Clear Sky Science · en
Staphylococcus capitis strain producing dual bacteriocins, capidermicin and micrococcin P1, shows broad-spectrum antimicrobial activity
Why tiny skin bacteria matter for superbug threats
Antibiotic-resistant infections are rising worldwide, and doctors are running out of reliable drugs. One promising idea is to recruit our own “good” microbes to help fight the bad ones. This study focuses on a common skin bacterium, Staphylococcus capitis, and shows that a particular strain, called HBC3, naturally makes two powerful germ-killing molecules. Together, these molecules can knock down dangerous hospital pathogens, including methicillin-resistant Staphylococcus aureus (MRSA), suggesting that friendly skin bacteria themselves could become future infection-fighting tools.
A skin resident with hidden firepower
S. capitis normally lives quietly on our skin, especially on the scalp, and is usually harmless. The researchers screened 18 S. capitis strains collected from people’s noses to see whether any could inhibit MRSA, a major cause of hard-to-treat infections. One strain, HBC3, stood out. In lab tests, it strongly suppressed a wide variety of Gram-positive bacteria, including MRSA, vancomycin-resistant enterococci (VRE), Streptococcus pyogenes, Listeria monocytogenes, and Bacillus and Clostridium species that can cause serious disease. Notably, it showed no effect on several common Gram-negative bacteria such as Escherichia coli and Klebsiella pneumoniae, suggesting a focused but powerful action rather than indiscriminate killing. 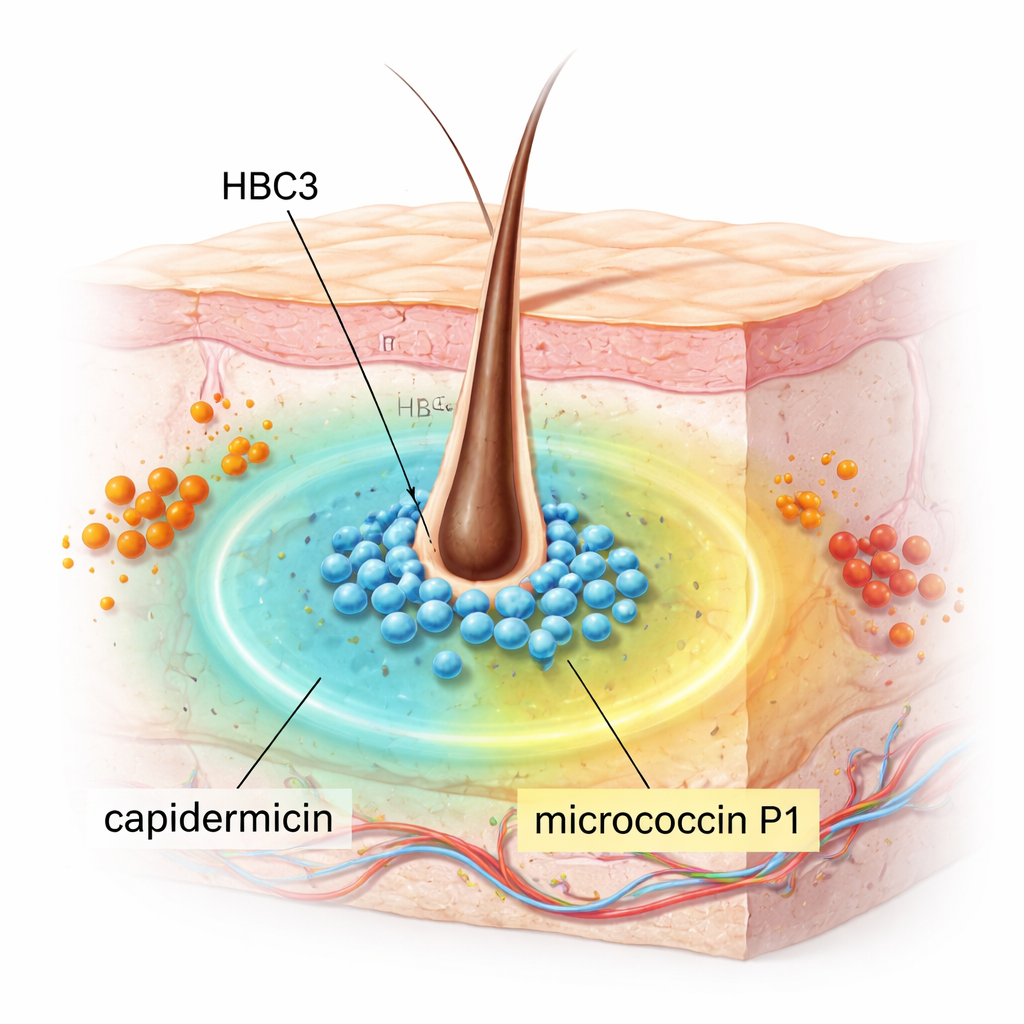
Two natural antibiotics on a single genetic “mini-chromosome”
To discover how HBC3 exerts this broad antimicrobial punch, the team decoded its entire genome. They identified a small circular DNA element, a plasmid named pHBC3_1, that carries the complete instructions for making two different bacteriocins—natural antibiotic-like peptides produced by bacteria. One is capidermicin, a small positively charged peptide previously seen in other S. capitis strains. The other is micrococcin P1 (MP1), a thiopeptide that blocks protein production in target bacteria and had not been reported before in S. capitis. When this plasmid was removed from HBC3, the strain completely lost its antibacterial activity, confirming that these genes are essential for its defensive abilities.
Different targets, shared goal
By purifying each bacteriocin separately, the scientists tested how well they inhibited various microbes. MP1 showed broad and strong activity against many Gram-positive cocci, including MRSA and enterococci, at low concentrations. Capidermicin, in contrast, was most effective against rod-shaped Gram-positive bacteria such as Bacillus coagulans and Listeria, but had weak or no effect on staphylococci in the conditions used here. When the team combined both peptides against B. coagulans, they observed an additive effect: lower doses together achieved stronger growth inhibition than either alone. This complementary targeting means HBC3 can suppress a wider range of rivals by deploying two different molecular weapons tuned to different bacterial shapes and cell surfaces. 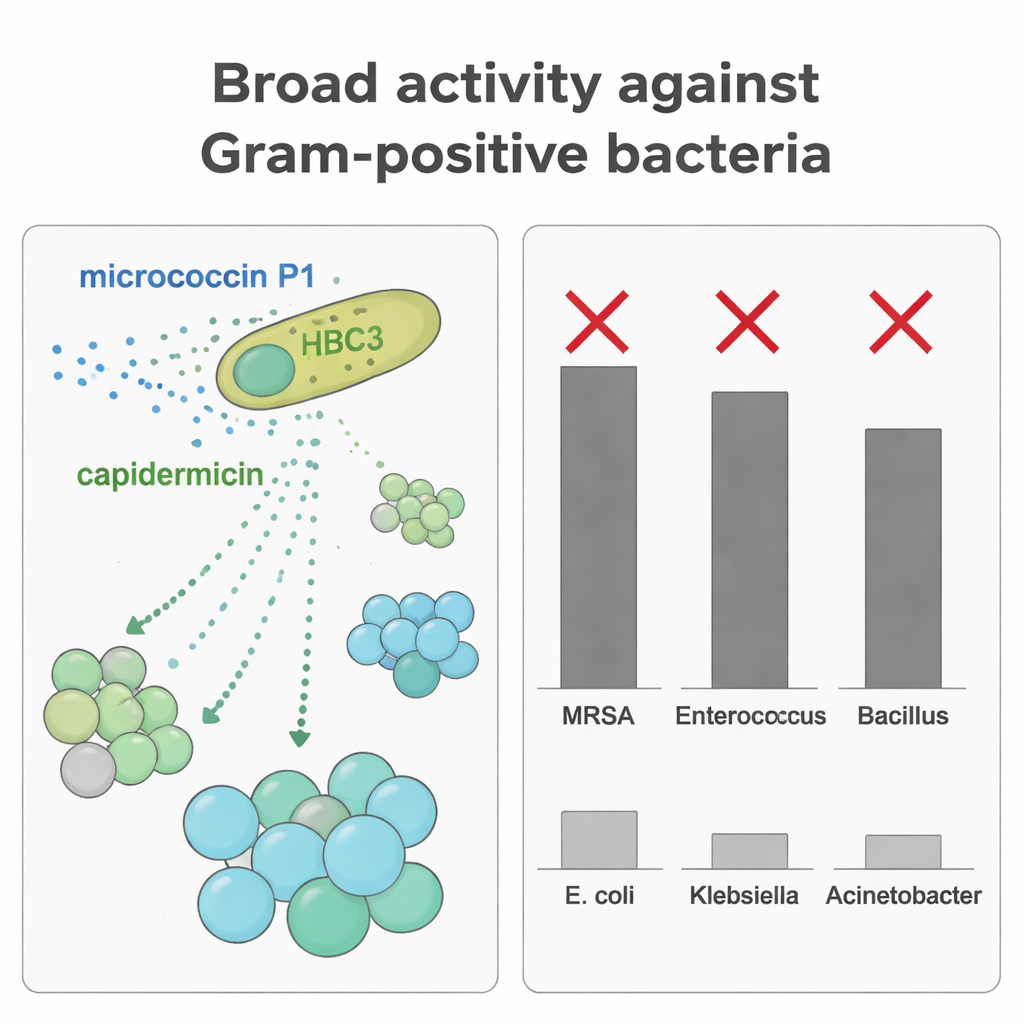
Winning space in crowded microbial neighborhoods
To mimic real-life competition, the researchers co-cultured HBC3 with MRSA, VRE, and B. coagulans on the same plates. The normal HBC3 strain almost completely eliminated these pathogens from the mixed communities, while a version lacking the plasmid allowed them to thrive. Genetic comparisons suggest that the dual-bacteriocin plasmid likely arose from past fusion and reshuffling of separate plasmids, helped by mobile DNA elements that can move gene clusters between bacteria. This composite plasmid gives HBC3 a clear edge in crowded environments like skin or mucous membranes, where many microbes compete for nutrients and space.
From skin ally to future probiotic?
The study concludes that S. capitis HBC3 is equipped with a rare combination of two distinct bacteriocins on one plasmid, enabling it to strongly inhibit multiple clinically important Gram-positive pathogens, including antibiotic-resistant strains. Because HBC3 itself is not part of a known high-risk lineage and does not carry detectable drug resistance genes, it may be a candidate for development as a targeted probiotic or decolonization agent—designed, for example, to selectively knock down S. aureus on skin or in the nose while sparing more benign neighbors. Before such applications become reality, safety, effectiveness in animals and humans, and long-term impacts on the normal microbiota will need careful testing, but the work highlights how our own microbes may help counter the ongoing superbug crisis.
Citation: Ohdan, K., Suzuki, Y., Kawada-Matsuo, M. et al. Staphylococcus capitis strain producing dual bacteriocins, capidermicin and micrococcin P1, shows broad-spectrum antimicrobial activity. Sci Rep 16, 6835 (2026). https://doi.org/10.1038/s41598-026-36393-6
Keywords: bacteriocins, Staphylococcus capitis, micrococcin P1, capidermicin, antibiotic resistance