Clear Sky Science · en
Experimental petroleum resin production and optimization using response surface modeling
Why this sticky story matters
From road-marking paint and roofing materials to packaging tape and printing inks, many everyday products rely on petroleum resins—the invisible "glue helpers" that add tack, shine, and durability. Making these resins is energy‑intensive and often wasteful, and small changes in how they are produced can greatly affect their color, strength, and cost. This study shows how a careful mix of lab experiments and smart statistics can tune the resin‑making process so manufacturers get more useful product with fewer trade‑offs.
Turning leftover fuel into useful glue helpers
Petroleum resins are made from complex hydrocarbon mixtures that emerge as by‑products when oil is cracked in refineries. The authors use a stream called pyrolysis gasoline, which contains a blend of ring‑shaped (aromatic) and chain‑like (aliphatic) molecules. This feedstock is attractive because it can yield resins that work well in many formulations, but its complexity also makes the chemistry hard to control. In this work, the team focuses on how three simple knobs—reaction temperature, the amount of aluminum chloride catalyst, and reaction time—shape four key outcomes: how much resin is produced, how heavy the molecules are, how high the softening point is (a measure of heat resistance), and how light or dark the color appears. 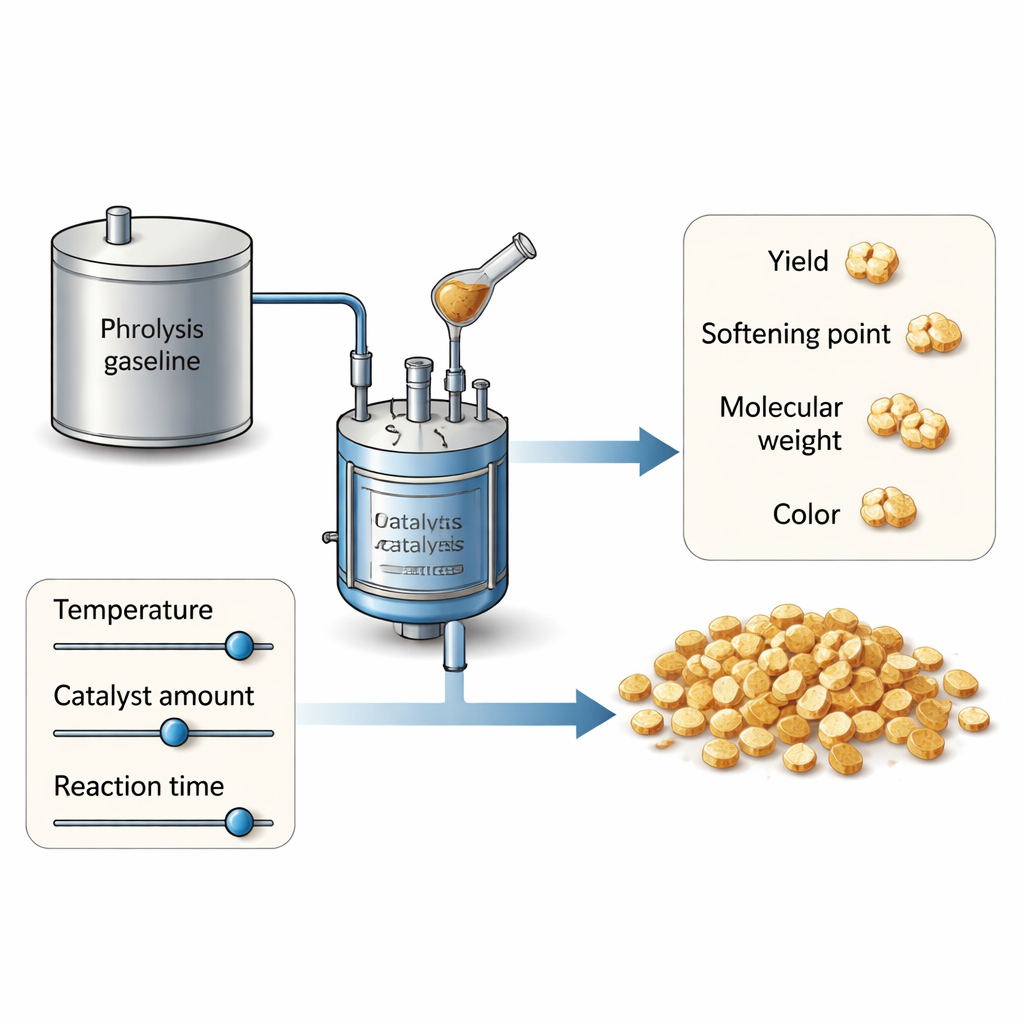
Designing the experiment like a map, not a guessing game
Instead of changing one setting at a time and hoping for the best, the researchers use a structured planning method called response surface methodology. They perform 20 carefully arranged experiments that cover a wide range of temperatures (20–100 °C), catalyst doses (0.1–3 percent by weight), and reaction times (1–3 hours). This design lets them see not only how each knob matters on its own, but also how the knobs interact. Statistical models are then fitted to the data to predict resin yield, molecular weight, softening point, and color anywhere within the tested range. The models prove remarkably accurate: they explain more than 94 percent of the variation in every property, and checks against new experiments show prediction errors of only a few percent.
Peeking inside the resin
To understand what kind of material they are actually making, the team probes the resins with several standard laboratory techniques. Infrared and nuclear magnetic resonance measurements reveal that the products are hybrid materials containing both aromatic and aliphatic segments, in proportions consistent with the starting feed. Calorimetry shows a glass transition temperature around 70 °C and no melting point, confirming that the resins are amorphous, rubber‑like solids rather than crystalline plastics. These traits are typical of commercial petroleum resins used as tackifiers in adhesives and coatings. Visually, the samples range from dark brown chunks to lighter tan pieces, mirroring the measured color values and highlighting how processing choices translate into appearance and usability. 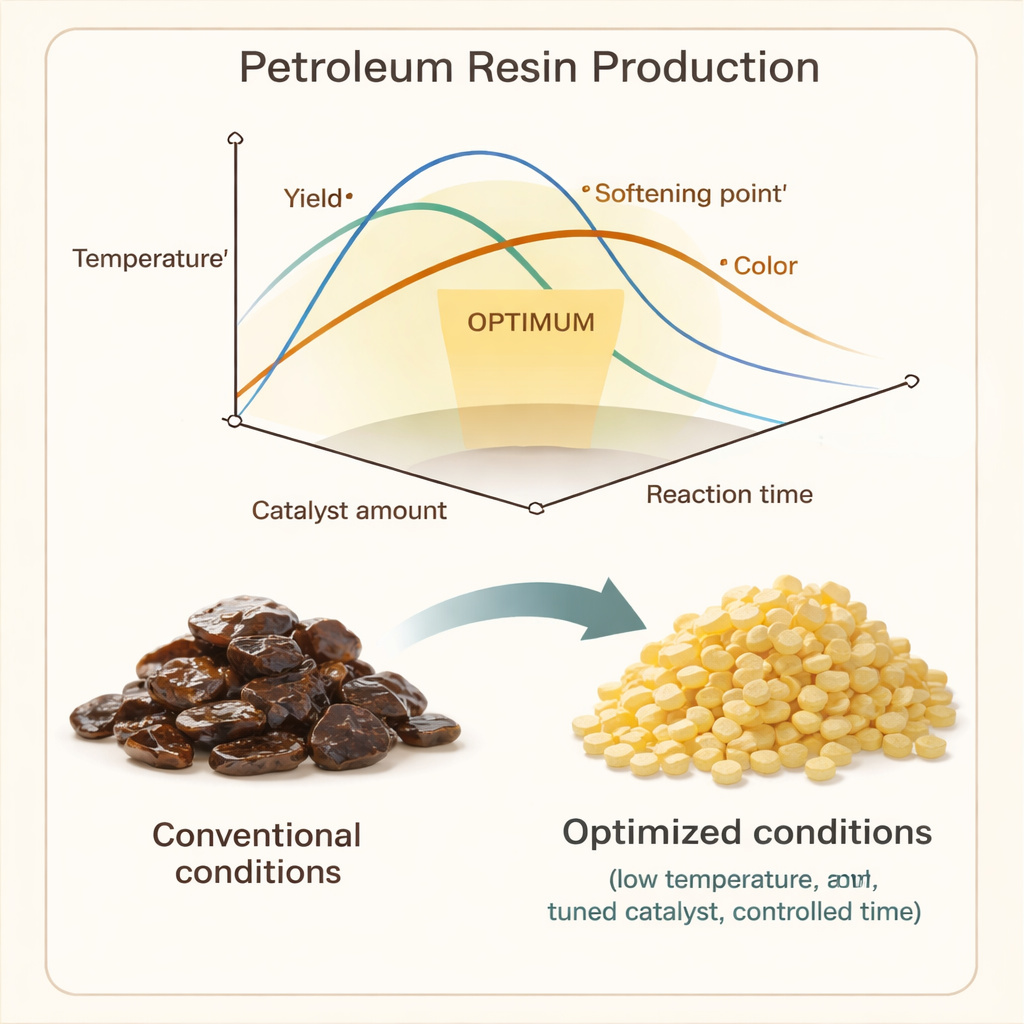
Finding the sweet spot in a three‑way trade‑off
The heart of the study is figuring out how to balance competing goals. Higher temperatures tend to speed up side reactions that cut chains short, reducing yield, lowering softening point, but helping produce a paler color. More catalyst first improves the reaction but, beyond a point, encourages unwanted branching and breakdown. Longer times steadily boost yield but do little for other properties once most reactive molecules have been consumed. By combining all these tendencies in a multi‑objective search, the authors locate an operating window that simultaneously gives high yield, high softening point, and acceptably light color while keeping molecular weight within a practical range.
What better resin looks like in practice
The optimized recipe they uncover is surprisingly mild: a low temperature of 20 °C, a moderate catalyst loading of about 1.13 percent, and a reaction time of 86 minutes. Under these conditions, the process yields about 22.5 percent resin with a molecular weight a little over 2000 g/mol, a high softening point of 152 °C, and a relatively light Gardner color of 3.7. Independent experiments confirm these predictions to within a few percent, demonstrating that the statistical model can reliably guide real‑world production decisions. Compared with earlier studies, this recipe sacrifices some raw yield but offers noticeably better heat resistance and color, which can be more valuable in many applications.
A roadmap for smarter chemical manufacturing
For a non‑specialist, the main message is that even messy, industrial mixtures can be tamed using carefully planned experiments and data‑driven models. By treating the production line as something to be mapped and optimized rather than tweaked by trial and error, the researchers show how to turn refinery by‑products into high‑value resins with predictable quality. Their framework—combining designed experiments, statistical modeling, and chemical insight—can be applied to other chemical processes where industry must juggle yield, performance, and appearance all at once.
Citation: Rostami, MT., Shahverdi, H., Javanbakht, V. et al. Experimental petroleum resin production and optimization using response surface modeling. Sci Rep 16, 6481 (2026). https://doi.org/10.1038/s41598-026-36409-1
Keywords: petroleum resin, polymerization, process optimization, industrial chemistry, statistical modeling