Clear Sky Science · en
Integrative analyses of metastatic cancer transcriptome reveal clinically distinct cellular States and ecosystems
Why the Neighborhood Around Tumors Matters
Most people know that cancer can spread, or metastasize, to distant organs—and that this spreading causes the majority of cancer deaths. But fewer people realize that each metastatic tumor is more like a bustling city than a simple lump of cancer cells. It contains many kinds of normal and cancerous cells that interact, compete, and cooperate in ways that can either slow the disease or help it grow. This study maps those hidden “neighborhoods” across thousands of metastatic tumors, offering clues about why some patients live longer and respond better to treatment than others.
Looking Inside Thousands of Metastatic Tumors
The researchers analyzed gene activity data from 2,822 patients with metastatic cancer spanning 25 tumor types, including prostate, skin, and breast cancers. Instead of examining single cells under a microscope, they used advanced computational tools to “unmix” bulk tissue data, estimating what different cell types were present and how active their genes were. With a machine-learning framework called EcoTyper, they grouped cells into recurring patterns of gene activity, which they call cellular states, and then looked at how these states cluster into broader ecosystems inside tumors.
Many Cell Types, Many Personalities
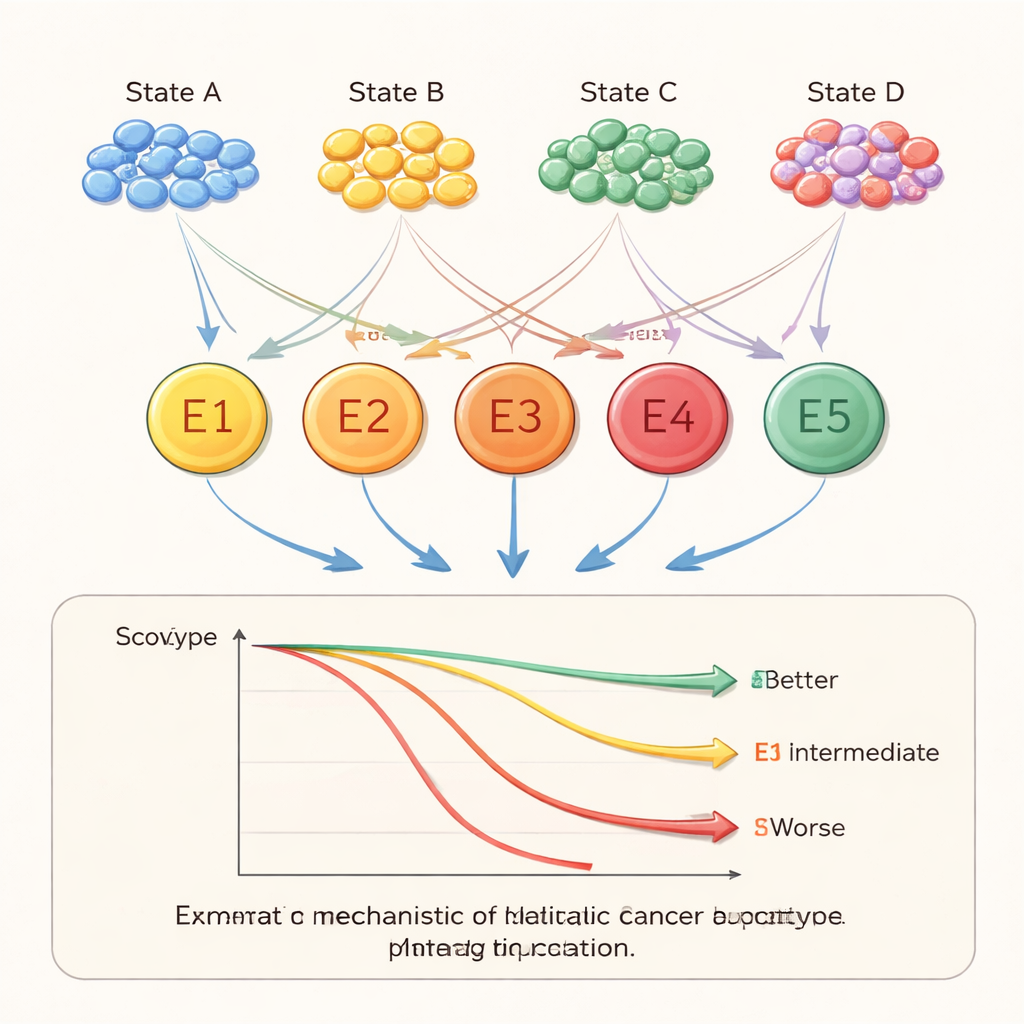
Within the metastatic tumors, the team focused on 12 major kinds of cells, such as cancerous epithelial cells, immune cells (including T cells, B cells, and natural killer cells), blood vessel cells, and fibroblasts that shape the tissue scaffold. Across these 12 types, they identified 45 distinct cellular states—essentially “personality modes” that the cells can adopt. For example, some T cell states were geared toward attack, while others appeared exhausted and less capable of fighting cancer. Certain fibroblast states were linked to remodeling the tissue in ways that can encourage tumor spread. These states did not appear randomly: their frequencies varied depending on the original cancer type and the organ where it had metastasized, revealing a strong influence of both tumor origin and local environment.
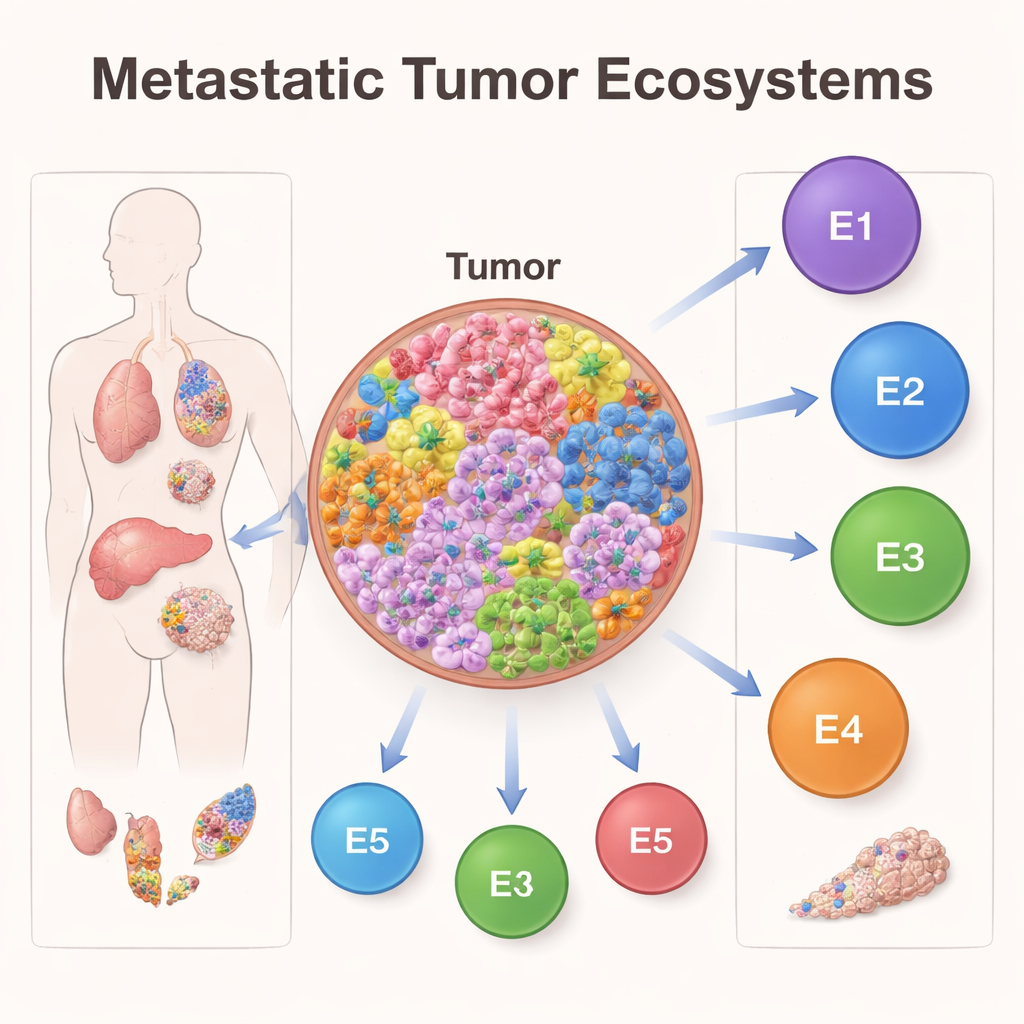
Five Tumor Ecosystems Linked to Survival
When the researchers examined which cellular states tended to appear together, they uncovered five major ecosystems, or “ecotypes,” inside metastatic tumors. Each ecotype was a characteristic mix of cell types and states. Some were rich in many immune and stromal cells, while others were more dominated by malignant cells. These ecotypes were not just academic categories—they strongly tracked with how long patients survived. One ecotype, labeled E5, was associated with relatively good outcomes, whereas others, especially E2 and E3, were tied to poorer survival. The same ecotype patterns appeared across different cancers and organs, suggesting they capture general rules of how metastatic tumors are organized.
Immune Activity, Treatment Response, and Control Switches
To understand what these ecosystems are doing, the team looked at which biological pathways were active in their genes. Many cellular states were enriched in immune functions or classic cancer pathways, such as responses to low oxygen or signals that drive cell division and invasion. One ecotype (E1) showed strong immune and inflammatory activity and high levels of immune and stromal cells, while another (E2) was enriched in growth and cell-cycle pathways linked to aggressive disease. The researchers also examined markers related to modern immunotherapies. Some ecotypes had features suggesting that tumors might evade immune attack and respond poorly to checkpoint-blocking drugs, whereas others looked more likely to benefit. Finally, by scanning for transcription factors—master switches that control gene programs—they highlighted several candidates, including SPIB, SRF, and NR1D1, that appear to help shape these ecosystems and were themselves associated with patient prognosis.
What This Means for Patients
For a non-specialist, the key message is that metastatic tumors are not all alike, even when they share the same diagnosis. They are built from recurring combinations of cell “personalities” and ecosystems that can either restrain or accelerate disease, and these patterns leave detectable footprints in gene activity data. By charting 45 cellular states and five major tumor ecosystems across thousands of patients, this work offers a high-resolution atlas of the metastatic tumor microenvironment. In the future, such maps may help doctors predict which patients are at higher risk, who is most likely to benefit from particular treatments—especially immunotherapies—and which cellular switches might be the most promising targets for new drugs.
Citation: Zhang, C., Li, S., Yu, Y. et al. Integrative analyses of metastatic cancer transcriptome reveal clinically distinct cellular States and ecosystems. Sci Rep 16, 7343 (2026). https://doi.org/10.1038/s41598-026-36512-3
Keywords: metastatic cancer, tumor microenvironment, cellular states, immune ecosystem, precision oncology