Clear Sky Science · en
Genomic profiling of extended-spectrum beta-lactamase-producing Escherichia coli isolated from poultry and poultry farm workers in Accra, Ghana
Why germs in chickens matter to all of us
Antibiotics have transformed medicine, but their power is fading as more bacteria learn to resist them. This study from Ghana looks at a surprisingly important source of hard-to-treat germs: everyday poultry farms on the edges of a big city. By examining bacteria from both chickens and the people who care for them, the researchers show how farm practices can help create and share dangerous, drug‑resistant microbes that may ultimately affect anyone who eats chicken, lives near farms, or needs antibiotics to treat an infection.
Chicken farms next to family homes
In many parts of Ghana, small- and medium-sized poultry farms sit very close to homes and shops. Farmers often rely on antibiotics to keep flocks healthy and to protect their investment, sometimes without strict veterinary guidance. The team visited 20 such farms around Accra, the capital, and collected 300 swabs from chickens and 60 stool samples from farm workers. They focused on a kind of Escherichia coli (E. coli) that produces enzymes called extended‑spectrum beta‑lactamases, or ESBLs. These enzymes can break down important antibiotics like many penicillins and cephalosporins, turning once‑reliable drugs into blunt tools.
How common were these tough germs?
The findings were striking. Among the E. coli recovered, more than four out of five chicken isolates and over two out of three human isolates made ESBLs. Nearly every one of these bacteria was resistant to key antibiotics used in hospitals, and many were also resistant to older, cheaper medicines such as tetracycline and sulfamethoxazole‑trimethoprim. This pattern means that common first‑line drugs may fail if these germs cause infections, pushing doctors toward more expensive or scarce options.
Inside the bacterial toolbox
To understand what makes these bacteria so formidable, the researchers sequenced the full DNA of 17 especially drug‑resistant strains taken from both chickens and workers. They found a recurring gene, called blaCTX-M-15, in every one of them. This gene is already notorious in hospitals worldwide for helping E. coli shrug off powerful antibiotics. It was often found together with many other resistance genes that protect the bacteria from several different drug families at once. The team also discovered gene sets usually linked to disease‑causing E. coli in birds—features that help the bacteria cling to cells, steal iron from the body, and survive attacks from the immune system. In some strains, the same genetic backgrounds and traits appeared in both poultry and people on the same farm, hinting that bacteria—or the resistance DNA they carry—can move between species.
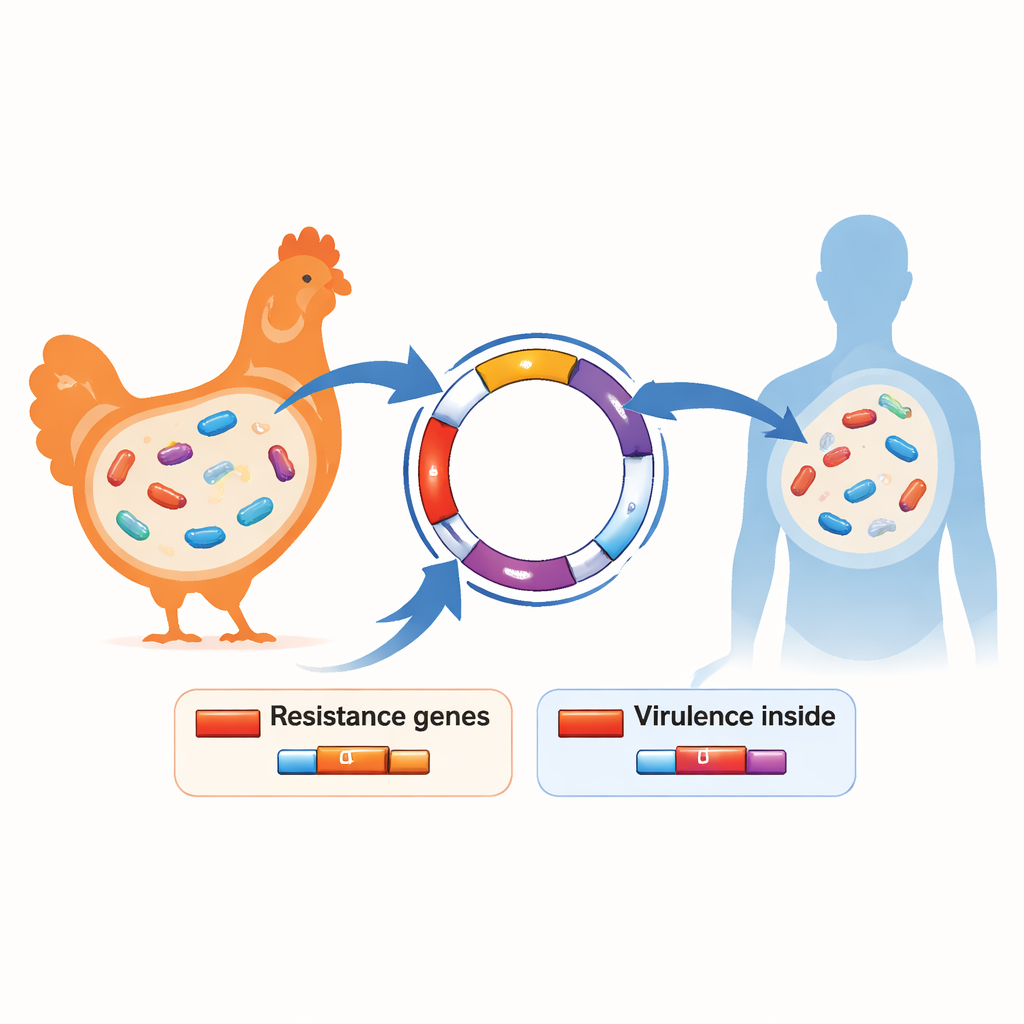
Traveling genes and shared risks
Another key piece of the puzzle was the plasmids—small circular bits of DNA that bacteria can swap like trading cards. The study found many different plasmid types, several of which were loaded with resistance and virulence genes. Because plasmids can jump from one bacterium to another, even across related species, they act as high‑speed shuttles spreading resistance through the farm environment and, potentially, into nearby communities. Genetic fingerprints showed that some bacterial lineages common in human medicine, such as certain sequence types known to cause serious infections, are also thriving in chickens. That overlap reinforces the idea that farms can be both a source and a mixing ground for troublesome strains.
What this means for farmers and the public
For non‑scientists, the message is clear: the way we raise animals can directly influence how well our antibiotics work. The study shows that poultry farms in peri‑urban Ghana harbor large numbers of E. coli that are resistant to many important drugs and that share genetic features with bacteria infecting people. These germs are boosted by frequent antibiotic use and move more easily when hygiene and biosecurity are weak. The authors argue that simple steps—using antibiotics only when truly needed, improving cleanliness and protective gear on farms, and tracking resistance genes through ongoing DNA monitoring—can help slow the spread of these dangerous bacteria. Protecting the effectiveness of antibiotics, they conclude, will require treating animal health, human health, and environmental health as tightly linked parts of the same system.
Citation: Okyere, I.J., Semevor, G.O., Ablordey, A. et al. Genomic profiling of extended-spectrum beta-lactamase-producing Escherichia coli isolated from poultry and poultry farm workers in Accra, Ghana. Sci Rep 16, 7400 (2026). https://doi.org/10.1038/s41598-026-36471-9
Keywords: antimicrobial resistance, poultry farming, Escherichia coli, Ghana, antibiotic stewardship