Clear Sky Science · en
Association of RAS mutational status with clinical outcomes in metastatic colorectal cancer treated with trifluridine/tipiracil or regorafenib
Why this study matters to patients and families
Metastatic colorectal cancer—colon or rectal cancer that has spread—is a leading cause of cancer death worldwide. When standard drugs stop working, patients and doctors face tough choices about what to try next. This study asks a very practical question: can simple genetic tests on a tumor help match patients to two widely used last‑line medicines, trifluridine/tipiracil and regorafenib, so that people live longer and feel better?
Looking at genes to guide tough treatment choices
Modern cancer care often begins with testing the tumor’s DNA. One key set of genes, called RAS, helps control how cells grow and divide. Changes, or mutations, in these genes can make cancer cells grow uncontrollably and resist certain drugs. The researchers reviewed medical records from 263 adults with metastatic colorectal cancer treated at a Taiwanese cancer center between 2018 and 2023. All had already received standard chemotherapy and targeted therapies and were then given either trifluridine/tipiracil alone, trifluridine/tipiracil plus another targeted drug, or regorafenib. Before starting these treatments, every patient had their RAS genes tested and was placed into one of three groups: normal RAS (RAS wild‑type), a common mutation in one spot of the KRAS gene (KRAS G12), or other, less common RAS mutations.
Three treatment paths in the real world
Trifluridine/tipiracil is a chemotherapy pill, and regorafenib is a pill that blocks several growth‑related signals in cancer cells and blood vessels. Some patients in this study received trifluridine/tipiracil by itself, while others took it in combination with an additional targeted drug that blocks tumor blood vessels or growth signals. Because this was a real‑world review and not a randomized trial, doctors chose the treatment based on each patient’s prior side effects, tumor burden, and overall condition. The investigators then tracked how long patients lived without their cancer getting worse (progression‑free survival) and how long they lived overall, as well as how often tumors shrank or at least stopped growing.
Gene differences shape which drug works better
Across the entire group, the combination of trifluridine/tipiracil plus a targeted drug clearly did best. Patients on this combination typically went about five months before their disease worsened and lived a median of just over 15 months after starting treatment—longer than those on trifluridine/tipiracil alone or on regorafenib. Tumors were more likely to shrink or stabilize with the combination as well. When the researchers looked more closely at RAS gene groups, important patterns emerged. For patients whose tumors had normal RAS or one of the rarer RAS mutations, trifluridine/tipiracil alone still outperformed regorafenib in terms of survival. However, for patients whose tumors carried the common KRAS G12 mutation, regorafenib gave better survival than trifluridine/tipiracil alone, though the combination treatment still did best of all.
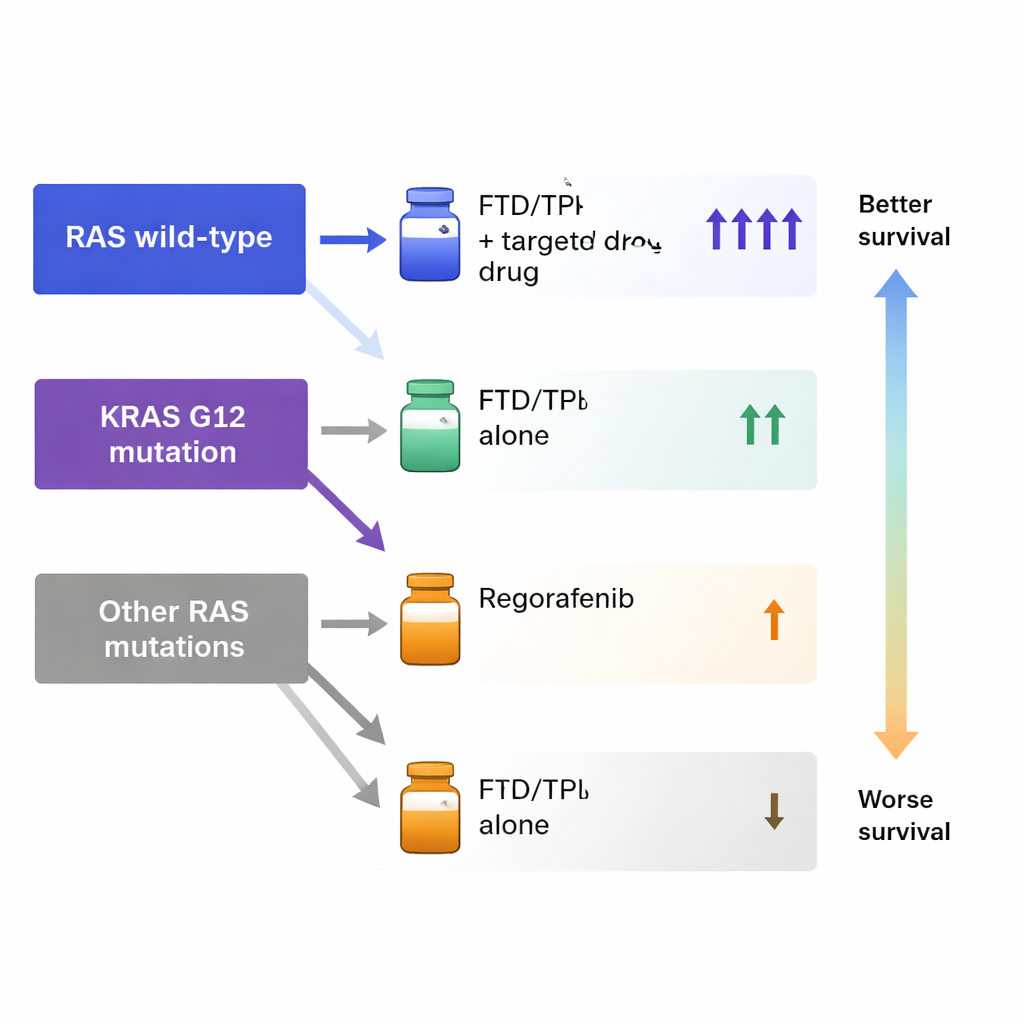
What this means for precision cancer care
These findings suggest that a relatively simple genetic test—already routinely done for many colorectal cancer patients—could help guide the choice between two difficult late‑stage treatment options. Rather than viewing trifluridine/tipiracil and regorafenib as interchangeable after standard therapy fails, doctors might favor trifluridine/tipiracil, especially in combination with a targeted drug, for most patients. For the subgroup with KRAS G12 mutations, regorafenib may be the better single‑drug option if combination therapy is not possible. The study also hints that other clinical features, such as the original stage of the cancer and certain DNA repair markers, may further refine these decisions.
Take‑home message for non‑specialists
For people living with advanced colorectal cancer, this research offers cautious but hopeful news: the genetic fingerprint of a tumor, particularly changes in RAS genes, can help point toward the medicine most likely to buy more time. In this study, adding a targeted drug to trifluridine/tipiracil gave the longest survival for nearly all patients, no matter what their RAS status was. Among those with a specific KRAS G12 mutation, regorafenib appeared safer to choose than trifluridine/tipiracil alone. Because this was a retrospective, single‑center analysis, its results need to be confirmed in larger, randomized trials. Still, it provides a practical, gene‑based roadmap that doctors can consider today when tailoring last‑line treatment for metastatic colorectal cancer.
Citation: Hsieh, MC., Rau, KM., Liu, KW. et al. Association of RAS mutational status with clinical outcomes in metastatic colorectal cancer treated with trifluridine/tipiracil or regorafenib. Sci Rep 16, 5294 (2026). https://doi.org/10.1038/s41598-026-36509-y
Keywords: metastatic colorectal cancer, RAS mutation, KRAS G12, trifluridine tipiracil, regorafenib