Clear Sky Science · en
A density functional theory study of cyclophosphamide and purinethol adsorption on a covalent triazine framework (CTF-2) for drug delivery applications
Why Smarter Chemotherapy Matters
Chemotherapy drugs save lives, but they often act like a floodlight instead of a spotlight—attacking healthy cells along with cancerous ones. This can lead to serious side effects, from fatigue and infections to organ damage. The study discussed here explores an ultra-thin, highly porous material called a covalent triazine framework (CTF-2) as a “smart sponge” that can carry two long‑used anticancer drugs—cyclophosphamide and purinethol—more safely and efficiently to where they are needed, and then let them go under tumor‑like conditions.
A New Kind of Molecular Sponge
CTF-2 is a man‑made material built from rings of carbon and nitrogen linked into a flat, sheet‑like network with a large central cavity and plenty of open space. Because it is thin, stable, and full of pores, it offers a huge internal surface area for trapping drug molecules. The authors focused on two important cancer medicines: cyclophosphamide, widely used for blood cancers and solid tumors, and purinethol (also called mercaptopurine), used against leukemias and some inflammatory bowel diseases. The idea is to dock these drugs gently onto the CTF-2 surface so they can be carried through the bloodstream and released more selectively, reducing damage to healthy tissue.
Probing the Invisible with Computer Experiments
Instead of working in a wet lab, the team used advanced quantum‑level computer calculations (density functional theory and related methods) to ask how strongly each drug would cling to CTF-2 and what holds the complexes together. They optimized the shapes of CTF-2 plus each drug, measured the distances between atoms where they touch, and computed how much energy is gained when a drug sticks to the framework. For cyclophosphamide, the calculated adsorption energy was about −1.04 electron volts, a bit stronger than the −0.82 electron volts found for purinethol. These values, which remain clearly favorable even after correcting for technical artifacts and simulating water as a solvent, indicate that both drugs bind firmly enough to be loaded but not so strongly that they cannot later be released.
Gentle Forces Doing Important Work
The study shows that the “glue” between drugs and CTF-2 is not a hard chemical bond but a web of gentle, non‑covalent forces. Using specialized analyses of electron density—essentially maps of where the shared electrons reside—the researchers found that van der Waals attractions (the same weak forces that let geckos climb walls) and mild electrostatic interactions dominate. These interactions occur at short but non‑bonding distances between hydrogen, nitrogen, oxygen, sulfur, and carbon atoms on the drugs and on the framework. Additional calculations that split the total attraction into different components confirmed that electrostatic and dispersion (van der Waals) forces provide most of the stabilizing pull, while repulsive forces prevent the drug from sinking too deeply or binding irreversibly.
Signals of Stable Loading and Tunable Release
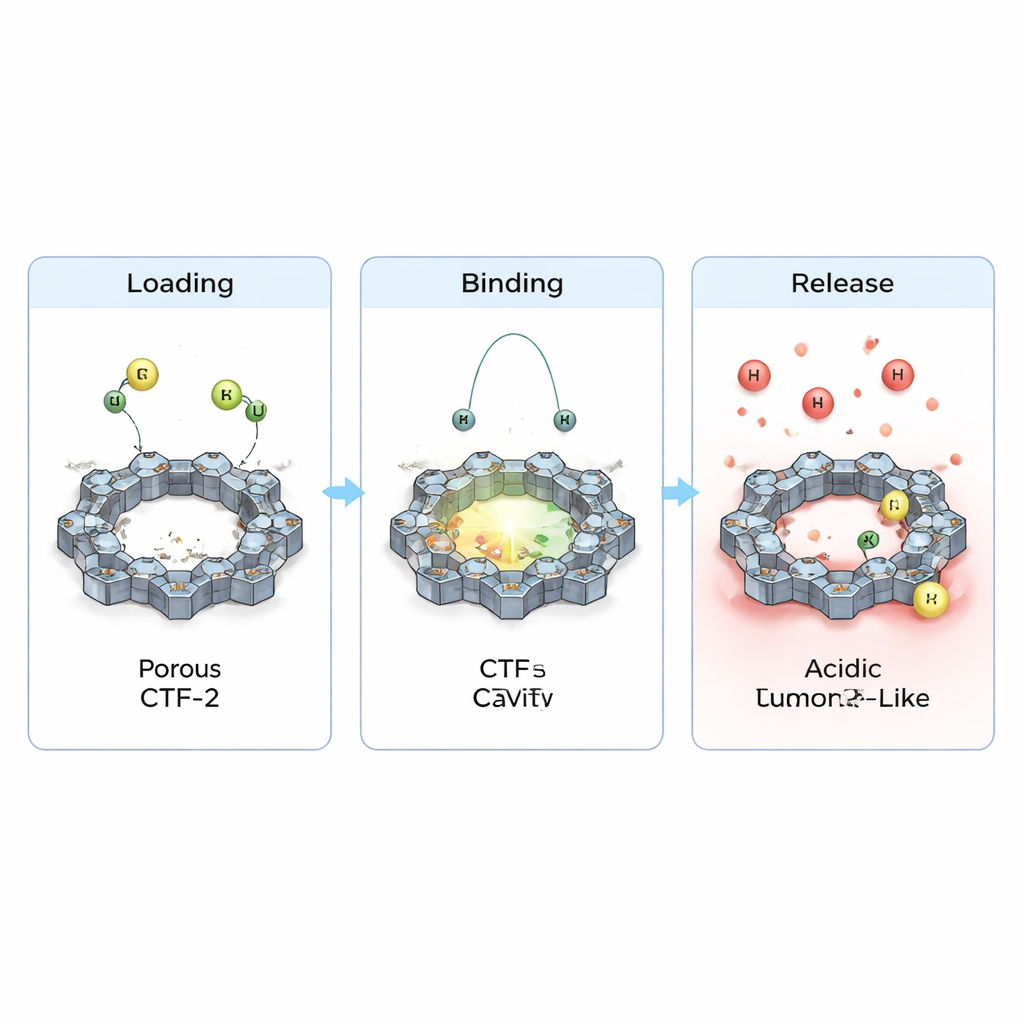
The team also examined how electrons shift when the drugs sit on CTF-2, and how this changes basic electrical properties. They observed small but clear transfers of charge between each drug and the framework and a slight narrowing of the energy gap that separates filled from empty electronic states. This implies that the material’s reactivity and conductivity are subtly tuned upon loading, a useful feature for any future sensing or theranostic applications. Crucially, when they mimicked the more acidic environment found near tumors by adding extra protons, the calculated binding weakened and the contact distances increased. In simple terms, the simulations suggest that CTF-2 can hold onto the drugs under normal blood‑like conditions, but will tend to let them go more readily in tumor‑like, acidic surroundings—exactly the behavior desired for targeted delivery.
What This Means for Future Cancer Treatments
Put together, these calculations paint CTF-2 as a promising carrier for cyclophosphamide and purinethol: it can pack them onto its porous surface in a stable way, protect them during circulation, and then release them when the local chemistry changes around a tumor. Although the work is theoretical and will need experimental confirmation, it suggests that finely designed, nitrogen‑rich frameworks like CTF-2 could help transform old but effective chemotherapy drugs into smarter medicines—delivering potent doses where they are needed most while easing the collateral damage to healthy cells.
Citation: Tariq, T., Yar, M., Bayach, I. et al. A density functional theory study of cyclophosphamide and purinethol adsorption on a covalent triazine framework (CTF-2) for drug delivery applications. Sci Rep 16, 6125 (2026). https://doi.org/10.1038/s41598-026-36405-5
Keywords: nanocarrier, drug delivery, cancer chemotherapy, covalent triazine framework, computational modeling