Clear Sky Science · en
Trade-off between canonical and unusual recombination sites promotes diversity and stability of gene cassette arrays of mobile integrons
How Bacteria Shuffle Their Defenses
Bacteria in hospitals face a constant barrage of antibiotics, yet many survive and spread. One major reason is a genetic gadget called an integron, which lets bacteria rapidly collect and rearrange resistance genes. This paper explores an unusual way these gadgets work, showing how bacteria balance two needs at once: keeping their resistance genes stable enough to rely on, but flexible enough to adapt to new drugs.
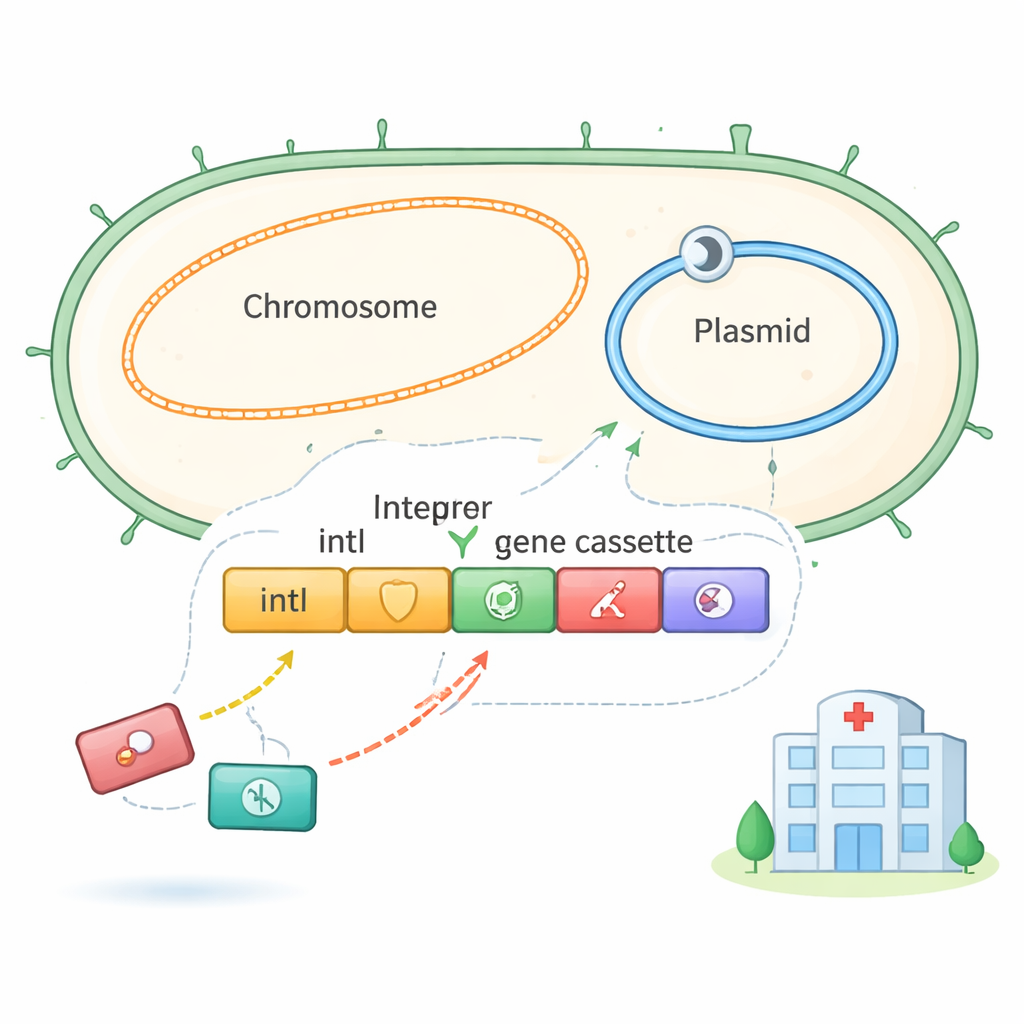
A Genetic Toolbox on Standby
Many Gram-negative bacteria carry integrons, small genetic platforms that act like docking stations for “gene cassettes.” Each cassette typically holds a single gene, often for antibiotic resistance, along with a short DNA tag that tells the integron’s enzyme, IntI, where to cut and paste it. Two main tag types are known: attI, usually at the start of the integron, and attC, which marks standard cassettes. When conditions change, such as under antibiotic stress, IntI can reshuffle these cassettes—adding new ones, removing others, or changing their order—allowing the bacterium to quickly adjust its defenses.
Strange Tags with Important Roles
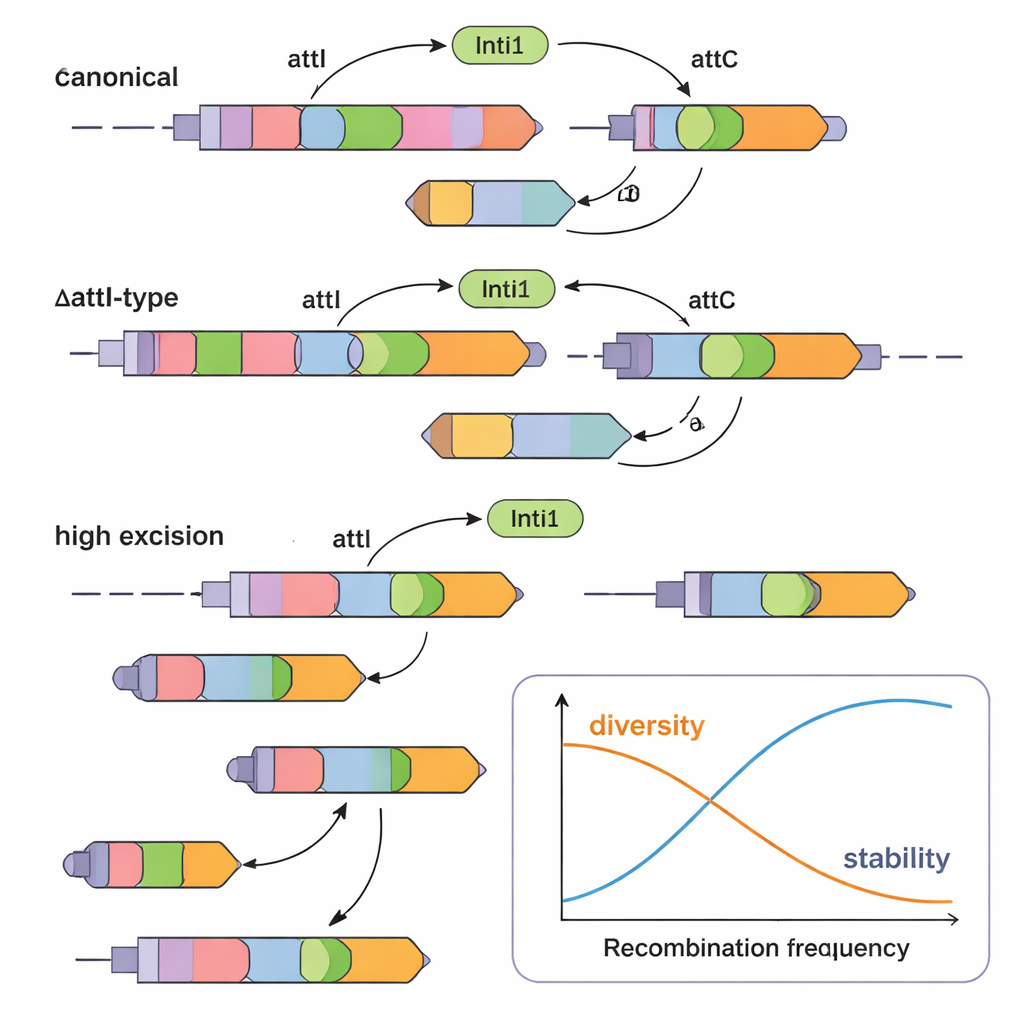
Beyond the well-studied attI and attC tags, the authors focused on odd, partly deleted versions of attI, called ΔattI sites. These appear in “unusual” gene cassettes where the usual attC tag has been replaced by a fragment of attI. By scanning over 1,700 complete integrons from public databases, the team found eight distinct ΔattI types across class 1 and class 2 integrons, mostly in hospital-related bacteria. They identified 26 unusual ΔattI-type cassettes, many encoding resistance to multiple antibiotic families. One, carrying the blaOXA-10 resistance gene, was especially common, highlighting that even these strange-looking cassettes are widespread and clinically relevant.
Putting Unusual Cassettes to the Test
To see whether these ΔattI-type cassettes actually function, the researchers recreated several examples in the lab. They placed them on plasmids in E. coli along with the IntI1 enzyme and measured how often cassettes were cut out (excision) or inserted into an attI site. They compared canonical designs (with a normal attC at one end) to unusual ones where the downstream attC was replaced by either a short or long ΔattI fragment. The surprising result: many ΔattI-type cassettes were fully active. Some were excised less efficiently than classical cassettes but inserted just as well, while others, especially those with a certain ΔattI2 fragment, were excised at rates similar to standard cassettes. This shows that even partially deleted recombination tags can still serve as robust substrates for the integron machinery.
Why Duplicated Genes Rarely Last
The authors also tackled another puzzle: why it is rare to see the same resistance cassette duplicated back-to-back in integrons, even though this could boost resistance by increasing gene dosage. They engineered artificial cassettes flanked by identical recombination sites on both sides—either two attI1 sites or two identical attC sites around the same gene. In these designs, IntI1 cut them out with striking efficiency, often above 97% and up to 100%. In other words, as soon as a perfect tandem cassette forms, it is almost instantly removed. This built-in instability helps explain why duplicated cassettes are uncommon in nature unless strong antibiotic pressure actively favors keeping them.
A Built-In Balance Between Change and Control
Seen together, the results reveal a subtle balance. Integrons use a mix of attI, attC, and ΔattI sites to fine-tune how easily different cassettes move. Canonical arrangements support efficient reshuffling, while ΔattI-type cassettes and the rapid loss of perfect duplicates prevent the array from filling up with copies of the same gene. This trade-off allows bacteria to maintain a diverse set of resistance options while avoiding overly unstable or redundant configurations. For a lay observer, the key message is that bacteria are not just passively collecting resistance genes; they use a sophisticated DNA system that actively manages which genes are kept, which are copied, and which are discarded, helping them thrive in the antibiotic-rich environment of modern hospitals.
Citation: Gonzales Machuca, A., Molina, M.C., Álvarez, V.E. et al. Trade-off between canonical and unusual recombination sites promotes diversity and stability of gene cassette arrays of mobile integrons. Sci Rep 16, 6133 (2026). https://doi.org/10.1038/s41598-026-36353-0
Keywords: antibiotic resistance, integrons, gene cassettes, bacterial evolution, hospital infections