Clear Sky Science · en
Bis-1,2,4-triazole derivatives as potential antioxidants for pneumonia therapy
Why protecting lungs from "rust" matters
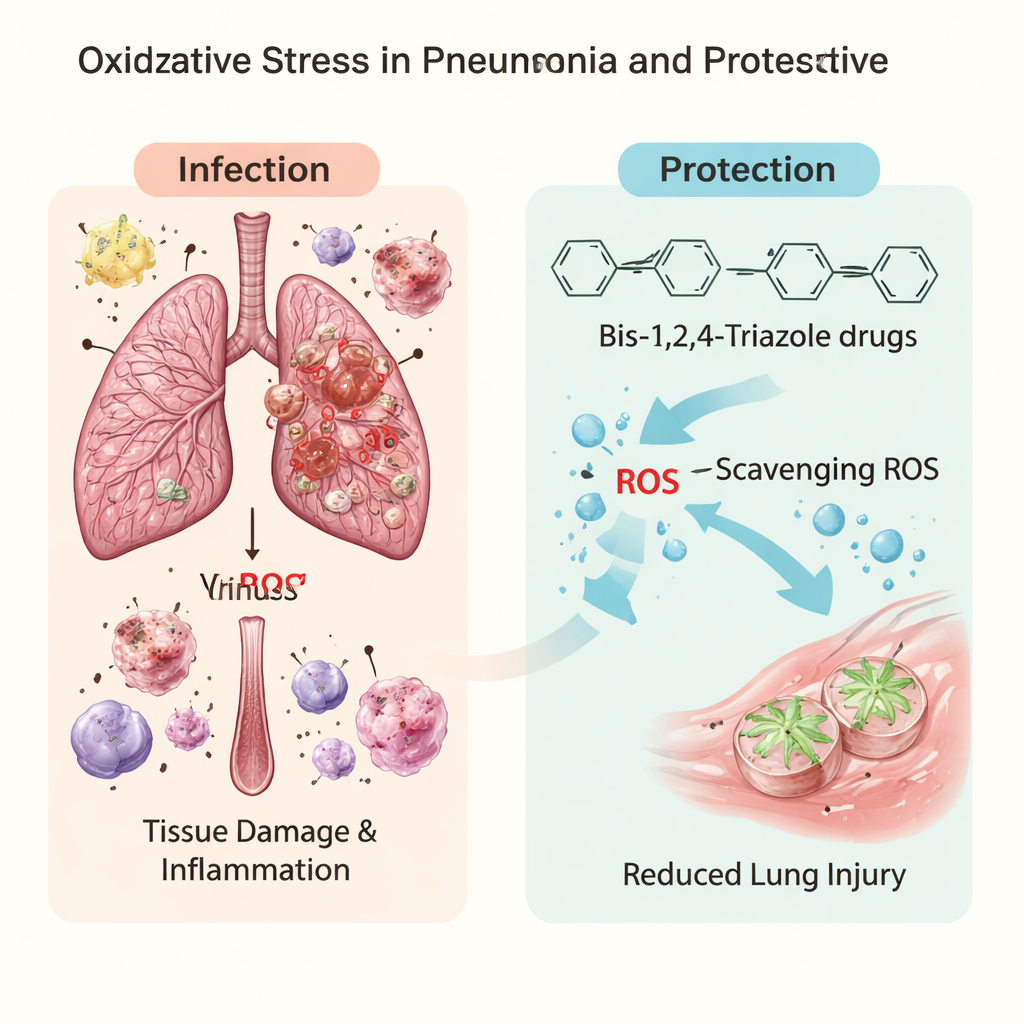
Pneumonia is often thought of simply as a bad lung infection that antibiotics can fix. But many people with severe pneumonia die not just from germs, but from the body’s own overreaction. As immune cells fight invading bacteria or viruses, they release highly reactive molecules—chemical "sparks" that can damage lung tissue much like rust eats away at metal. This study explores a new family of lab‑made molecules, called bis‑1,2,4‑triazoles, designed to act as both powerful antioxidants and blockers of bacterial weaponry. The goal is to develop add‑on medicines that protect the lungs while antibiotics clear the infection.
Hidden damage during a lung infection
When microbes invade the lungs, immune cells unleash bursts of reactive oxygen and nitrogen species, or ROS/RNS. In small amounts these help kill pathogens; in excess they attack the body’s own fats, proteins, and DNA. Recent clinical work shows that people with severe pneumonia, including COVID‑19 pneumonia, have clear signs of oxidative stress and redox imbalance. This chemical overload weakens the lung’s delicate air sacs, fuels runaway inflammation, and contributes to acute respiratory distress syndrome. That has led researchers to ask whether targeted antioxidants could act like firebreaks—soaking up these reactive molecules before they can burn through lung tissue.
Designing new small molecules to shield the lungs
The team focused on a ring‑shaped chemical structure called a 1,2,4‑triazole, already common in many drugs. Instead of using a single ring, they linked two rings together to form bis‑1,2,4‑triazoles, which can interact with both water‑loving and fat‑loving parts of cells and with metal ions. Six such compounds had previously been made and shown to have antimicrobial effects and to inhibit an enzyme linked to inflammation and cancer. In this work, the authors asked whether adjusting features like the length of a carbon chain or adding a nitro group on an attached ring could make these molecules especially good at neutralizing ROS and interfering with bacterial virulence factors involved in pneumonia.
Testing antioxidant power in the lab
To gauge how well the six molecules could mop up free radicals, the researchers used a standard color‑change test with a stable radical called DPPH. As an antioxidant removes this radical, the solution fades from purple to yellow, and the degree of fading can be precisely measured. All six bis‑triazoles showed dose‑dependent radical‑scavenging activity, but two stood out. A compound bearing a flexible six‑carbon (hexyl) chain and another carrying a para‑nitrophenyl ring came closest to matching vitamin C, a classic antioxidant. They needed roughly twice the concentration of vitamin C to reach the same 50% radical‑blocking mark, yet still performed strongly enough to be considered promising drug leads. Their structures appear to help them nestle into fatty lung membranes and stabilize the radicals they catch.
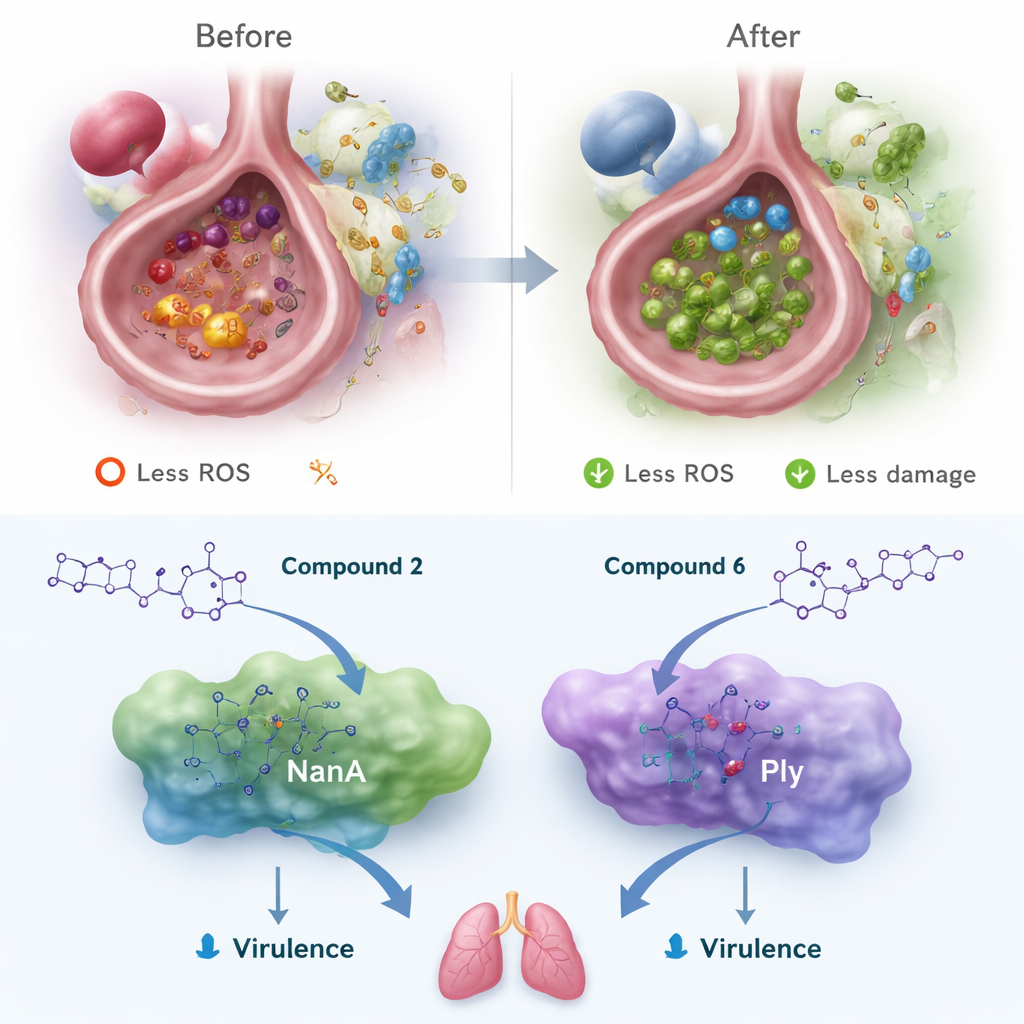
Hitting bacterial weapons while staying drug‑like
Beyond antioxidants, the team used computer simulations to see whether the new molecules could jam two key proteins from Streptococcus pneumoniae: NanA, which helps bacteria latch onto and invade tissues, and pneumolysin (Ply), a toxin that punches holes in cell membranes. The standout nitrophenyl compound bound most tightly in silico, forming multiple hydrogen bonds and stacking interactions inside the proteins’ active pockets. Although not as sticky as some bulky natural polyphenols, these bis‑triazoles showed that their compact scaffold can occupy the same vulnerable regions of NanA and Ply. Parallel computer models of absorption, metabolism, and toxicity suggested that all six compounds, especially the two leaders, have high predicted gut absorption, low toxicity, no major interference with common drug‑metabolizing enzymes, and no tendency to disturb heart rhythm channels—key features of a "drug‑like" profile.
What this could mean for future pneumonia treatment
Taken together, the results point to bis‑1,2,4‑triazoles—particularly the two best‑performing molecules—as early prototypes of dual‑action helpers for pneumonia therapy. In principle, such compounds could both soak up harmful reactive molecules that erode lung tissue and blunt bacterial tools that worsen disease, all while being safe enough for oral use and largely staying out of the brain. The work is still at a preclinical stage: the compounds must now be tested for safety in human lung cells and in animal models of pneumonia, and their structures will likely be refined to boost potency and solubility. But the study offers a clear message for non‑specialists: future pneumonia treatments may not rely on antibiotics alone; they may also include small molecules that protect our lungs from the collateral damage of our own immune response.
Citation: Korol, N., Symkanych, O., Pallah, O. et al. Bis-1,2,4-triazole derivatives as potential antioxidants for pneumonia therapy. Sci Rep 16, 5640 (2026). https://doi.org/10.1038/s41598-026-36386-5
Keywords: pneumonia, oxidative stress, antioxidants, bacterial virulence, drug design