Clear Sky Science · en
African swine fever outbreaks in German pig holdings – experiences, epidemiological considerations and genome sequences
Why a Pig Virus Matters to All of Us
African swine fever is a deadly viral disease of pigs that does not infect humans but can devastate farms, food supplies, and rural economies. Once the virus enters a country, it is notoriously hard to remove. This study takes a close look at nine outbreaks of African swine fever in German pig farms between 2021 and 2024, asking how the virus got in, how it moved through herds, and what detailed genetic analyses of the virus can reveal about its journeys.
Germany’s Battle with a Costly Pig Disease
After African swine fever reached Europe from the Caucasus region in 2007, it spread widely through wild boar and domestic pigs. Germany first detected infected wild boar in 2020, and by late 2025 had recorded 18 outbreaks in domestic pig holdings. This paper focuses on nine of those outbreaks that occurred outside a special 2024 cluster, offering the first full overview of their circumstances. The farms ranged from two-pig backyard holdings to large commercial units with thousands of animals and generally good hygiene. Despite these differences, each outbreak raised the same uncomfortable question: how did a virus carried mainly by wild boar and people’s movements breach farm defenses?
Different Farms, Similar Weak Spots
The nine outbreaks paint a varied picture. In eastern Germany, several farms lay inside or near zones where infected wild boar were already known. In small backyard farms with almost no protective barriers, investigators judged it very likely that contaminated grass, bedding, or dirt carried in on boots brought the virus from the wild into pig pens. In contrast, some large, well-fenced farms far from any known wild boar cases still became infected. In these cases, investigators suspected what they call “point introductions” linked to human activity: perhaps a tiny lapse in hygiene, an item of contaminated equipment, or visitors and seasonal workers coming from affected countries. Often, the exact route could not be proven, only ranked from more to less likely.
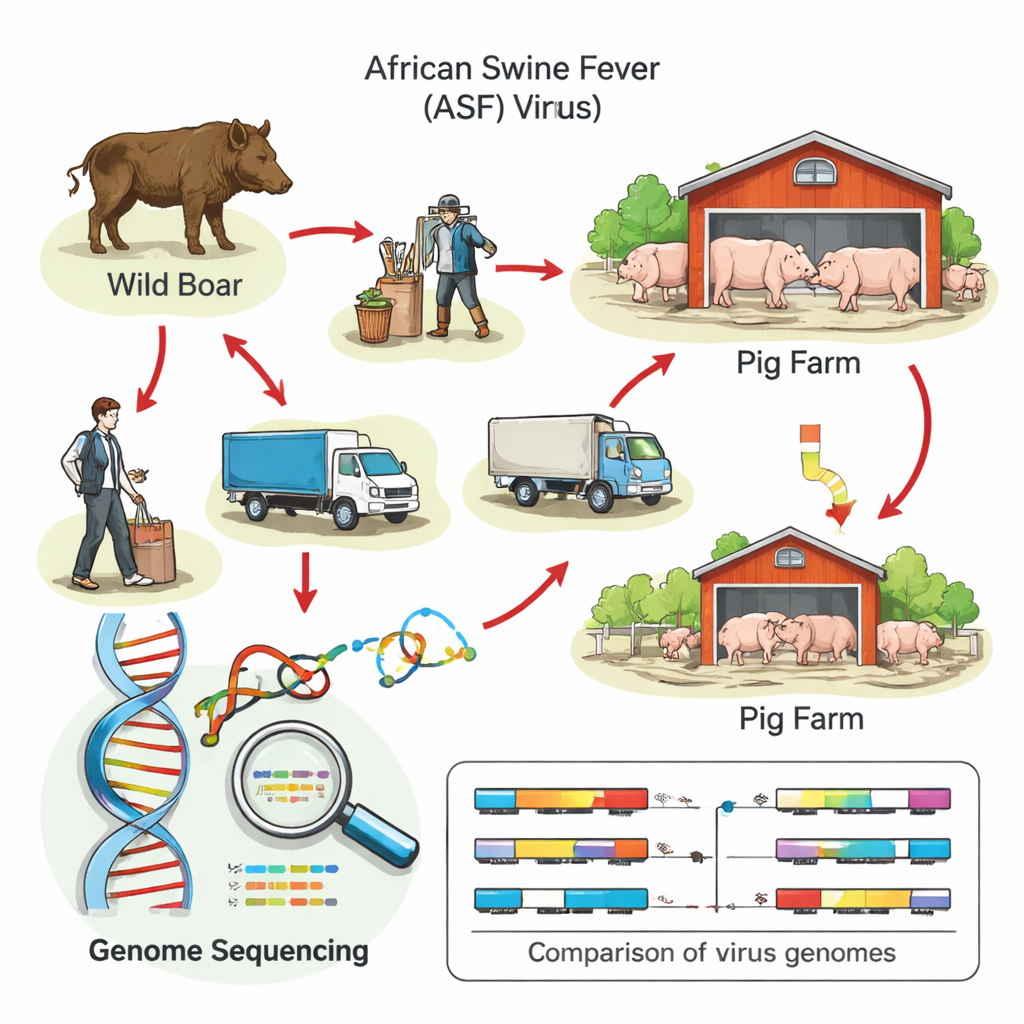
What the Virus’s Genetic Fingerprints Reveal
To go beyond educated guesswork, the researchers turned to whole-genome sequencing—reading out the virus’s full genetic code. By comparing each outbreak strain to a reference virus and to viruses found in wild boar, they could group them into distinct genetic “lineages” and identify telltale mutations that act like barcodes. Many farm outbreaks in Brandenburg and Mecklenburg-Western Pomerania carried the same genetic markers as nearby wild boar viruses, supporting the idea that infection pressure from the surrounding landscape played a key role. In one striking case in southern Germany, however, the virus lacked every marker seen in the country’s wild boar and instead closely matched strains from Moldova and Italy, pointing to a separate introduction from abroad rather than silent spread inside Germany.
Slow Spread Inside Farms, Fast Consequences Outside
The study also challenges the image of African swine fever as a “fast and furious” disease. On several farms, only one compartment or a single group of pigs was affected—even weeks or months after the virus likely arrived. Where pigs were split into clearly separated units and basic internal hygiene was respected, the virus moved surprisingly slowly between them. By contrast, in small holdings where all pigs mixed freely, every animal soon became infected. In multiple cases, veterinarians first treated sick pigs for presumed bacterial illness, delaying tests for African swine fever. That delay not only risked further spread but also blurred farmers’ memories about movements of people and materials, making it harder to reconstruct how the virus entered in the first place.
Lessons for Keeping Farms Safe
For readers outside veterinary science, the main takeaway is both sobering and hopeful. There is no vaccine yet for African swine fever, so prevention hinges almost entirely on biosecurity—the everyday habits that keep virus-laden mud, meat scraps, or tools from crossing the farm gate. This study shows that outbreaks can strike tiny backyard pens and high-tech facilities alike, often through small, easily overlooked lapses. At the same time, the careful matching of virus genomes between wild boar and domestic pigs demonstrates that modern genetic tools can help investigators trace likely infection routes and distinguish local spillover from long-distance introductions. The authors conclude that strict, consistently followed hygiene rules, ongoing training and awareness, and smart use of genome sequencing together offer the best chance to shield pig farms—and the livelihoods that depend on them—from this costly disease.
Citation: Schulz, K., Calvelage, S., Rogoll, L. et al. African swine fever outbreaks in German pig holdings – experiences, epidemiological considerations and genome sequences. Sci Rep 16, 4350 (2026). https://doi.org/10.1038/s41598-026-36441-1
Keywords: African swine fever, pig farms, wild boar, biosecurity, genome sequencing