Clear Sky Science · en
Unraveling COPD pathogenesis: a multi-omics approach to identify metabolites and genetic links
Why lung disease and body chemistry are linked
Chronic obstructive pulmonary disease (COPD) is best known as a smoker’s lung disease, but this study shows that what happens in our blood chemistry may be just as important as what we breathe in. By combining big genetic datasets, detailed measurements of small molecules in the blood, and lab experiments on human airway cells, the researchers uncover how disrupted fat processing in the body may help drive COPD—and how a familiar inhaler drug, Salbutamol, could be helping in a surprising new way.
Looking inside COPD beyond smoking
COPD affects hundreds of millions of people worldwide and is projected to become the third leading cause of death. While smoking and air pollution are major culprits, they do not fully explain who gets COPD or how fast it progresses. The team behind this work asked whether specific blood molecules, called metabolites, are not just markers of damage but active players in the disease. They focused on more than 1,400 metabolites measured in large European studies and linked these to COPD risk using people’s genetic differences as a kind of natural experiment.
Using genes to test cause and effect
To move beyond simple correlation, the researchers used a technique called Mendelian randomization. In plain terms, they tracked whether people who are born with gene variants that raise or lower certain metabolites also have higher or lower chances of developing COPD. This approach helps separate true causes from lifestyle habits that can muddy the picture, like coffee drinking or smoking. Out of 1,400 metabolites, six initially stood out as linked to COPD risk. However, a second, more stringent genetic test showed that only two—Carnitine C14 and 3-hydroxyoleoylcarnitine—share the same underlying genetic signal as COPD itself. Both are related to how the body handles long-chain fatty acids, suggesting that fat metabolism, not just inflammation and airflow, lies at the heart of COPD.
From molecules to key control switches
Once the two reliable metabolites were identified, the researchers traced them back to the metabolic “roads” they travel. These roads converged on fatty acid metabolism and highlighted a set of enzymes that act like traffic lights, controlling how much fat is built or broken down—especially the enzymes ACACA and ACACB. By mining existing drug and protein-interaction databases, the team discovered that these enzymes sit in the same network as ADRB2, the protein target of Salbutamol, a common rescue inhaler for COPD. Analyses of lung tissue from patients showed that ACACA and ACACB were turned up, while ADRB2 was turned down, consistent with a state of fatty acid overload in diseased lungs.
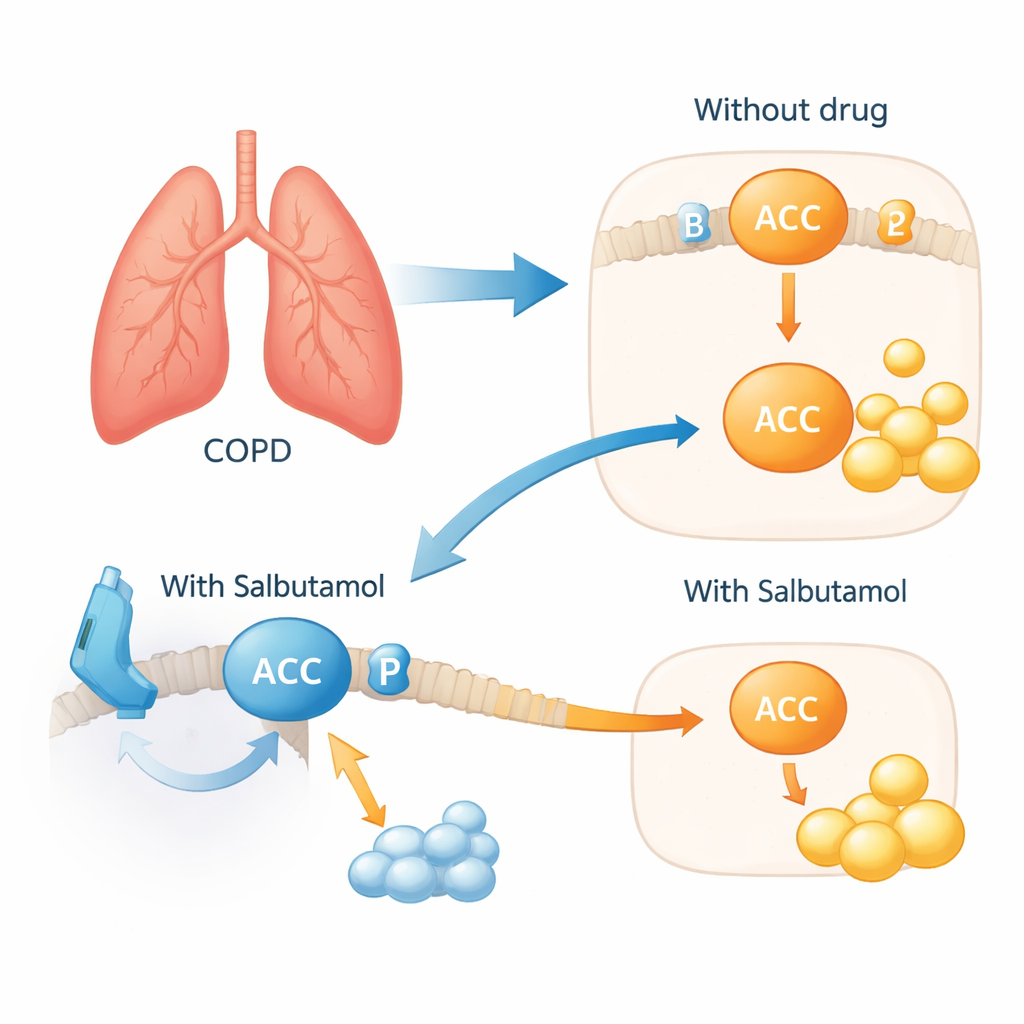
An inhaler that also calms runaway fats
To test whether this network matters in living cells, the scientists exposed human bronchial cells to cigarette smoke extract to mimic COPD-like stress. The cells responded by cranking up ACACA and ACACB and reducing a protective chemical tag on ACACA that normally keeps fat production in check. When Salbutamol was added, this harmful pattern reversed: the protective tag on ACACA was restored and signs of excessive fat synthesis dropped. In simple terms, the drug appeared to put the brakes back on fat-building machinery inside airway cells, acting through the ADRB2 "switch" at the cell surface. Lifestyle-based genetic analyses further supported the idea that the two key metabolites are tied to underlying biology rather than just to habits like coffee intake, which explained away several other candidate molecules.
What this means for patients and future treatments
For non-specialists, the take-home message is that COPD may be driven in part by a metabolic traffic jam in how lung cells process fats, and that a widely used bronchodilator may also be quietly helping to clear that jam. Carnitine C14 and 3-hydroxyoleoylcarnitine emerge as strong blood markers that could one day help flag people at higher risk or track disease progression. The enzymes ACACA and ACACB, sitting at key junctions in fat metabolism and linked to the Salbutamol target ADRB2, offer new handles for drug developers. Although more work in diverse populations and in-depth metabolic studies is needed, this multi-omics approach opens a path toward COPD treatments that do more than open airways—they may also restore healthier fuel use inside the lungs.
Citation: Zeng, M., Liu, J., Cao, X. et al. Unraveling COPD pathogenesis: a multi-omics approach to identify metabolites and genetic links. Sci Rep 16, 6013 (2026). https://doi.org/10.1038/s41598-026-36368-7
Keywords: COPD, fatty acid metabolism, Salbutamol, carnitine biomarkers, multi-omics