Clear Sky Science · en
MGMT upregulation mediates Temozolomide resistance conferred USP5 dependency
Why some brain tumors stop responding to a key drug
For people with the aggressive brain cancer glioblastoma, one of the main drugs doctors rely on is temozolomide. At first, this chemotherapy can slow the disease, but tumors often learn how to shrug it off. This study asks a practical question with life‑or‑death consequences: what exactly allows these cancer cells to escape the drug, and can we find new weak points to attack so the treatment starts working again?
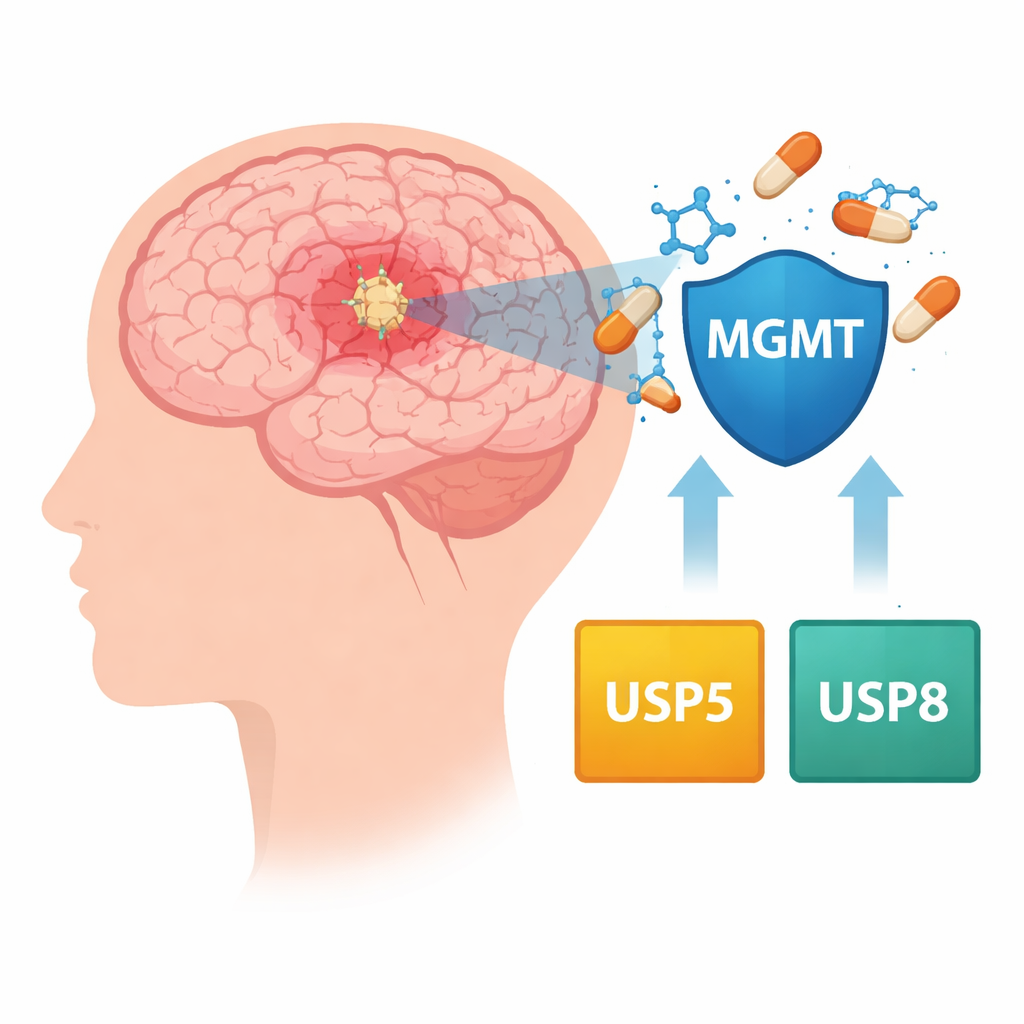
A drug that helps at first, then fades
Temozolomide works by damaging the DNA of rapidly dividing tumor cells, forcing them to die. Many glioblastomas, however, produce a repair protein called MGMT that can undo this damage. Patients whose tumors switch on MGMT tend to do worse because the drug’s effect is cancelled out. Doctors can partly predict response by checking whether the MGMT gene is chemically switched off (methylated) or on (unmethylated), but this does not fully explain why resistance develops or how MGMT levels stay high inside tumor cells over time.
Hidden helpers that protect the repair protein
The authors looked at another layer of control: the molecular “cleanup crews” that decide which proteins get destroyed and which are spared. Cells normally tag worn‑out proteins for breakdown, while a group of enzymes called deubiquitinating enzymes (DUBs) can rescue proteins from this fate. By mining large public cancer databases and examining drug‑resistant glioblastoma cell lines grown in the lab, the team homed in on two such enzymes, USP5 and USP8. In tumor samples from hundreds of patients, higher USP5 levels tended to travel together with higher MGMT levels, and patients whose tumors had both genes turned up high generally died sooner.
Building resistance in the lab
To mimic what happens in patients, the researchers slowly exposed two common glioblastoma cell lines to rising doses of temozolomide until the cells became resistant. In these hardened cells, several DUBs rose, but USP5, USP8, USP10 and the repair protein MGMT were especially elevated. When the team used small RNA molecules to silence USP5 or USP8, the resistant cells suddenly became vulnerable: they activated self‑destruct programs, including apoptosis and a form of stress‑driven self‑digestion known as autophagy. At the same time, MGMT levels dropped sharply, and another resistance‑linked protein, USP10, was also reduced, suggesting these enzymes sit near the top of a survival network.
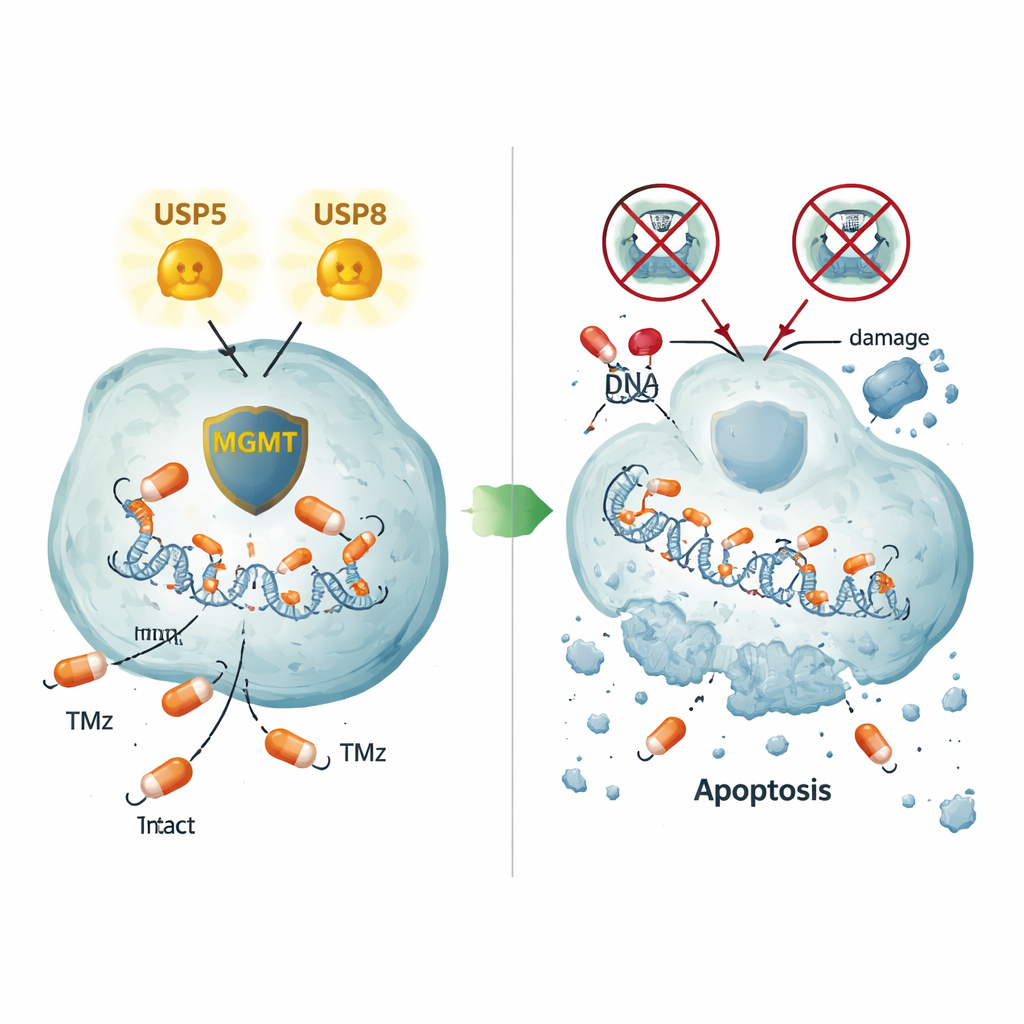
Two different ways to keep MGMT alive
Careful imaging of tumor cells and human tumor tissue showed USP5 and MGMT often sitting together in the same regions of the cell, especially in the cytoplasm. Blocking USP5 not only lowered MGMT but also caused MGMT to be broken down by the cell’s protein‑shredding machinery, a process that could be reversed with the drug bortezomib, which blocks the proteasome. Overproducing USP5 in otherwise MGMT‑negative cells was enough to raise MGMT protein levels, even in a non‑brain cell line, confirming a direct stabilizing effect. USP8 turned out to act differently: knocking it down also reduced MGMT, but without changing USP5 itself, revealing a second, USP5‑independent route that helps keep MGMT around. Even in a resistant cell line that did not make MGMT at all, turning off USP5 or USP8 still triggered extensive cell death, pointing to additional resistance pathways controlled by these enzymes.
Links to the immune system and new treatment ideas
Beyond drug resistance, the study connects USP5 to how glioblastoma interacts with the immune system. Analysis of genomic data suggested that changes in USP5 levels and gene copy number shift the balance of various immune cells around the tumor, including killer T cells and regulatory T cells. Earlier work has also shown that USP5 can stabilize PD‑L1, a protein tumors use to switch off attacking T cells. Taken together, the findings suggest that USP5 may help tumors both survive chemotherapy and evade immune attack, making it a particularly attractive drug target.
What this means for future brain cancer therapy
In plain terms, this work identifies USP5 and USP8 as key bodyguards that protect the MGMT repair protein and help glioblastoma cells resist temozolomide. By dismantling these bodyguards, the authors were able to kill drug‑resistant cells in the lab and lower MGMT levels, even in some cases where MGMT‑based resistance was not the only problem. That raises the hope that future medicines aimed at USP5 or USP8, possibly combined with existing chemotherapy and immune‑based drugs, could make stubborn brain tumors sensitive again and give patients more time.
Citation: Bhardwaj, S., Sanjay, Sharma, D. et al. MGMT upregulation mediates Temozolomide resistance conferred USP5 dependency. Sci Rep 16, 6118 (2026). https://doi.org/10.1038/s41598-026-36379-4
Keywords: glioblastoma, temozolomide resistance, MGMT, USP5, deubiquitinating enzymes