Clear Sky Science · en
An integrative clinical and bioinformatic analysis identifies MicroRNAs as biomarkers of ischemic stroke severity
Why a blood test for stroke matters
When someone has a stroke, every minute counts. Yet even with modern brain scans, it can be hard to quickly judge how severe the damage is and who is at highest risk of a poor recovery. This study explores whether tiny molecules circulating in the blood, called microRNAs, could serve as fast, reliable fingerprints of what is happening inside the brain during an ischemic stroke. If successful, such a blood test might help doctors triage patients more effectively, guide treatment choices, and better explain likely outcomes to families.
Tiny messengers in the bloodstream
MicroRNAs are very small pieces of genetic material that help control how genes are turned on and off. Cells in the body, including blood cells and brain cells, release microRNAs into the bloodstream, where they are surprisingly stable and easy to measure. Previous animal and human studies have hinted that certain microRNAs change after a stroke and might mirror how badly the brain is injured. The authors focused on a panel of microRNAs already linked to heart and brain disease, especially miR-16-5p and two closely related forms called miR-125a-3p and miR-125a-5p, to see whether their levels in whole blood could distinguish stroke patients from high‑risk controls and reflect stroke severity.
How the study was carried out
The research team enrolled 60 people admitted with an acute ischemic stroke and compared them with 30 age- and sex‑matched individuals who had cardiovascular risk factors but no stroke. Blood samples were taken soon after hospital arrival and again seven days later. Using a sensitive laboratory method, quantitative PCR, the scientists measured the amount of each selected microRNA in the blood. They then compared levels between groups and tracked how they changed over time. To judge clinical severity and outcome, they used two common scales in stroke medicine: the NIH Stroke Scale, which rates neurological deficit, and the modified Rankin Scale, which measures how disabled a person is in daily life.
What the blood signals revealed
The study found clear differences in microRNA patterns between stroke patients and controls. MiR-125a-3p was consistently lower in stroke patients both at admission and on day seven, while miR-125a-5p was higher at admission but dropped by the end of the first week. Both offered moderate ability to distinguish stroke patients from controls, suggesting potential diagnostic value. The most striking signal, however, came from miR-16-5p. Patients who arrived with higher miR-16-5p levels were more likely to have moderate-to-severe neurological deficits and worse functional outcomes a month later. Even after accounting for age, sex, inflammation (measured by C‑reactive protein), smoking, heart disease, and platelet counts, high miR-16-5p and elevated C‑reactive protein remained independent predictors of an unfavorable early outcome.
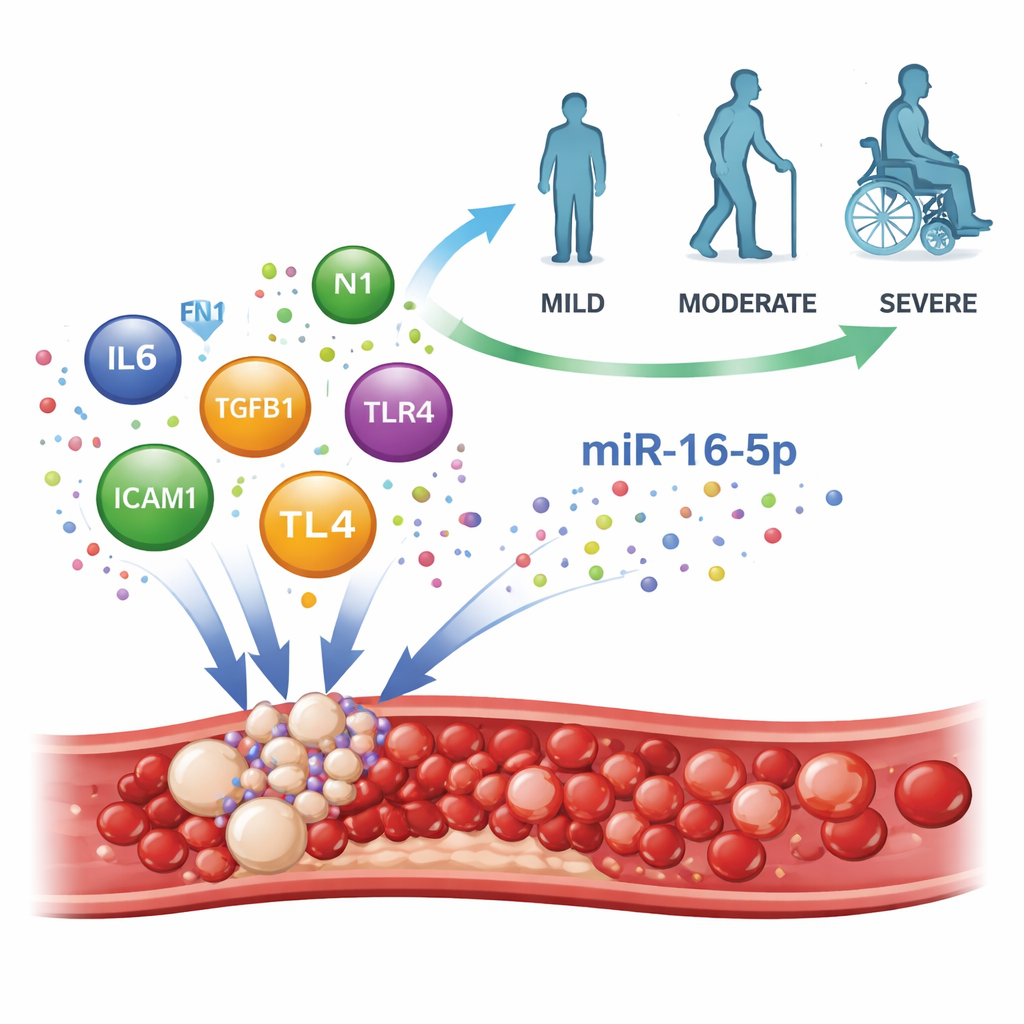
Clues to the biology behind the numbers
To understand why miR-16-5p might track stroke severity, the authors used large biological databases and computer models to map which genes and pathways this microRNA can influence. They found that miR-16-5p targets were heavily involved in responses to low oxygen, blood clotting, platelet activation, and inflammation—core processes in ischemic stroke. Network analysis highlighted several key molecules, including IL‑6, fibronectin (FN1), TGF‑β1, ICAM‑1, and TLR4, all known to shape how blood vessels, platelets, and immune cells behave after a stroke. Machine‑learning methods further underscored that high miR-16-5p, together with signs of inflammation, helped classify patients with more severe disability, supporting the idea that this microRNA sits at the crossroads of clotting and inflammatory damage.
What this could mean for patients
For non-specialists, the takeaway is that a simple blood draw may one day help doctors see beyond the brain scan and into the invisible molecular storm of a stroke. In this study, different microRNAs showed distinct patterns soon after ischemic stroke, with miR-16-5p standing out as a marker linked to greater early brain injury and poorer short‑term outcome, while miR-125a-3p and miR-125a-5p aided in distinguishing stroke from high‑risk controls. These findings strengthen the idea that platelet‑derived and inflammation‑related microRNAs mirror the biological events that drive stroke damage. However, the authors caution that miR-16-5p is not unique to stroke and is also influenced by age and other cardiovascular conditions. Larger, long‑term studies are needed before such tests can guide everyday care, but this work lays important groundwork toward blood‑based tools that could help personalize stroke diagnosis and prognosis.
Citation: Eyileten, C., Wicik, Z., Shahzadi, A. et al. An integrative clinical and bioinformatic analysis identifies MicroRNAs as biomarkers of ischemic stroke severity. Sci Rep 16, 6242 (2026). https://doi.org/10.1038/s41598-026-36494-2
Keywords: ischemic stroke, microRNA biomarkers, stroke severity, platelet activation, inflammation