Clear Sky Science · en
Identification of bacterial key genes and therapeutic targets in hypertensive patients with type 2 diabetes through bioinformatics analysis
Why Your Gut Matters to Blood Pressure and Blood Sugar
High blood pressure and type 2 diabetes often show up together, raising the risk of heart attacks, strokes, and kidney disease. Doctors have long treated these conditions separately, focusing on drugs that act on the heart, blood vessels, or insulin. This study asks a different question: could tiny microbes living in our intestines—and the genes they carry—help drive both problems at once, and might they reveal new treatment options?
The Hidden World Inside the Intestine
The researchers analyzed stool samples from 124 adults, comparing 29 people with both hypertension and type 2 diabetes to 95 healthy volunteers. Using genetic sequencing of bacterial markers, they built a detailed picture of which microbes were present and how varied each person’s gut community was. People with both conditions had richer, more evenly distributed bacterial communities that clustered separately from those of healthy individuals, meaning their gut ecosystems were clearly reorganized rather than just slightly disturbed.
A Microbial Shift from Helpful to Harmful
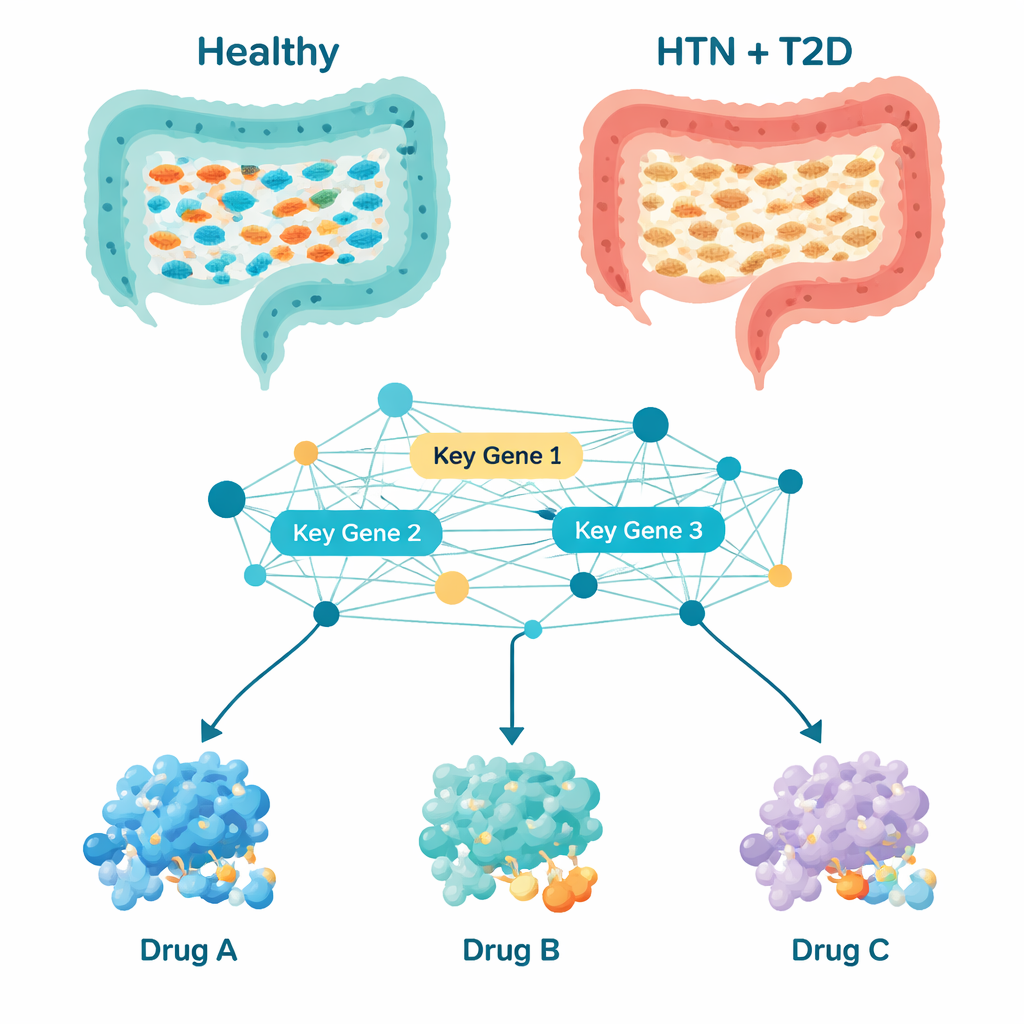
When the team zoomed in on which types of bacteria were most abundant, they saw a distinct pattern. In healthy people, bacteria that help produce short-chain fatty acids—compounds known to support gut lining health and help control inflammation and metabolism—were common. These included groups like Bacteroides, Prevotella, Roseburia, and Akkermansia. In people with both hypertension and diabetes, many of these helpful microbes were depleted. At the same time, bacteria linked in other studies to inflammation and metabolic imbalance, such as Megasphaera, Lactobacillus, Streptococcus, and Veillonella, were more common. Nineteen bacterial groups were consistently different between patients and healthy controls, suggesting a coordinated shift toward a gut environment that may favor low-grade inflammation and metabolic stress.
From Microbes to Molecular “Control Knobs”
Counting microbes alone cannot explain how they influence the body, so the scientists used computational tools to infer what the gut bacteria are likely doing. They predicted which metabolic pathways—chains of chemical reactions—were more or less active in patients. Out of more than a thousand pathways, 195 stood out as altered. Many related to how bacteria make proteins, process energy, and handle building blocks like nucleotides and amino acids. By building a network of how the corresponding bacterial proteins interact, the team pinpointed ten “hub” genes that sat at key junctions in this network. These genes, with names like gltB, gyrB, fusA, and mdh, act like control knobs for core bacterial functions such as energy production, DNA copying, protein synthesis, and fatty acid and nucleotide metabolism. Because these activities are tightly connected to inflammation, blood vessel health, and glucose control, changes in these microbial genes may ripple outward to affect blood pressure and blood sugar.
Searching for New Uses for Existing Drugs
Armed with this list of bacterial control genes, the researchers turned to computer-based drug screening. They gathered 189 drugs already studied for high blood pressure, type 2 diabetes, or related metabolic problems, and used molecular docking—essentially a 3D puzzle—to see which compounds might stick most tightly to the proteins made by the ten key bacterial genes. Three candidates stood out: Naringin and Neohesperidin, plant-derived compounds found in citrus fruits, and Bromocriptine, a drug already approved for type 2 diabetes. Detailed simulations over 100 billionths of a second suggested that complexes involving Neohesperidin and Bromocriptine were especially stable, meaning these drugs might reliably bind and influence the target bacterial proteins in real life. Additional checks of how drug-like these compounds appear, and how they might be absorbed, distributed, and cleared by the body, pointed to Bromocriptine as the most practical near-term candidate, although all three will require careful safety and dosing studies.
What This Means for Future Care
In plain terms, this work maps a three-step chain: people with both high blood pressure and type 2 diabetes tend to have a distinct gut microbiome; that microbiome carries a set of bacterial genes that may push metabolism and blood vessels in harmful directions; and some existing drugs appear capable of targeting those bacterial genes. The study does not prove that changing these microbes or their genes will cure disease, nor does it test any treatments in patients. But it lays out a blueprint for microbiome-based diagnostics and therapies, where doctors might one day modify gut bacteria or use tailored drugs to ease the combined burden of high blood pressure and diabetes.
Citation: Rahat, M.T.I., Sumi, M.S.A., Nurejannath, M. et al. Identification of bacterial key genes and therapeutic targets in hypertensive patients with type 2 diabetes through bioinformatics analysis. Sci Rep 16, 6431 (2026). https://doi.org/10.1038/s41598-026-36467-5
Keywords: gut microbiome, hypertension, type 2 diabetes, bacterial genes, drug repurposing