Clear Sky Science · en
Integrated BSI bacteria identifier-on-chip using approximate k-mer matching
Why faster germ detection matters
For patients receiving life-saving stem cell transplants, infections in the bloodstream can turn recovery into a medical emergency within hours. Doctors know that every minute counts, but today’s lab tools for finding the culprit bacteria can be slow or require powerful computers. This study presents a tiny, energy-efficient computer chip—called PC-CAM—that can rapidly scan DNA from patient samples and flag dangerous bacteria in real time, potentially at the hospital bedside.
The infection risk after a gut-saving treatment
Some transplant patients develop a severe complication in which the donated immune cells attack the patient’s gut. One promising treatment is fecal microbiota transplantation, where healthy gut microbes from a donor are introduced to the patient. While this can rescue the gut, it sometimes brings an added danger: harmful bacteria from the intestine can escape into the bloodstream, causing life-threatening infections. To treat these infections effectively, doctors must quickly identify which bacterial species are present, but traditional tests can take hours to days and may miss unfamiliar strains.
From slow genome piecing to quick DNA fragments
Modern DNA sequencers can read genetic material directly from blood or stool samples in near real time. The bottleneck is no longer reading the DNA, but interpreting it. Conventional methods first try to reconstruct whole genomes from millions of short DNA fragments and then match these assembled genomes to a reference library—a computationally heavy process. The authors replace this with a simpler strategy: they break DNA into short pieces of fixed length, called k-mers, and look for these pieces directly in a database of known bacterial sequences. Instead of demanding a perfect letter-for-letter match, their system tolerates a certain number of differences, which allows it to cope with the inevitable errors of DNA sequencing and natural mutations. 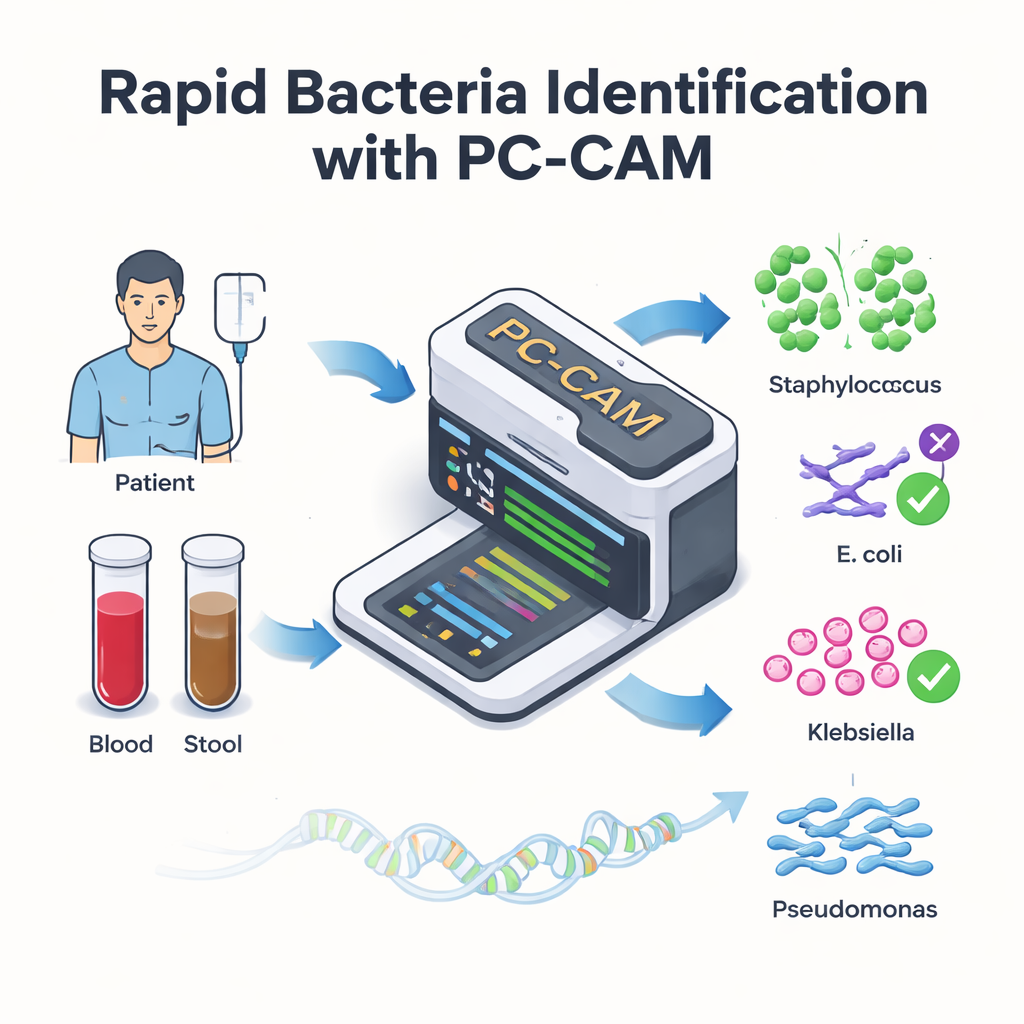
A smart memory chip built for DNA searching
At the heart of PC-CAM is a specialized type of memory called content-addressable memory, which can compare incoming data against everything it stores at once. The researchers designed an “approximate search” version of this memory (ACAM) that can be tuned to accept near-matches rather than exact ones. Each short DNA fragment is encoded and stored as a row in this memory. When a new fragment from a patient sample arrives, the chip activates a search line through all rows simultaneously and measures how quickly tiny electrical signals discharge. By adjusting two control voltages, the system sets a threshold for how many mismatches it will tolerate, effectively dialing in how forgiving the match should be. This hardware-level comparison allows the chip to sift through patterns far faster and with less energy than a general-purpose computer.
How well the chip finds real bacteria
The team tested PC-CAM on real-world datasets from patients who had undergone fecal microbiota transplantation, comparing bacteria detected in their blood and stool to the results of a full, software-based genome analysis. Even when the chip stored only a small, carefully chosen subset of DNA fragments from each bacterial genome, it largely agreed with the more computationally expensive method, correctly identifying key pathogens in multiple patients. In extended experiments using simulated DNA reads from various bacteria and different error rates, the chip’s sensitivity (its ability to catch true matches) rose as it was allowed to tolerate more differences, while specificity (its ability to avoid false alarms) declined at very high tolerance levels. The authors show that simple post-processing steps—such as requiring multiple agreeing fragments or discarding weak matches—can reduce these false positives. 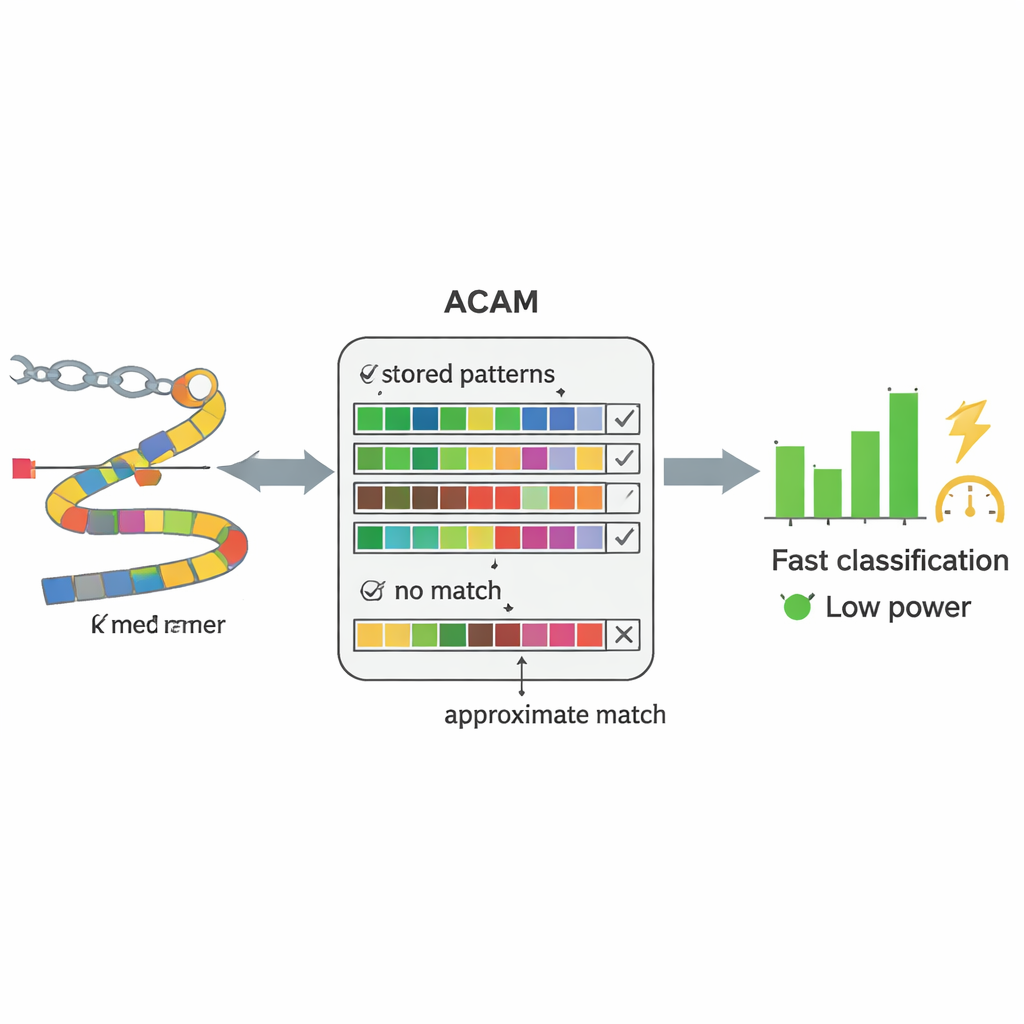
Tiny hardware with big speed and low power
PC-CAM integrates the ACAM memory with a small RISC-V processor on a chip just a few square millimeters in size, fabricated in a standard 65-nanometer technology. Measurements on real hardware show that it can classify about 960,000 short DNA reads every second while consuming around 1.27 milliwatts of power—less than many digital wristwatches. When compared with a leading software tool running on a high-end desktop processor, the chip was roughly 1,900 times faster for this kind of classification task. Although its accuracy is modestly reduced by storing a diluted version of each genome, the authors argue that this trade-off is acceptable when speed and power are the primary constraints, such as in emergency care or portable devices.
What this means for patients and beyond
In plain terms, the study shows that a tiny, low-power chip can act as a specialized search engine for bacterial DNA, spotting dangerous germs in blood or stool samples almost instantly. While it does not replace full-blown genetic analysis in a major laboratory, it can provide rapid, on-the-spot guidance about which bacteria are present, helping doctors choose antibiotics sooner and more accurately. The same approach could be extended to monitoring antibiotic resistance, checking food and water for contamination, or watching crops for disease—all using compact hardware that brings sophisticated DNA analysis out of the data center and closer to where decisions must be made.
Citation: Garzón, E., Galindo, V., Harary, Y. et al. Integrated BSI bacteria identifier-on-chip using approximate k-mer matching. Sci Rep 16, 5722 (2026). https://doi.org/10.1038/s41598-026-36497-z
Keywords: bloodstream infection, rapid pathogen detection, DNA sequencing, lab-on-a-chip, microbiome