Clear Sky Science · en
Novel carbonate, oxide, and hydroxide nanohybrids based on Mg, Ba, and Ca for efficient Safranin O dye adsorption
Why cleaning up bright dyes in water matters
Brilliant red dyes may look harmless in a test tube, but in rivers and lakes they can block sunlight, disrupt food webs, and carry toxic effects up the food chain. One such colorant, Safranin O, is widely used in laboratories and industry and is notoriously hard to remove once it reaches water. This study explores a new class of tiny mineral hybrids—built from common elements like magnesium, calcium, and barium—that can pull Safranin O out of water with remarkable efficiency and be reused many times, offering a practical route toward cleaner, safer wastewater.
Color in, color out: a simple idea with tiny particles
The researchers set out to design a solid material that could act like a sponge for Safranin O without dissolving or losing strength in real wastewater. They used a versatile preparation route called a Pechini sol–gel method to mix metal salts with an organic resin at the molecular level, then heated the mixture to either 600 or 800 °C. The resulting products—named BMC600 and BMC800—are multiphase nanocomposites, meaning they contain several different mineral components in one particle. These include magnesium oxide (MgO), calcium carbonate (CaCO3), barium carbonate (BaCO3), and calcium hydroxide (Ca(OH)2). Each of these brings slightly different chemical “personalities,” and together they create many active spots where dye molecules can latch on.
Peering inside the dye sponge
To understand what they had made, the team used a suite of modern characterization tools. X-ray diffraction confirmed that both BMC600 and BMC800 contained the same four crystalline phases, with crystal domains on the order of 60–70 nanometers. Electron microscopy revealed that the lower-temperature sample, BMC600, consisted of smaller, more finely divided particles than BMC800. High‑resolution images showed quasi‑spherical nanoparticles averaging about 29 nanometers in BMC600 but roughly six times larger in BMC800. Because adsorption happens at surfaces, these smaller, less-sintered particles in BMC600 expose more reactive area and defects for the dye to bind, a structural advantage that later shows up in performance tests.
How the dye sticks and how well it works
When the new materials were mixed with Safranin O solutions, several trends emerged. At very acidic conditions (pH 2), both materials removed only a small fraction of the dye, but at mildly alkaline pH 10 their performance jumped dramatically: BMC600 removed about 82% and BMC800 about 68% under standard test conditions. This switch is linked to surface charge. Below a certain pH point, the particles’ surfaces are positively charged and repel the positively charged Safranin O molecules. Above that point, the surfaces become negatively charged and electrostatically attract the dye. Infrared spectroscopy confirmed that surface hydroxyl groups and carbonate groups also participate, forming hydrogen bonds and other weak interactions with the dye. Together, these forces create strong yet reversible binding. When the team varied contact time and concentration, they found that BMC600 worked faster and had a higher maximum capacity than BMC800, holding up to about 318 milligrams of dye per gram of adsorbent, compared with 270 milligrams per gram for BMC800. The data fit a simple “monolayer” adsorption picture, where dye molecules arrange themselves in a single layer over the most favorable sites.
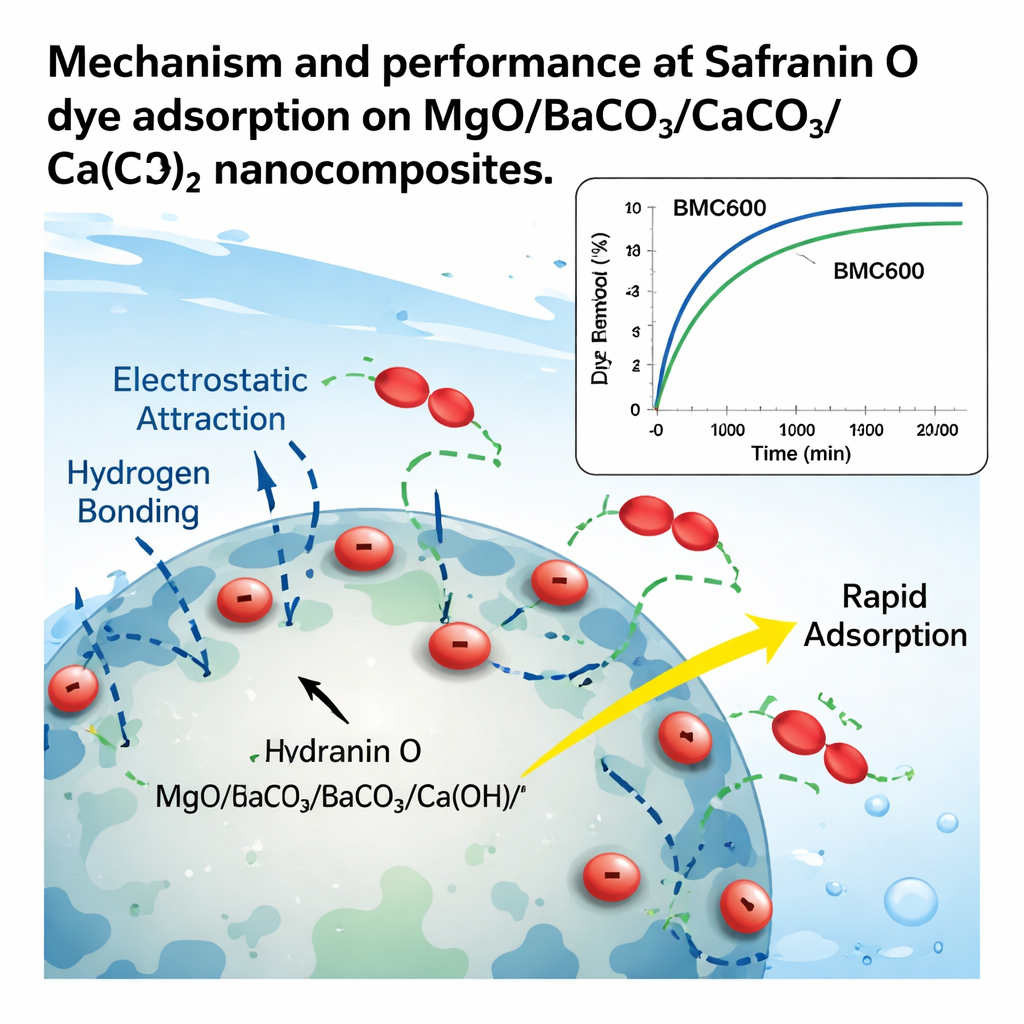
Energy, competition, and reuse in real-world conditions
Temperature and competing substances can make or break a water treatment material. Here, raising the temperature reduced how much Safranin O was captured, indicating an exothermic, or heat‑releasing, physical adsorption process: the dye prefers to stay attached at cooler temperatures and is slightly less favored when warmed. Despite that, the overall process remained spontaneous over the tested range, and thermodynamic analysis suggested that the main interactions are relatively gentle, not permanent chemical bonds—good news for regeneration. The nanocomposites also held up well when other common ions and dyes were present; ordinary salts caused only modest drops in capacity, although other positively charged dyes competed strongly. Crucially, the adsorbents could be cleaned and reused: washing with hydrochloric acid released up to about 99.7% of the bound Safranin O, and after five adsorption–desorption cycles BMC600 still retained roughly 88% of its initial performance. A back‑of‑the‑envelope cost analysis suggested that, thanks to their high capacity, these materials could remove dye at a cost that compares favorably with many existing options.
What this means for cleaner water
In everyday terms, these nanocomposites behave like robust, reusable mineral sponges tailored for a stubborn red dye. By combining several simple minerals into one nanoscale framework and tuning the heating step, the researchers created surfaces that are strongly attractive to Safranin O under the right pH, yet can be reset with an acid rinse. While more work is needed to scale up and test with actual industrial effluents, the study shows that cleverly engineered, low‑cost inorganic hybrids can match or surpass many state‑of‑the‑art adsorbents. If integrated into treatment plants as filters or packed beds, such materials could help strip vivid, potentially harmful colors from wastewater before it returns to the environment.
Citation: Abdelrahman, E.A., Basha, M.T. Novel carbonate, oxide, and hydroxide nanohybrids based on Mg, Ba, and Ca for efficient Safranin O dye adsorption. Sci Rep 16, 2624 (2026). https://doi.org/10.1038/s41598-026-36376-7
Keywords: wastewater treatment, dye pollution, nanocomposite adsorbent, Safranin O removal, water purification