Clear Sky Science · en
CDC25C downregulation suppresses HCC growth via mitochondrial stress-induced autophagy and apoptosis
Why this liver cancer study matters
Liver cancer is one of the deadliest cancers worldwide, and many patients are diagnosed too late for surgery or standard treatments to work well. This study explores a single cellular “switch,” a protein called CDC25C, that appears to drive liver cancer growth while being less critical for healthy liver cells. By turning this switch down, the researchers found they could stress and ultimately kill cancer cells through the cell’s own self-cleaning and self-destruct systems, pointing to a promising and more selective treatment strategy.
A growth switch inside liver tumors
Every cell must decide when to divide and when to stop. CDC25C is one of the key proteins that pushes cells through the division cycle. In liver cancer, this switch is often stuck in the “on” position, helping tumors grow and spread. The team used mouse liver cancer cells and normal mouse liver cells, as well as tumor-bearing mice, to artificially lower CDC25C levels. They confirmed, using standard genetic and protein tests, that CDC25C was sharply reduced in their experimental group compared with controls, establishing a solid model to study what happens when this growth switch is dialed down.
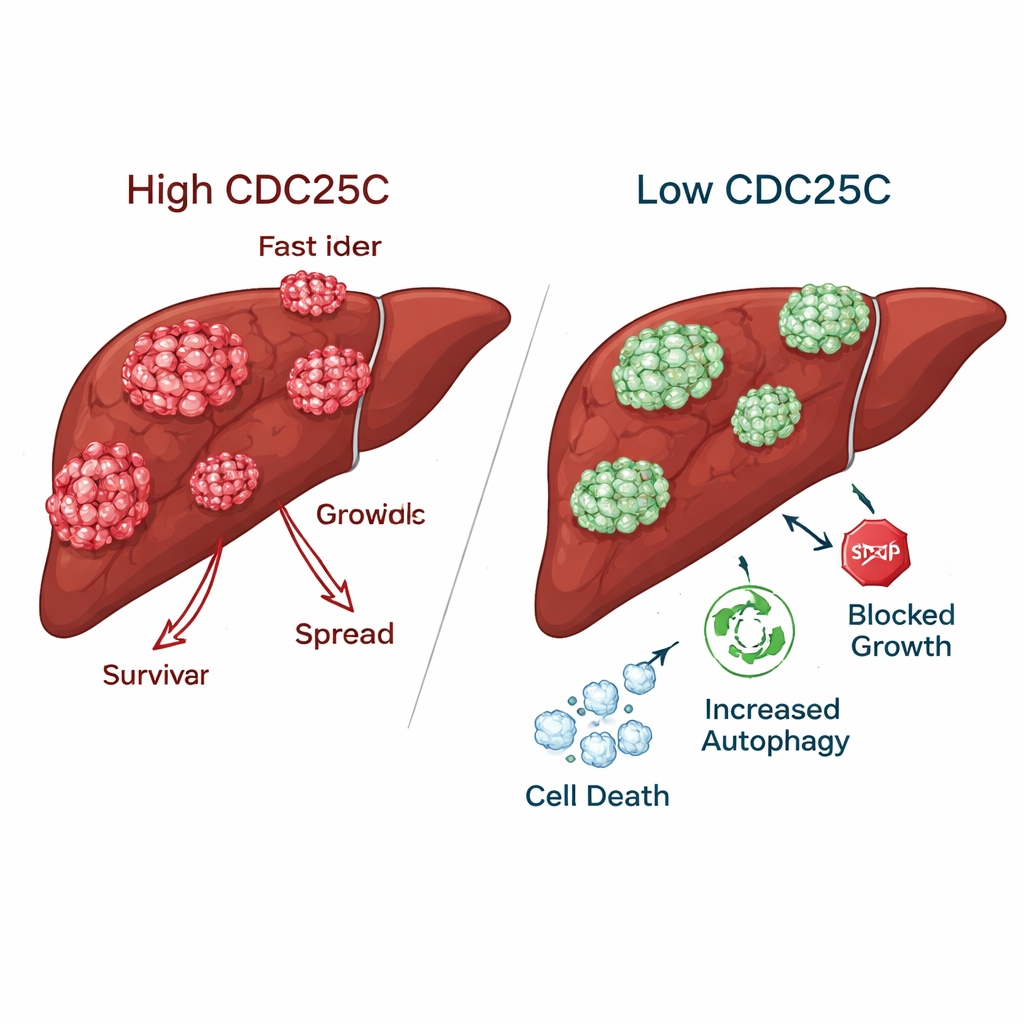
Slowing cancer while sparing healthy liver cells
When CDC25C was reduced in liver cancer cells, the tumors lost many of their aggressive traits. Cancer cells formed far fewer colonies, migrated more slowly across artificial “wounds,” and were less able to move through and invade barriers in laboratory tests. In contrast, normal liver cells actually showed better growth and movement when CDC25C was lowered, suggesting that healthy tissue may tolerate or even benefit from CDC25C inhibition. In mice, tumors engineered with reduced CDC25C showed lower levels of this protein in both gene and protein measurements, confirming that the same effect occurred in living animals.
Powerhouse stress and the cell’s cleanup crew
Looking inside the cells with electron microscopes, the researchers saw that cancer cells with low CDC25C contained swollen, damaged mitochondria—the tiny power plants of the cell—along with bubble-like structures called autophagosomes that signal activation of the cell’s internal recycling system, autophagy. They then measured key signs of mitochondrial stress: calcium levels and reactive oxygen species (ROS), both of which were elevated in cancer cells after CDC25C was reduced. Markers of the mitochondrial stress response, including helper proteins like HSP60 and two mitochondrial “quality control” enzymes, were also increased, as was CHOP, a stress-related factor that links mitochondrial trouble to broader cellular responses. Strikingly, these stress signals moved in the opposite direction in normal liver cells, reinforcing the idea that cancer and healthy cells respond very differently to CDC25C loss.
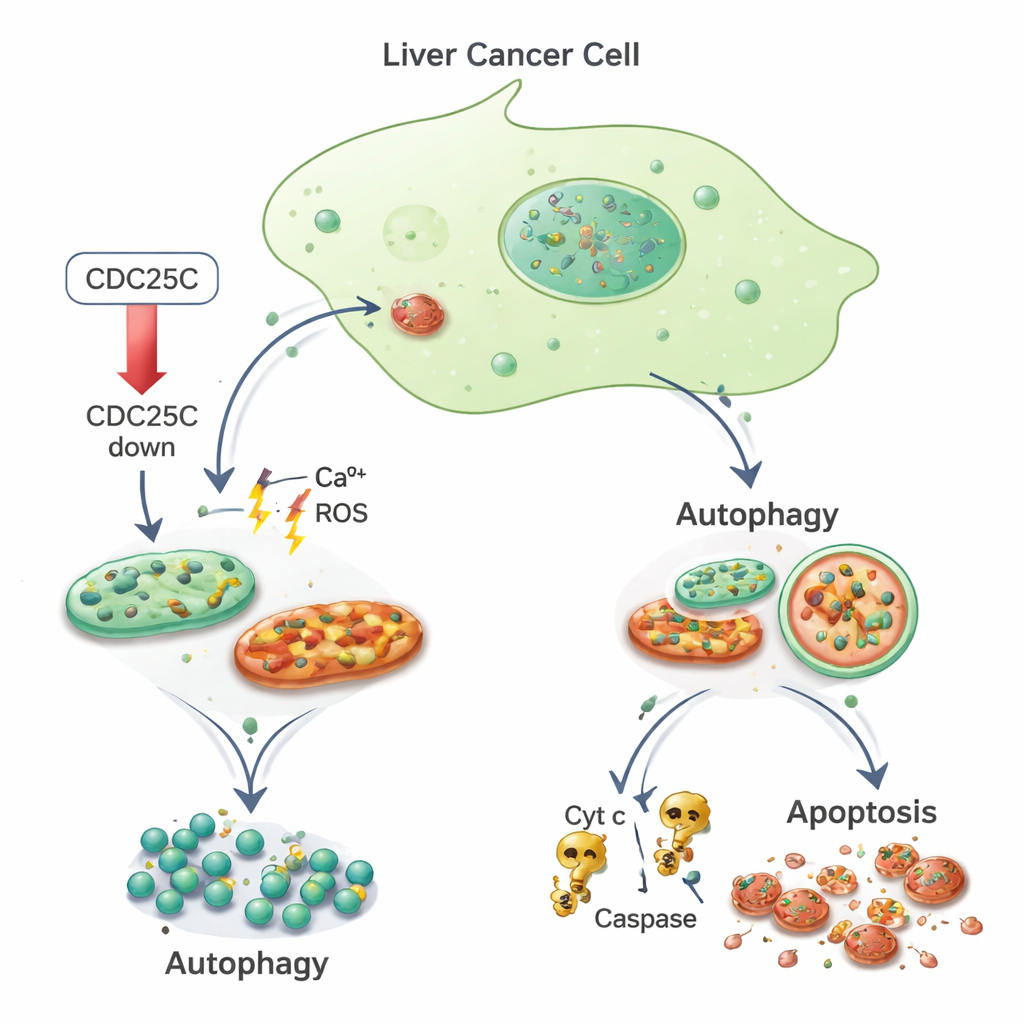
From stress to self-digestion and cell death
The team next asked how these stressed cancer cells were coping. They found that markers of autophagy were boosted in both cancer and normal liver cells when CDC25C was reduced. Proteins such as LC3 and p62, which reflect the formation and activity of autophagosomes, rose in level, indicating that cells were stepping up their cleanup machinery. At the same time, in cancer cells only, mitochondrial damage appears to push cells beyond repair and into programmed cell death. Using fluorescent stains and flow cytometry, the researchers saw more cancer cells with fragmented, brightly stained nuclei—a hallmark of apoptosis—and reduced mitochondrial membrane potential. They also found increased levels of cytochrome c and the enzymes Caspase-3 and Caspase-9, classic components of the mitochondria-driven death pathway. Normal liver cells, by contrast, showed less apoptosis under the same CDC25C-lowering conditions.
What this could mean for future treatments
For a lay reader, the key message is that turning down CDC25C seems to hit liver cancer cells where they are vulnerable, overloading their powerhouses, pushing their recycling systems into overdrive, and ultimately triggering clean self-destruction—while mostly sparing normal liver cells. The authors conclude that CDC25C is more than a simple growth switch; it also shapes how cells handle stress, cleanup, and death. Because cancer cells rely heavily on CDC25C and respond to its loss with fatal mitochondrial stress and apoptosis, drugs that target this protein, or its downstream stress pathways, could one day offer a more selective way to treat liver cancer with fewer harms to healthy liver tissue.
Citation: Miao, X., Zheng, F., Mo, X. et al. CDC25C downregulation suppresses HCC growth via mitochondrial stress-induced autophagy and apoptosis. Sci Rep 16, 5631 (2026). https://doi.org/10.1038/s41598-026-36351-2
Keywords: hepatocellular carcinoma, CDC25C, mitochondrial stress, autophagy, apoptosis