Clear Sky Science · en
Pseudolaric acid B promotes lung cancer cells ferroptosis depending on JNK/ERK-mediated upregulation of survivin
Plant Compound Takes Aim at Lung Tumors
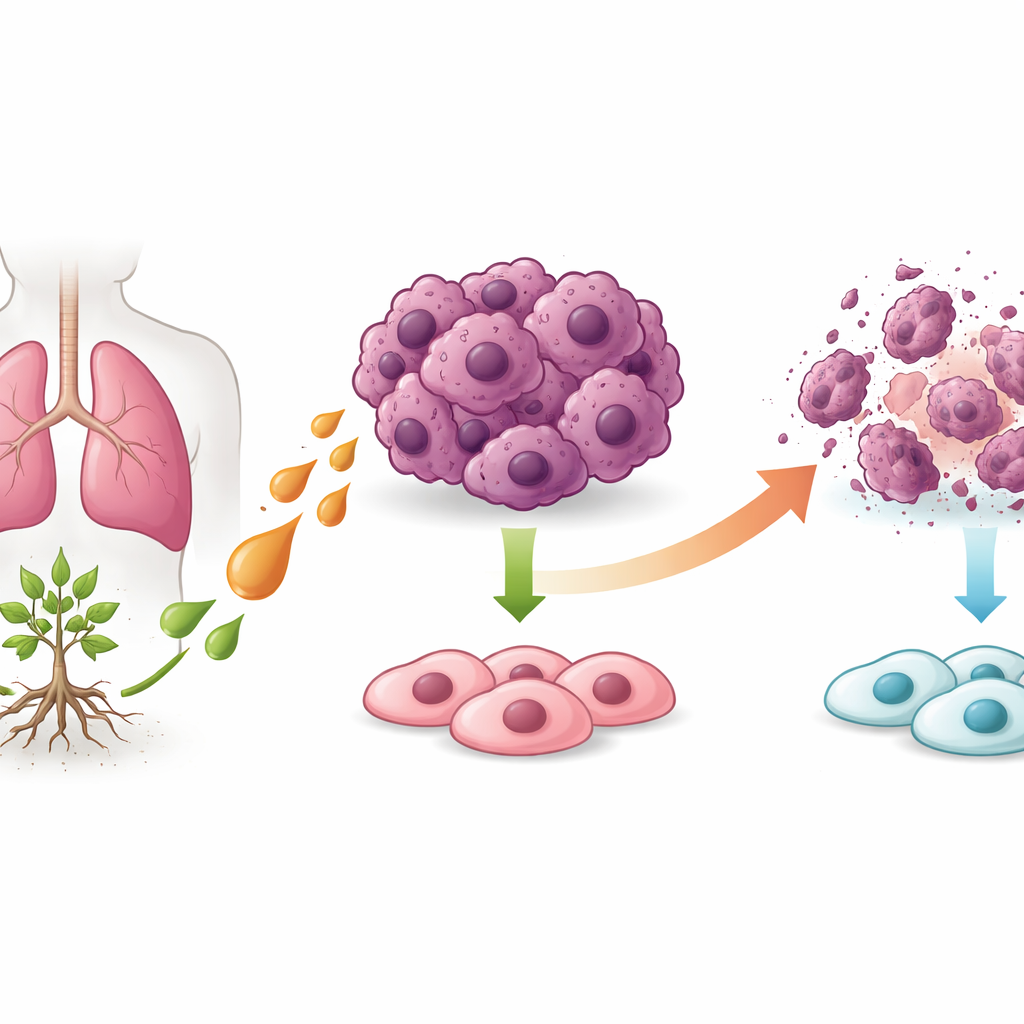
Lung cancer remains one of the deadliest cancers worldwide, and many patients either do not respond to modern drugs or suffer serious side effects. This study explores a natural substance called pseudolaric acid B, extracted from the bark of a traditional Chinese medicinal tree, to see whether it can selectively kill lung cancer cells while sparing healthy lung tissue. The researchers uncover an unexpected way this compound destroys tumor cells by triggering a special form of cell death that depends on iron and fat damage inside the cells.
Why a Tree Bark Molecule Matters
Doctors urgently need new treatments that can defeat drug-resistant tumors without harming normal cells. Plant-derived medicines have already given us major cancer drugs such as paclitaxel and vincristine. Pseudolaric acid B has shown anticancer effects in several tumor types, but how it works in lung cancer has been unclear and sometimes conflicting. Here, scientists tested pseudolaric acid B on human lung cancer cells and normal bronchial cells in the lab, and then in mice bearing human lung tumors, to map out exactly how this natural compound acts.
Selective Attack on Cancer Cells
In cell culture, pseudolaric acid B strongly slowed the growth of two different lung cancer cell lines but had little impact on normal bronchial cells. The treated cancer cells stopped multiplying, lost their ability to form dense colonies, and showed clear signs of damage. Interestingly, a classic form of cell suicide called apoptosis played only a minor role: blocking key suicide enzymes or shutting down the recycling process known as autophagy did not rescue the cells. At the same time, proteins known as “inhibitors of apoptosis,” which usually help cancer cells survive, were sharply reduced, suggesting that the drug was pushing cells toward a different death pathway.
Iron-Driven Cell Death Inside Tumors
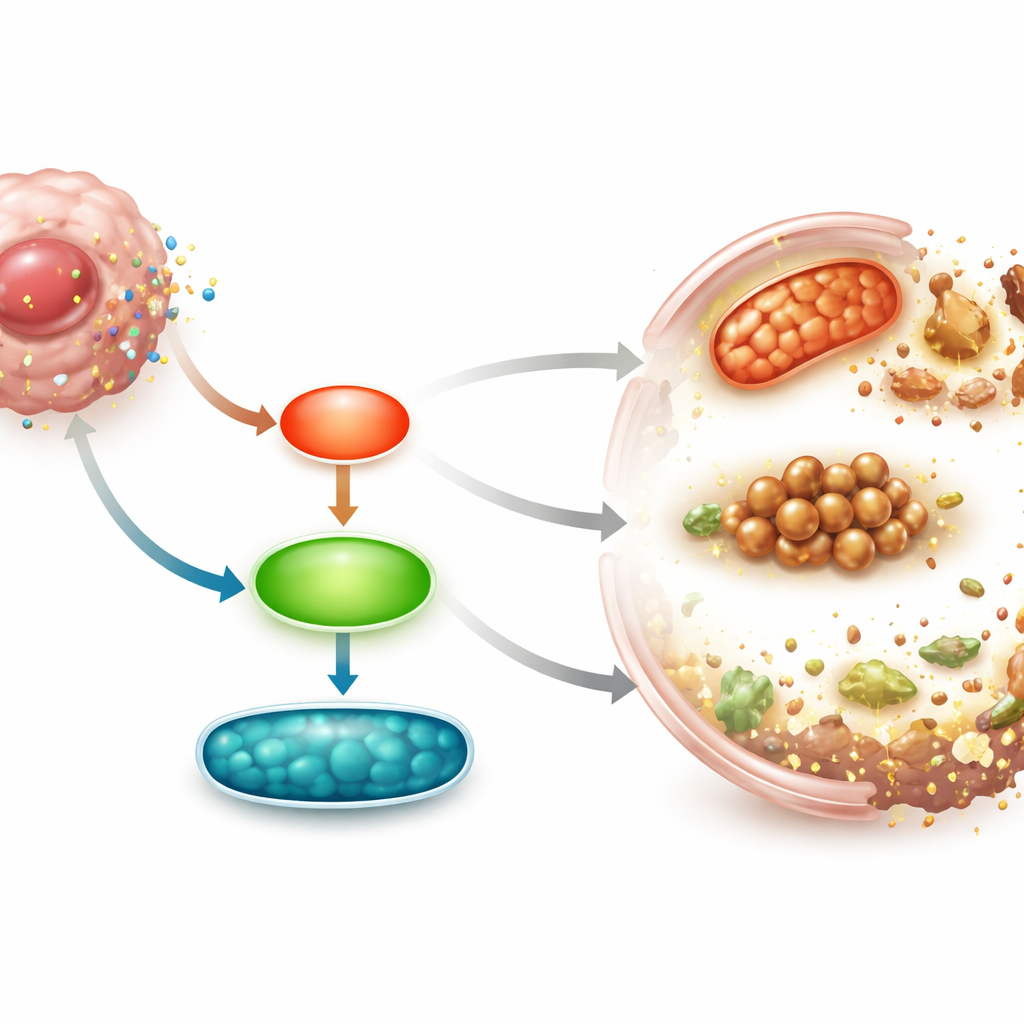
By analyzing gene activity across the entire genome, the team found that many of the affected genes pointed to ferroptosis, a regulated type of cell death driven by iron and the breakdown of cell membrane fats. They then looked inside the cells and saw telltale hallmarks of ferroptosis: shrunken, damaged mitochondria; high levels of reactive oxygen species; increased ferrous iron; and more oxidized lipids in the cell membranes. Key protective proteins that normally guard against ferroptosis, such as SLC7A11 and GPX4, were reduced at both the protein and RNA levels. When the researchers added an iron-chelating drug that blocks ferroptosis, cell death, iron buildup, and lipid damage were all strongly reversed, confirming that pseudolaric acid B mainly kills lung cancer cells through ferroptosis.
An Unexpected Helper: Survivin and Cell Signals
One surprise was the behavior of a protein called Survivin, a member of the same family as the inhibitors of apoptosis. While most of these survival proteins were decreased by treatment, Survivin levels rose sharply at the protein level, even though its RNA did not change. When the scientists blocked Survivin using a small-molecule inhibitor or gene-silencing RNA, pseudolaric acid B lost much of its killing power: lung cancer cells survived better, iron and lipid damage fell, and the protective ferroptosis proteins returned. Further pathway analysis revealed that two signaling routes inside cells, known as JNK and ERK, became more active after treatment. Inhibiting these signals reduced ferroptosis, restored the survival proteins, and lowered Survivin levels, showing that pseudolaric acid B triggers ferroptosis through a JNK/ERK–Survivin axis.
From Dishes to Living Tumors
To test whether these effects hold up in a living organism, the team implanted human lung cancer cells into mice to grow small tumors. Daily treatment with pseudolaric acid B significantly slowed tumor growth without causing obvious weight loss or other signs of toxicity. Tumor samples from treated animals carried high levels of a lipid damage marker, increased Survivin, and fewer dividing cells, all consistent with ongoing ferroptosis and reduced tumor vigor. These in vivo findings matched the laboratory experiments and supported the idea that the plant compound can curb tumor growth by driving iron-dependent cell death.
What This Means for Future Treatments
Overall, the study shows that pseudolaric acid B can selectively kill lung cancer cells by pushing them into ferroptosis, a form of cell death fueled by iron and membrane damage, rather than relying mainly on classical apoptosis. This effect depends on a signaling chain involving JNK and ERK proteins and an unexpected rise in the survival protein Survivin, which in this context becomes essential for ferroptosis to proceed. While much work remains before this compound could be tested in patients, these findings suggest that carefully harnessing ferroptosis, possibly with plant-derived molecules, could open new paths to treating stubborn lung cancers.
Citation: Li, Y., Yu, C., Yang, S. et al. Pseudolaric acid B promotes lung cancer cells ferroptosis depending on JNK/ERK-mediated upregulation of survivin. Sci Rep 16, 8294 (2026). https://doi.org/10.1038/s41598-026-36423-3
Keywords: lung cancer, pseudolaric acid B, ferroptosis, Survivin, MAPK signaling