Clear Sky Science · en
Urinary complement C3 fragment levels and their clinical relevance in MPO-ANCA-associated vasculitis
Why pee can reveal hidden inflammation
Doctors treating certain autoimmune diseases of the blood vessels face a major challenge: they need to know when the illness in the kidneys is flaring up, but the clearest tests often require a needle biopsy. This study asks a deceptively simple question with big implications for patients: can an ordinary urine sample show, in real time, how much harmful immune activity is smoldering inside the kidneys of people with a condition called MPO-ANCA–associated vasculitis?
A quiet but serious attack on small blood vessels
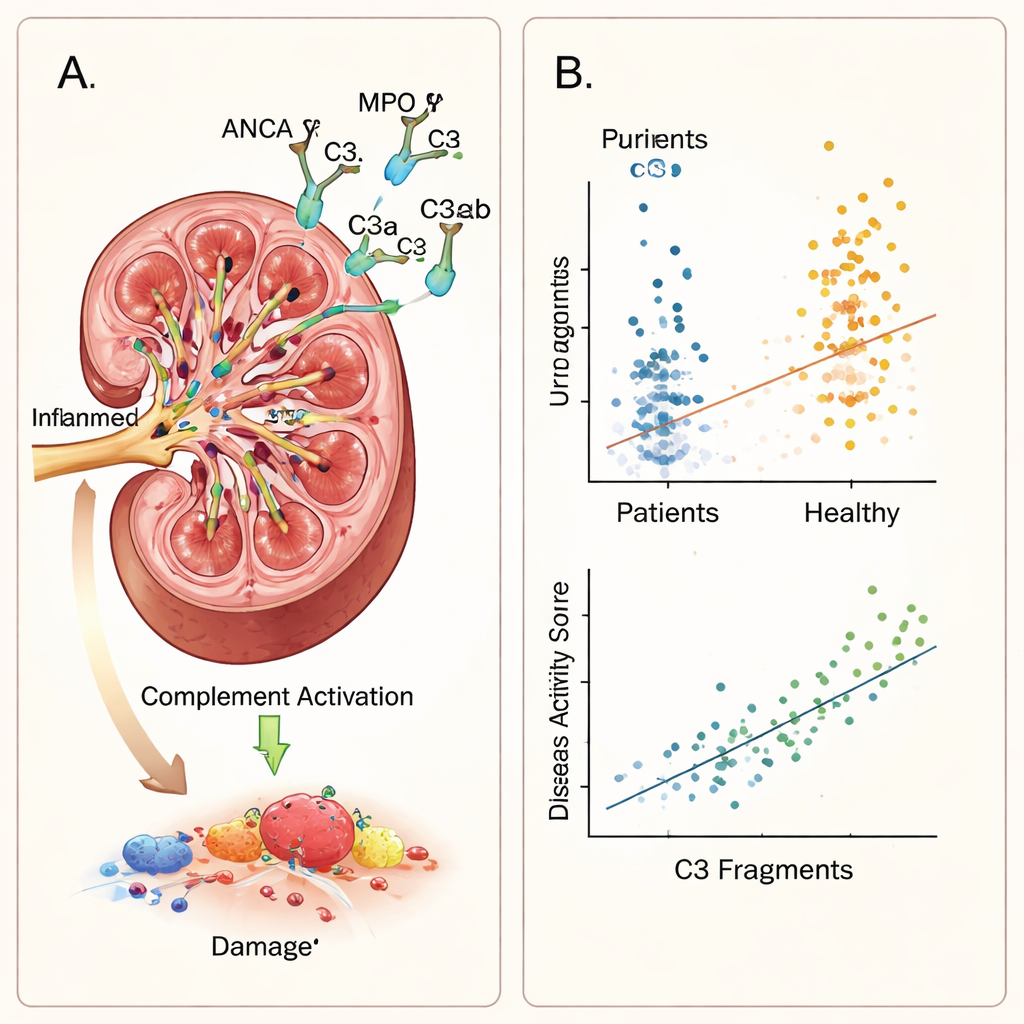
ANCA-associated vasculitis is a rare autoimmune disease in which the body’s own defenses mistakenly attack small blood vessels throughout the body, especially in the kidneys. Many patients develop kidney inflammation that can silently damage these organs until function is badly impaired. Traditionally, this kidney damage has been labeled “pauci-immune,” meaning only a few immune deposits are seen under the microscope. Yet over the past decade, scientists have realized that a powerful immune cascade called the complement system, particularly a branch known as the alternative pathway, plays a crucial role in driving this damage. The question is how to monitor that process without repeatedly sampling kidney tissue.
Following immune footprints in urine
The complement system works a bit like a row of falling dominoes: once triggered, larger components are chopped into smaller fragments that help summon and direct immune cells. One of the central players is a protein called C3. As C3 is cut, it yields a series of fragments—C3a, C3b, iC3b, C3c and C3d—that act as messengers and markers of activity. The researchers reasoned that if the kidneys are a major site of this immune reaction, then some of these C3 fragments should spill into the urine and might serve as a “liquid biopsy” of kidney inflammation. To test this, they collected single urine samples from 22 patients with myeloperoxidase (MPO)–ANCA vasculitis and from 20 healthy people of similar age, then used sensitive laboratory tests to measure the levels of each C3 fragment. They adjusted the results for urine concentration by comparing them to creatinine, a waste product naturally excreted by the kidneys.
Clear differences between patients and healthy people
The contrast between patients and healthy volunteers was striking. Every measured C3 fragment in urine—C3a, C3b, iC3b, C3c and C3d—was significantly higher in people with vasculitis, even after accounting for differences in urine concentration. Within the patient group, higher fragment levels lined up closely with several signs of active disease. They rose together with the Birmingham Vasculitis Activity Score, a standardized clinical scale capturing symptoms and organ involvement, and with two key urine findings: protein in the urine and blood in the urine. In statistical models that considered multiple factors at once, overall disease activity remained the strongest independent driver of fragment levels, and protein leakage from the kidneys was an additional contributor for most fragments. Notably, these immune markers did not track with serum creatinine, a common blood test of kidney filtration, suggesting they are detecting local immune activity rather than just loss of kidney function.
What these findings say about kidney immune activity
The pattern observed in the study supports the idea that complement activation in this disease is happening inside the kidney rather than simply being filtered in from the bloodstream. Kidney cells are known to be capable of producing complement proteins, and when the small vessels are inflamed, more C3 may be cut into active pieces right at the site of injury. Those fragments can then appear in the urine, offering a window into the ongoing immune attack. One fragment, iC3b, was especially interesting: it closely reflected overall disease activity but did not rise and fall with the amount of protein in the urine, hinting that it may be more tightly linked to immune signaling itself than to structural leakage in the filtering barrier.
From needle biopsies to “liquid biopsies”?
The study has important caveats: it was conducted at a single hospital, included only 22 patients, and captured each person at just one point in time. Not all participants had kidney biopsies, limiting detailed comparisons with tissue findings. Still, the work provides compelling early evidence that a simple urine test for complement C3 fragments can reflect how active MPO-ANCA vasculitis is in the kidneys, independent of traditional kidney function tests. For patients and clinicians, this raises the possibility that in the future, carefully designed urine tests could help track disease flares, guide treatment intensity, and perhaps evaluate new drugs that target the complement system—with far less reliance on invasive procedures.
Citation: Hu, Y., Shi, M., Huang, Q. et al. Urinary complement C3 fragment levels and their clinical relevance in MPO-ANCA-associated vasculitis. Sci Rep 16, 5643 (2026). https://doi.org/10.1038/s41598-026-36417-1
Keywords: vasculitis, autoimmune kidney disease, complement system, urine biomarkers, ANCA