Clear Sky Science · en
Sustainable production of battery-grade nickel via hydrogen reduction of saprolite
Cleaning Up the Nickel Behind Electric Cars
Nickel is a quiet workhorse of modern technology, especially in the high-performance batteries that power electric vehicles. Yet producing this metal is surprisingly dirty, releasing large amounts of carbon dioxide. This study explores a way to make “battery-grade” nickel with far lower emissions by replacing coal with hydrogen gas in a key smelting step, potentially shrinking the climate footprint of the electric-car revolution.
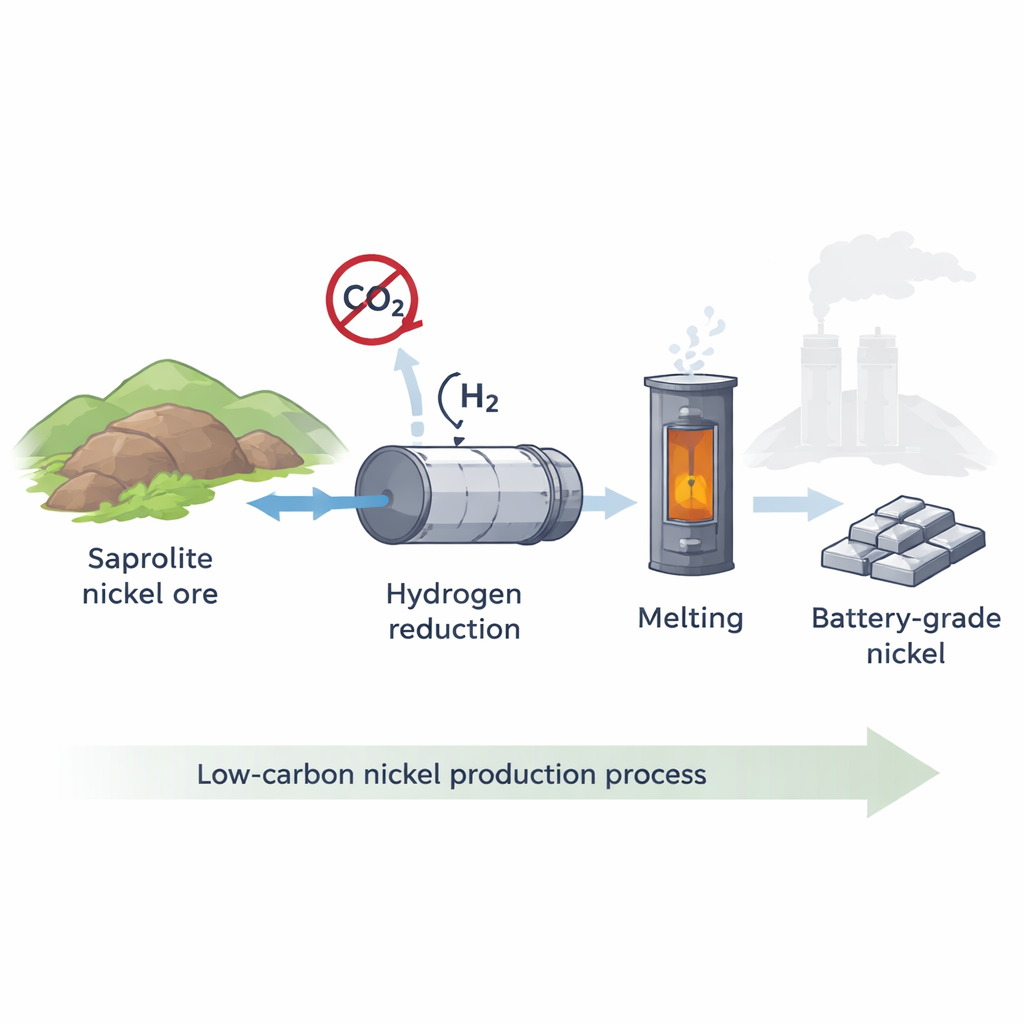
Why This Type of Nickel Ore Matters
Much of the world’s nickel comes from weathered tropical rocks called laterites. One major variety, saprolite ore, is rich in magnesium-bearing silicate minerals and typically contains more than 1.5% nickel. Today, nearly all saprolite is treated by a high-temperature route known as the rotary kiln–electric furnace (RKEF) process, which burns coal both as fuel and as a chemical reducing agent. Depending on conditions, this can emit from about 30 up to more than 60 tons of carbon dioxide for every ton of nickel produced. Alternatives such as aggressive acid leaching are often even more carbon intensive. As demand for nickel soars with electric vehicle growth and environmental scrutiny intensifies, there is strong pressure to find cleaner smelting technologies.
Using Hydrogen Instead of Coal
The researchers focused on a promising alternative: using hydrogen gas, rather than coal, to strip oxygen from the nickel- and iron-bearing minerals in saprolite. They built a one‑meter‑long rotating steel reactor that mimics the motion and gas–solid contact of an industrial kiln. Finely sized saprolite from New Caledonia was fed into this chamber, which was first heated under nitrogen and then exposed to a controlled stream of nearly pure hydrogen at temperatures between 800 and 950 °C. By carefully tracking weight loss and changes in mineral structure, they could see how fast and how completely the ore was reduced under different operating conditions, such as temperature, gas flow, and particle size.
Why Particle Size Is the Hidden Lever
Detailed mineral and chemical analysis showed that coarse particles are richer in magnesium silicates, while fine particles contain proportionally more iron minerals, but nickel itself is spread almost evenly across all grain sizes. That means physical separation of nickel-bearing minerals is not practical: the whole ore must be treated together. During hydrogen treatment at 900 °C, the samples quickly lost about 20% of their mass—a combined signal of water release from heated minerals and oxygen removal during metal formation. Strikingly, this mass loss reached its final value within just 15 minutes and changed little with longer times. Instead, two physical factors dominated performance: gas flow and particle size. Once the hydrogen flow exceeded about 3 liters per minute, supplying more gas brought no extra benefit. In contrast, grinding the ore finer gave a strong boost: the smallest particles, under 45 micrometers, achieved the highest and fastest reduction, because hydrogen could more easily diffuse through the thin silicate framework to reach nickel and iron atoms trapped inside.
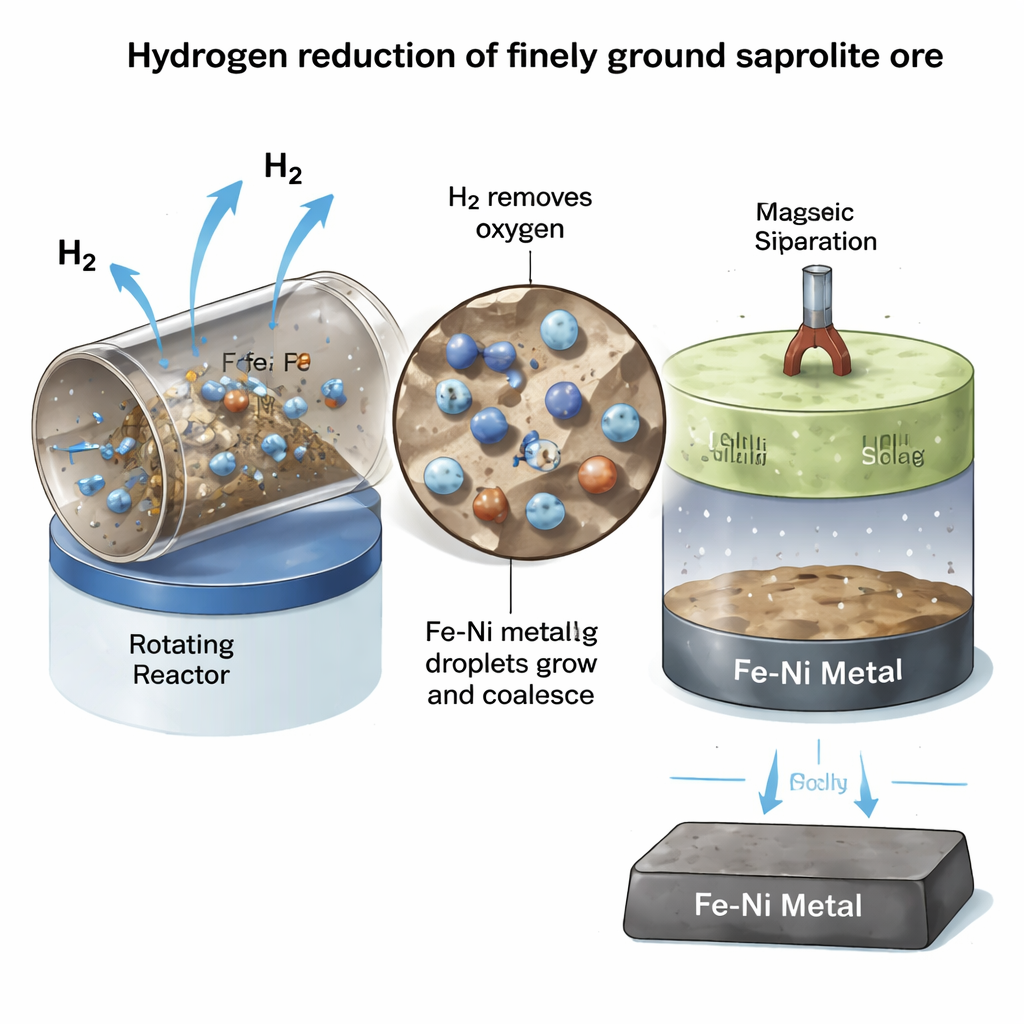
From Reduced Ore to Battery-Grade Metal
To see whether this hydrogen-treated ore could yield a usable product, the team melted the reduced powder in a high-temperature vertical furnace under an inert argon atmosphere. At 1550 °C, the material separated cleanly into two layers: a dense iron–nickel alloy that sank to the bottom and a lighter, magnesium-rich silicate slag that floated on top. Microscopic imaging and chemical mapping confirmed that the metal layer contained about 73% iron and 25% nickel—typical of industrial nickel pig iron—while the slag was largely free of metal. Because the alloy is strongly magnetic, it could be separated completely with simple magnetic equipment, pointing to an efficient route from ore to smelter-ready feedstock without adding extra chemicals or solid reductants.
What This Means for Cleaner Batteries
For non-specialists, the key message is that the way we process nickel can be made much cleaner without changing the type of ore we mine. By finely grinding saprolite and exposing it to a brisk flow of hydrogen at around 900 °C, the ore can be transformed in minutes into a material that melts into high-grade nickel pig iron, with a clear split between metal and waste rock. Because hydrogen produces water instead of carbon dioxide when it does the chemical “unbinding,” this approach could sharply cut emissions from nickel smelting if powered by low-carbon energy. The study lays out the operating window—temperature, gas flow, and particle size—that engineers can use to design low-carbon plants, and it highlights the next step: testing this hydrogen-based process in continuous pilot kilns to prove that greener nickel can be made reliably and at scale.
Citation: Park, T., Han, S., Lee, W. et al. Sustainable production of battery-grade nickel via hydrogen reduction of saprolite. Sci Rep 16, 5553 (2026). https://doi.org/10.1038/s41598-026-36516-z
Keywords: nickel batteries, hydrogen smelting, low carbon metals, laterite ore, electric vehicle materials