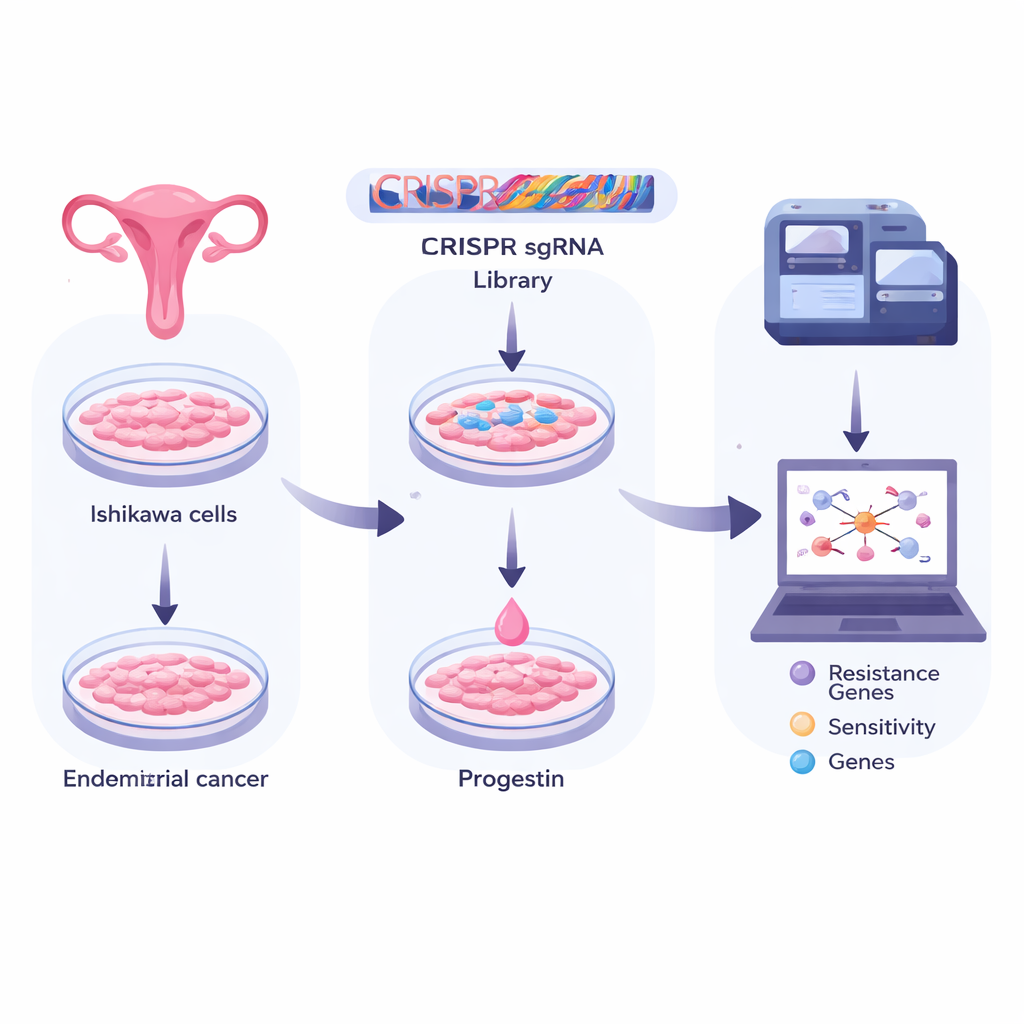
Using gene scissors to search the whole genome
The researchers worked with Ishikawa cells, a widely used model of endometrial cancer. They infected millions of these cells with a library of CRISPR “gene scissors,” each programmed to switch off a different human gene. This created a vast pool in which, in principle, every cell had a unique gene disabled. The team then split the cells into groups: one frozen as a baseline, one exposed only to a harmless solvent, and one treated for many days with a common synthetic progesterone (medroxyprogesterone, or MPA). By deep sequencing the CRISPR tags that survived in each group, they could see which gene knockouts helped cells live or die under progesterone treatment.
Sorting out genes that help or hinder drug response
Analyzing this flood of data required sophisticated statistics. The team applied two complementary methods, RRA and MLE, to rank genes according to how strongly they protected cells from progesterone (resistance genes) or made cells more likely to die (sensitivity genes). They found hundreds of candidates in each direction. Many of these genes clustered in pathways that handle the copying and repair of DNA, the making and processing of RNA, and general cell metabolism. In other words, the basic machinery that lets cells read, maintain, and act on their genetic information appears tightly linked to whether endometrial cancer cells obey progesterone’s “stop growing” signal.
Narrowing the list to the most important players
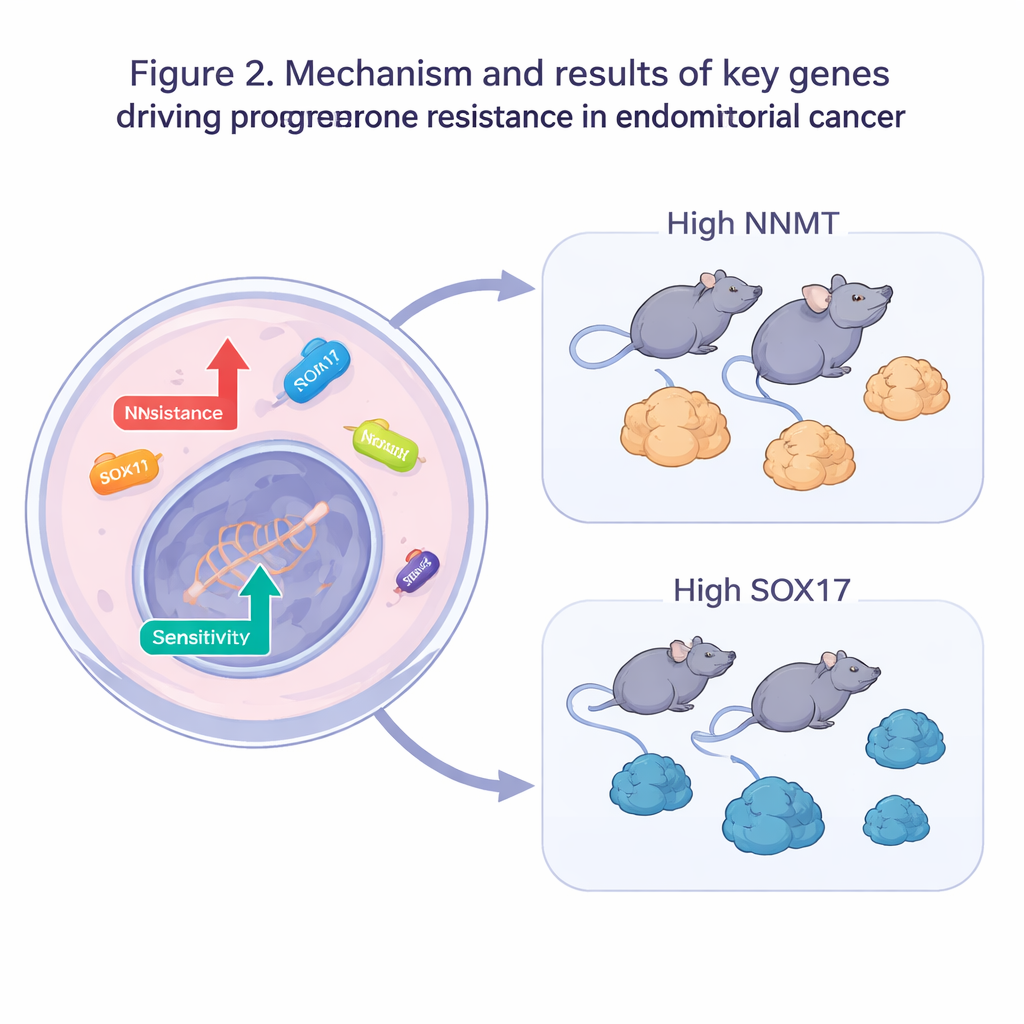
To avoid chasing false leads, the scientists cross-checked the CRISPR hits against an older dataset from their lab, in which they had compared ordinary cancer cells with a strain made resistant to progesterone over time. By looking for genes that appeared in both screens and showed consistent changes, they distilled the list down to five genes that seem to promote resistance and twenty that seem to fight it. Public cancer databases showed that many resistance-blocking genes are more active in the most common type of endometrial cancer, while several resistance-promoting genes correlate with poorer patient outcomes, suggesting that these signals matter in real tumors, not just in dishes.
Testing key genes in cells and in mice
The team then moved from big data to hands-on experiments. In progesterone-resistant cells, they shut down each of the five suspected resistance genes or boosted the five suspected sensitivity genes. When these altered cells were exposed to progesterone, many more died and much lower drug doses were needed to slow growth, confirming that tweaking single genes could restore hormone sensitivity. Two genes stood out: NNMT, which is involved in cell metabolism, and SOX17, a gene that helps control how cells develop and behave. In mouse tumor models, cranking up NNMT made tumors less responsive to progesterone, while boosting SOX17 made resistant tumors shrink more under the same treatment.
What these findings could mean for patients
Put simply, the study builds an early “wiring diagram” of how endometrial cancer cells decide whether to listen to progesterone. It suggests that resistance is not just about losing the progesterone receptor, but also about deeper changes in DNA and RNA handling and in key control genes such as NNMT and SOX17. In the future, blood or tissue tests that read out this gene network could help doctors predict who will benefit from hormone-based fertility-sparing therapy. Even more promising, drugs that dial down resistance genes or boost sensitivity genes might one day be combined with progesterone to keep this gentle treatment option effective for more people and for longer.
Citation: Li, X., Wang, S., Qiu, Z. et al. Construction and initial validation of key gene network for progesterone resistance in endometrial cancer based on genome-wide CRISPR screening. Sci Rep 16, 8030 (2026). https://doi.org/10.1038/s41598-026-36534-x
Keywords: endometrial cancer, progesterone resistance, CRISPR screening, gene networks, hormone therapy