Clear Sky Science · en
Development of a green chemistry based bioanalytical method using response surface methodology to analyze febuxostat and indomethacin in rabbit plasma
Why cleaner drug testing matters
Many people living with gout take more than one medicine at a time, such as febuxostat to lower uric acid and indomethacin to ease painful flares. To make sure these drugs work safely and effectively, scientists need to measure how much of each is circulating in the blood. This study shows how researchers developed a laboratory test that can measure both drugs together in rabbit blood while also reducing the use of harmful chemicals, offering a glimpse of how medical testing can become both smarter and greener.
Tracking two gout medicines at once
Doctors often prescribe febuxostat and indomethacin together: one controls uric acid levels long term, the other calms inflammation during attacks. Yet existing laboratory methods were not well suited to tracking both drugs at the same time in blood, especially in a way that considered environmental impact. The authors set out to build a single test that could monitor febuxostat and indomethacin side by side in blood plasma, providing clear information for future studies of dosing, drug interactions, and treatment response.
Making the lab method greener
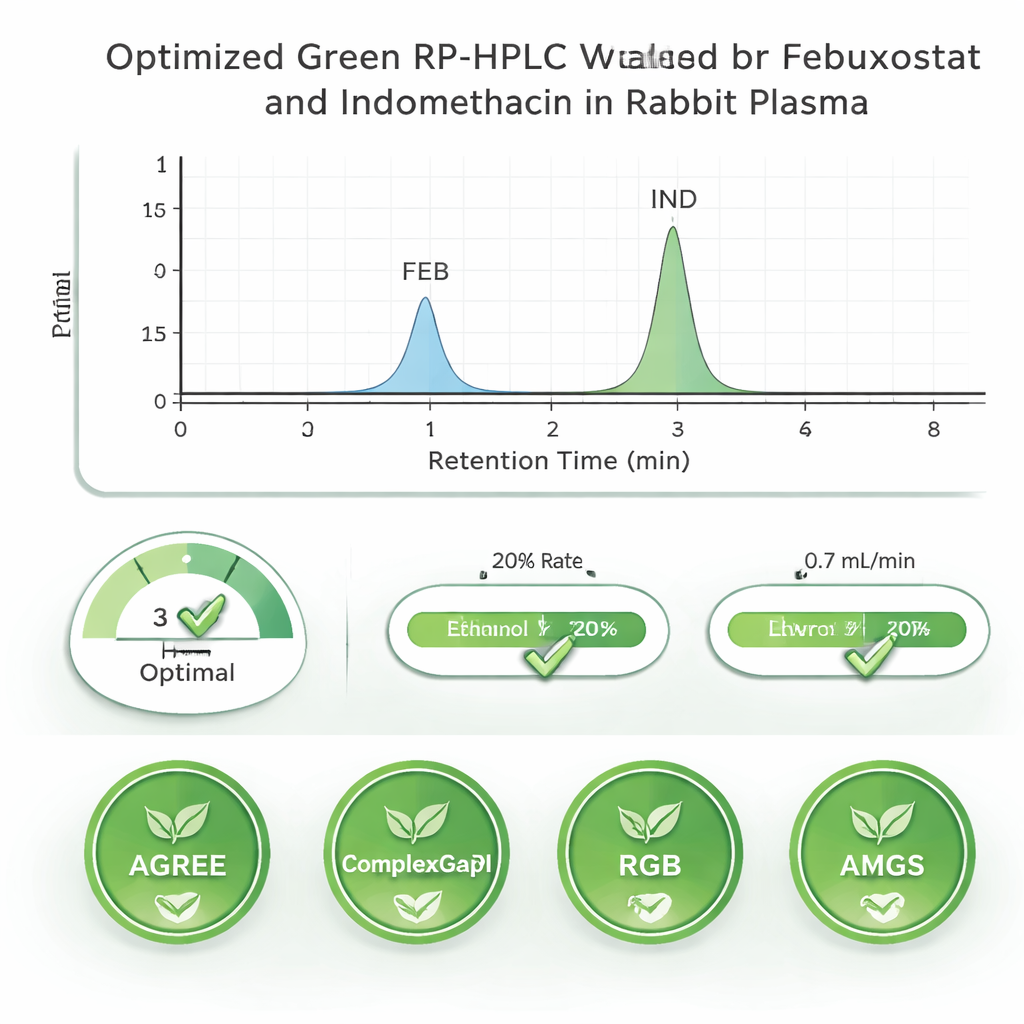
A major goal of the work was to make the test kinder to people and the planet. Many common lab solvents, such as acetonitrile and methanol, are effective but toxic and less sustainable. Here, the team chose ethanol, a less hazardous and more renewable solvent that still dissolves both drugs well. They used ethanol both to separate the drugs on a chromatography column and to pull them out of rabbit plasma in a simple protein-precipitation step. To judge how eco-friendly their approach was, they applied several modern “green” scoring tools—ComplexGAPI, AGREE, RGB, and AMGS—which look at factors such as chemical toxicity, waste generated, energy use, and overall practicality. The method achieved a very low waste factor and high greenness scores, indicating that it compares favorably with traditional approaches that rely on harsher solvents.
Designing the method from the ground up
Instead of relying on trial and error, the researchers used a structured “Quality by Design” strategy to fine-tune the test. They focused on three key settings: the acidity (pH) of the buffer, the percentage of ethanol, and the flow rate of the liquid running through the chromatography column. Using a statistical tool called a central composite design, they ran 20 carefully chosen experiments and measured how well the two drug signals were separated and how symmetrical the peaks looked. Computer analysis of these runs revealed which settings truly mattered and mapped out a “design space”—a safe operating window in which the method would remain reliable. Within this window, the team identified ideal conditions that gave clear, sharp peaks for both drugs in just a few minutes.
Proving the test is reliable
After settling on the best settings, the scientists put the method through a battery of checks similar to those required by regulatory agencies. They showed that the test responds in a straight line over a wide range of drug levels, from very low to relatively high concentrations in plasma. Repeated measurements on the same day and on different days gave almost identical results, demonstrating strong precision. They also confirmed that the method can recover most of the drugs from plasma, that natural substances in blood do not interfere, and that the drug levels remain stable under typical storage and handling conditions. Together, these results indicate that the method is accurate, consistent, and robust enough to support pharmacokinetic and monitoring studies.
What this means for patients and the environment
In simple terms, this work delivers a dependable lab test that can measure two key gout medicines at once while cutting back on hazardous chemicals and waste. Although the study was carried out in rabbit plasma and in a single laboratory, the approach can be adapted to other drugs and systems. As more researchers adopt these combined “quality by design” and green chemistry principles, medical testing can become not only more informative for patient care, but also safer for laboratory staff and less harmful to the environment.
Citation: Ansary, A., Piyongsola, Paul, B. et al. Development of a green chemistry based bioanalytical method using response surface methodology to analyze febuxostat and indomethacin in rabbit plasma. Sci Rep 16, 5645 (2026). https://doi.org/10.1038/s41598-026-36517-y
Keywords: gout treatment, green analytical chemistry, liquid chromatography, febuxostat, indomethacin