Clear Sky Science · en
Graphene oxide/metal–organic framework composite as an effective catalyst for esterification reactions
Turning Everyday Chemicals into Cleaner Fuels
Ester molecules are everywhere: in the flavors of fruit, the scents of perfumes, and as key ingredients in biodiesel fuels. Making esters on an industrial scale, however, often requires harsh acids and high temperatures that generate waste and corrode equipment. This study explores a new solid catalyst — a carefully engineered material called MOF-801@GO — that can drive ester-making reactions more efficiently and cleanly, potentially making bio-based fuels and fine chemicals cheaper and greener to produce.
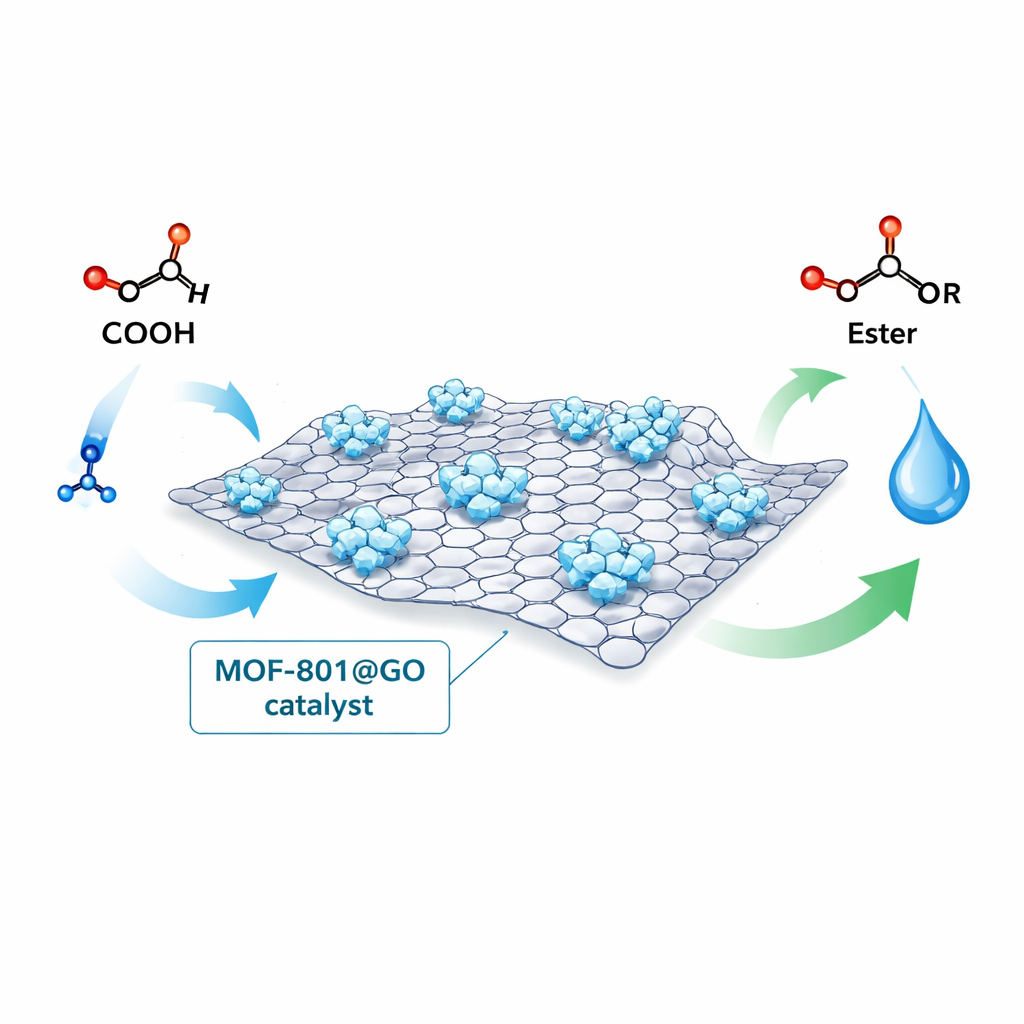
Building a Smart Solid Helper
The researchers combined two advanced materials to create their catalyst. The first is graphene oxide, an ultra-thin carbon sheet only one atom thick but several micrometers wide, covered with oxygen-containing groups that make it easy to disperse and modify. The second is MOF-801, a metal–organic framework made from zirconium atoms linked by small organic molecules into an open, sponge-like crystal. By growing MOF-801 particles directly on graphene oxide sheets, they formed a composite called MOF-801@GO in which the crystals are anchored and spread out across a large surface. This design aims to expose more active spots where reactant molecules can attach and react.
Checking the Material from All Angles
To confirm what they had made, the team used a suite of characterization tools. Infrared spectroscopy showed the chemical fingerprints of both the graphene oxide and the MOF-801 framework in the final composite, indicating that both components remained present and intact. Electron microscopes revealed that the graphene oxide formed wrinkled, sheet-like layers, while MOF-801 appeared as small, roughly micrometer-sized crystals decorating those sheets. X-ray diffraction patterns matched those of pure MOF-801, confirming that its crystal structure survived, while subtle changes signaled that the framework was well integrated with the graphene oxide rather than simply mixed in as a separate powder.
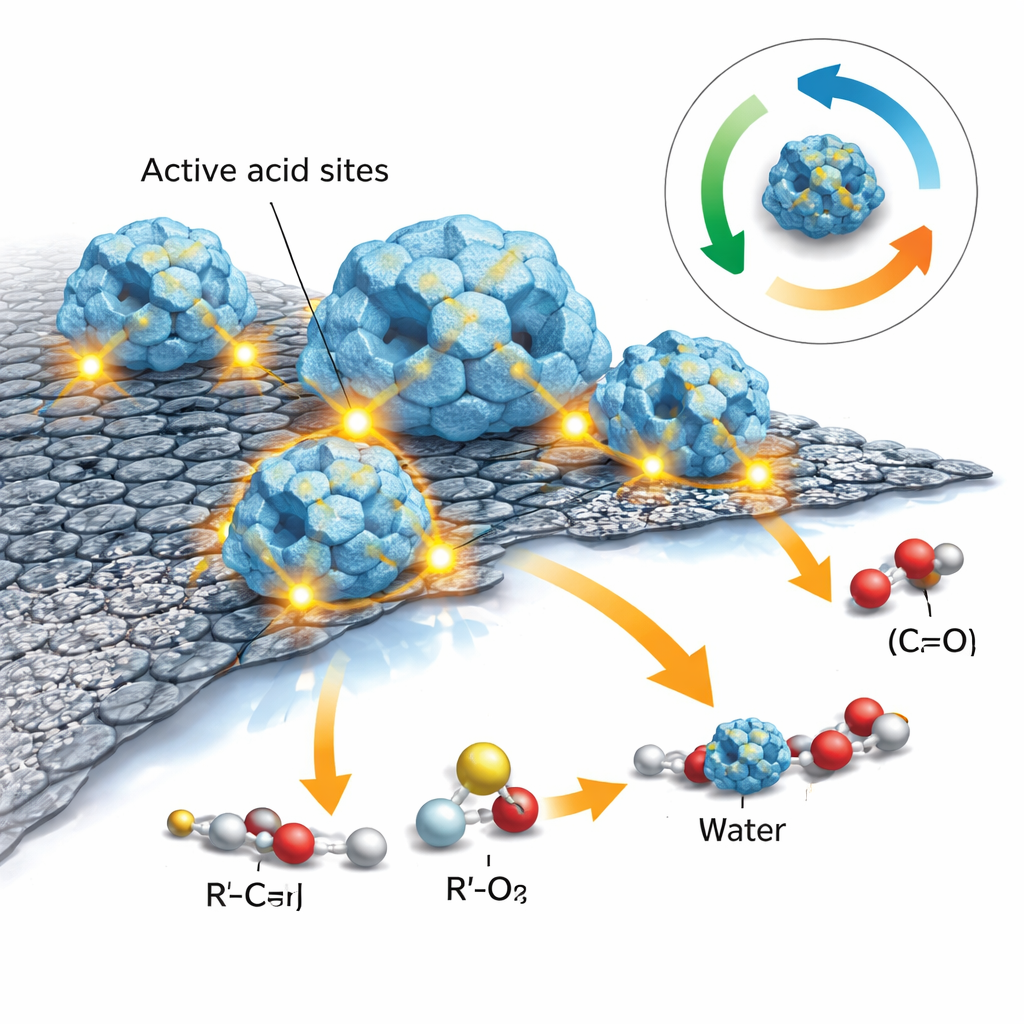
Why the Catalyst is So Active
Beyond just structure, the key question is how many and what types of “acid sites” the material offers, because these spots on the surface act as the workbenches where ester molecules are formed. Using a technique that tracks how ammonia gas is released from the material as it is heated, the authors found two main kinds of sites: weaker ones associated with hydroxyl and carboxyl groups on the graphene oxide and framework, and stronger ones tied to exposed zirconium centers inside MOF-801. The combination greatly increases the number of medium-strength acid sites compared with the unmodified MOF, suggesting that the interface between the graphene and the framework boosts the material’s ability to activate reactant molecules.
Making Esters Efficiently and Repeatedly
The team then tested the catalyst in standard esterification reactions, in which a carboxylic acid combines with an alcohol to form an ester and water. Using acetic acid with various alcohols under solvent-free conditions, small amounts of MOF-801@GO were enough to reach yields up to about 95–98% at moderate temperatures around 80 °C. In contrast, using only graphene oxide, only MOF-801, or a simple zirconium salt led to much lower conversions, highlighting the synergy of the composite material. The catalyst also worked well for a range of acids and alcohols, showing that it is not limited to a single reaction pair and could be broadly useful for producing diverse esters, including those used as biodiesel components.
Built to Last Through Many Cycles
For any industrial process, a catalyst must be not only active but also durable. Here, MOF-801@GO proved robust under repeated use. After each reaction, the solid catalyst could be separated, washed, and reused with only a small drop in performance after several cycles. Careful measurements showed very little loss of zirconium to the liquid phase, meaning the active metal stayed locked in the solid material. Imaging and spectroscopic tests on the used catalyst looked nearly identical to those on the fresh sample, confirming that its structure remained stable. A control test in which the solid was removed mid-reaction showed that the reaction essentially stopped, proving that the catalysis is truly due to the solid material rather than metal that has dissolved.
A Step Toward Greener Ester Production
In simple terms, this work presents a solid, reusable helper that can turn common acids and alcohols into esters under relatively mild and cleaner conditions. By fusing a porous zirconium-based framework onto flexible graphene oxide sheets, the researchers created a catalyst with many accessible active sites that stays intact over repeated uses. Such materials could help future chemical plants and biodiesel producers reduce waste, cut down on corrosive liquid acids, and make everyday products — from fuels to fragrances — in a more environmentally friendly way.
Citation: Masoudi, R., Zarnegaryan, A. & Dehbanipour, Z. Graphene oxide/metal–organic framework composite as an effective catalyst for esterification reactions. Sci Rep 16, 7771 (2026). https://doi.org/10.1038/s41598-026-36344-1
Keywords: graphene oxide, metal–organic framework, heterogeneous catalysis, esterification, biodiesel