Clear Sky Science · en
Host m6A modifications shape microbiota that drives cell specific ferroptosis as a causal pathway to chronic respiratory diseases
Why Your Microbes Matter to Your Lungs
Chronic breathing problems like asthma and chronic obstructive pulmonary disease (COPD) are usually blamed on smoking, pollution, or allergies. This study suggests another, less obvious player: the trillions of microbes living in our gut and on our skin. Using large-scale genetic data, the authors show that certain microbes may not just accompany lung disease, but help cause it—working through subtle chemical changes in our cells and a form of iron-driven cell damage. Understanding this hidden network could eventually lead to new ways to prevent or treat chronic respiratory diseases by nudging our microbes and their molecular pathways in healthier directions.
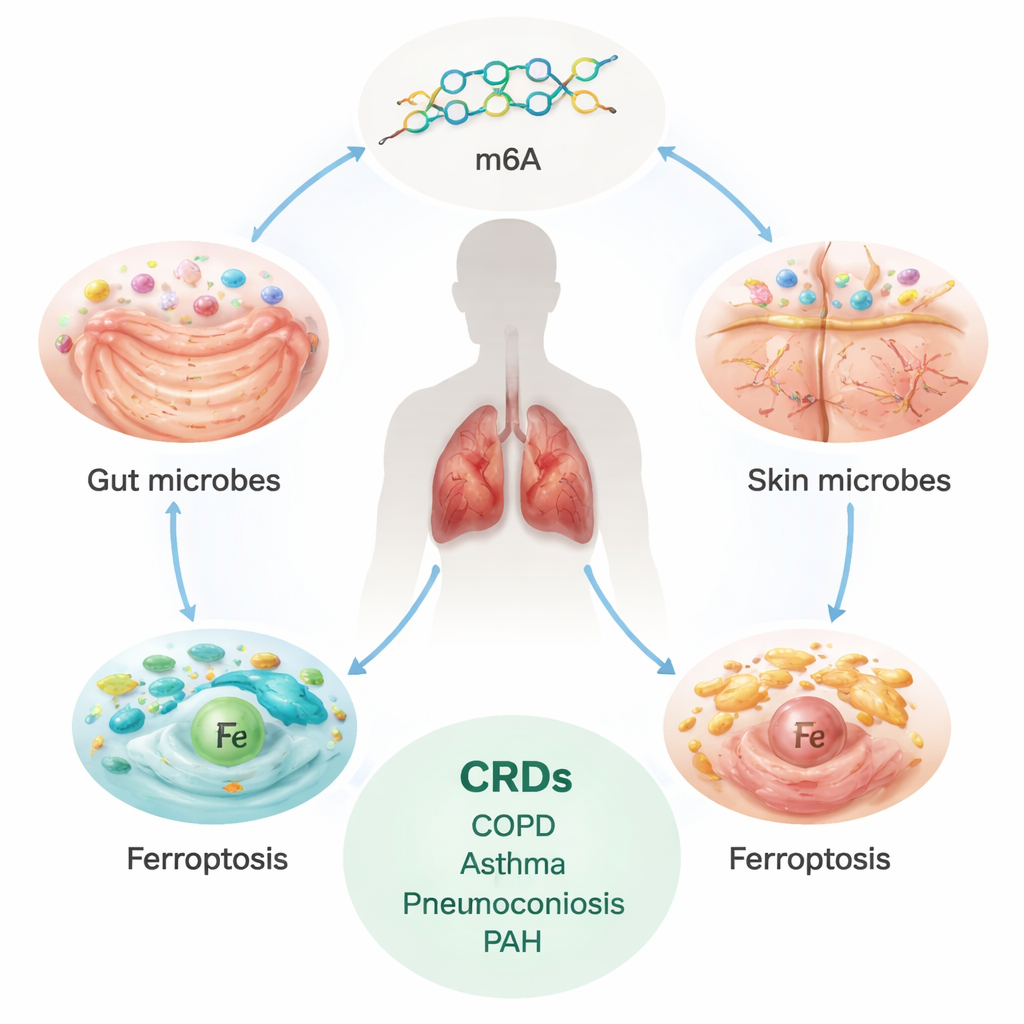
The Hidden Burden of Chronic Lung Disease
Chronic respiratory diseases, including COPD, asthma, interstitial lung disease (ILD), pneumoconiosis, and pulmonary arterial hypertension (PAH), affect hundreds of millions of people worldwide and cause millions of deaths every year. These conditions often progress slowly but relentlessly, scarring or remodeling the lungs and blood vessels and leaving people short of breath. Current treatments mainly manage symptoms; they do not fully stop or reverse the underlying damage. That has pushed scientists to search for deeper, root-level causes, especially ones that might be modified before severe disease sets in.
Microbial Neighbors in the Gut and on the Skin
We now know that the gut and skin are home to complex microbial communities that help train the immune system, shape metabolism, and maintain barrier defenses. But are these microbes actually driving lung disease, or simply changing as people get sick? To untangle cause from effect, the researchers used a method called Mendelian randomization, which leverages natural genetic differences between people as a kind of lifelong “experiment.” They combined genetic data on hundreds of gut and skin microbe types with data on five major chronic lung diseases from large European studies, testing whether inherited tendencies toward higher or lower levels of specific microbes are linked to the risk of each disease.
Microbes That Help or Harm the Lungs
The analysis identified dozens of gut microbes that either increased or decreased the risk of each respiratory disease, often in disease-specific patterns. For example, certain gut bacteria appeared to protect against COPD, asthma, or PAH, while others raised the odds of ILD or COPD. Skin microbes showed more modest but still detectable links, with some species associated with higher risk and others with lower risk across different conditions. The team also flipped the direction of analysis, finding that genetic risk for COPD, asthma, and other lung diseases, in turn, predicted changes in both gut and skin microbiota. This two-way traffic suggests a feedback loop: microbes can help shape lung disease, and lung disease can reshape our microbial ecosystems.
Two Molecular Switches: RNA Marks and Iron-Driven Cell Death
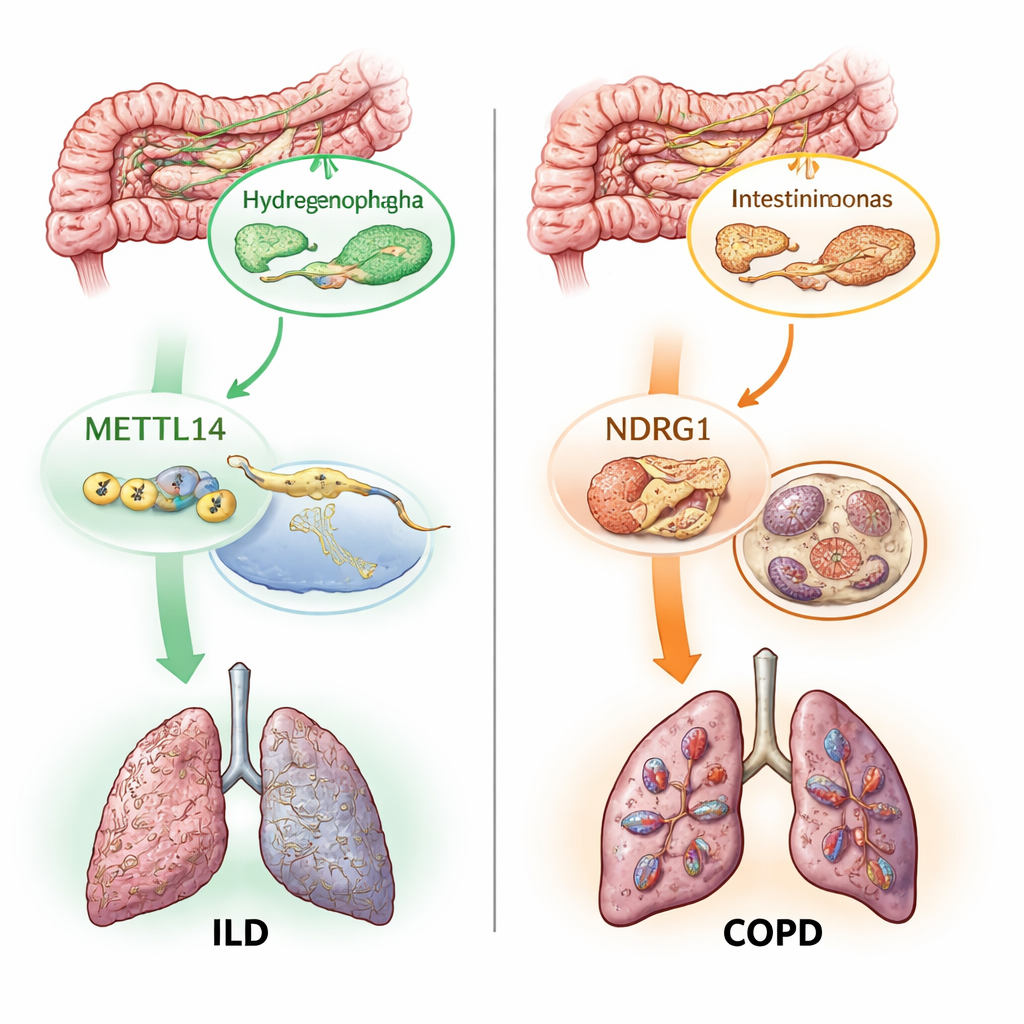
Beyond mapping which microbes matter, the authors asked how these microscopic partners might influence lung tissues. They focused on two molecular processes already suspected in lung damage. The first, called m6A modification, is a small chemical tag added to RNA that tunes how genes are turned on and off. The second, ferroptosis, is a form of iron-dependent cell death driven by runaway oxidation of fats in cell membranes. Using genetic proxies for 19 m6A-related genes and hundreds of ferroptosis-related genes, they tested whether changes in these pathways causally relate to lung disease. They found that one m6A writer gene, METTL14, was linked to lower risk of ILD, and several ferroptosis genes, including NDRG1, were linked to higher risk of COPD, ILD, and asthma.
Connecting the Dots: From Microbes to Cells to Disease
The most novel part of the work was tracing how these pieces fit into chains of cause and effect. Statistical mediation analyses suggested that some of METTL14’s protective effect on ILD runs through changes it induces in a particular gut microbe called Hydrogenophaga. In another pathway, a specific gut bacterium, Intestinimonas massiliensis, appeared to influence COPD risk partly by activating NDRG1 and the ferroptosis death program. The team then checked human lung-tissue data and confirmed that both METTL14 and NDRG1 are expressed differently in diseased versus healthy samples. Finally, using single-cell genetic maps of immune cells, they pinpointed NDRG1’s harmful effect on COPD to one immune cell subset, a type of monocyte, suggesting that future drugs targeting this gene may need to act in a cell-specific way.
What This Means for Future Treatments
For non-specialists, the key message is that lung health is not just about what we breathe but also about the microbes that live in and on us and the molecular switches they flip inside our cells. This study cannot prove every step experimentally, and it is limited mainly to people of European ancestry, but it offers strong genetic clues that gut microbes, RNA marks like m6A, and iron-driven cell death form a connected pathway leading to chronic lung damage. In the long run, this line of research could inspire new strategies that combine microbiome-based therapies with drugs targeting METTL14, NDRG1, or ferroptosis, aiming not just to ease symptoms but to interrupt the biological circuits that make chronic respiratory diseases progress.
Citation: Zhang, J., Liu, X., Liu, X. et al. Host m6A modifications shape microbiota that drives cell specific ferroptosis as a causal pathway to chronic respiratory diseases. Sci Rep 16, 5809 (2026). https://doi.org/10.1038/s41598-026-36513-2
Keywords: gut microbiome, chronic lung disease, asthma and COPD, RNA epigenetics, cell death pathways