Clear Sky Science · en
Imaging galvanic couples in complex sulphide assemblages using multi-modal elemental and photocurrent microscopy
Hidden batteries inside common rocks
Metal-rich rocks that host copper, zinc, and gold are not just passive lumps of stone. At tiny scales, they can behave like networks of microscopic batteries, with different minerals acting as positive and negative electrodes. These hidden electrical pairs help control how fast ores dissolve during processing, and how quickly mine wastes generate acid drainage that pollutes water. This study shows how to actually “see” those micro-batteries inside complex sulphide rocks, using a combination of high‑resolution chemical mapping and a specialised light‑based electrical imaging technique.
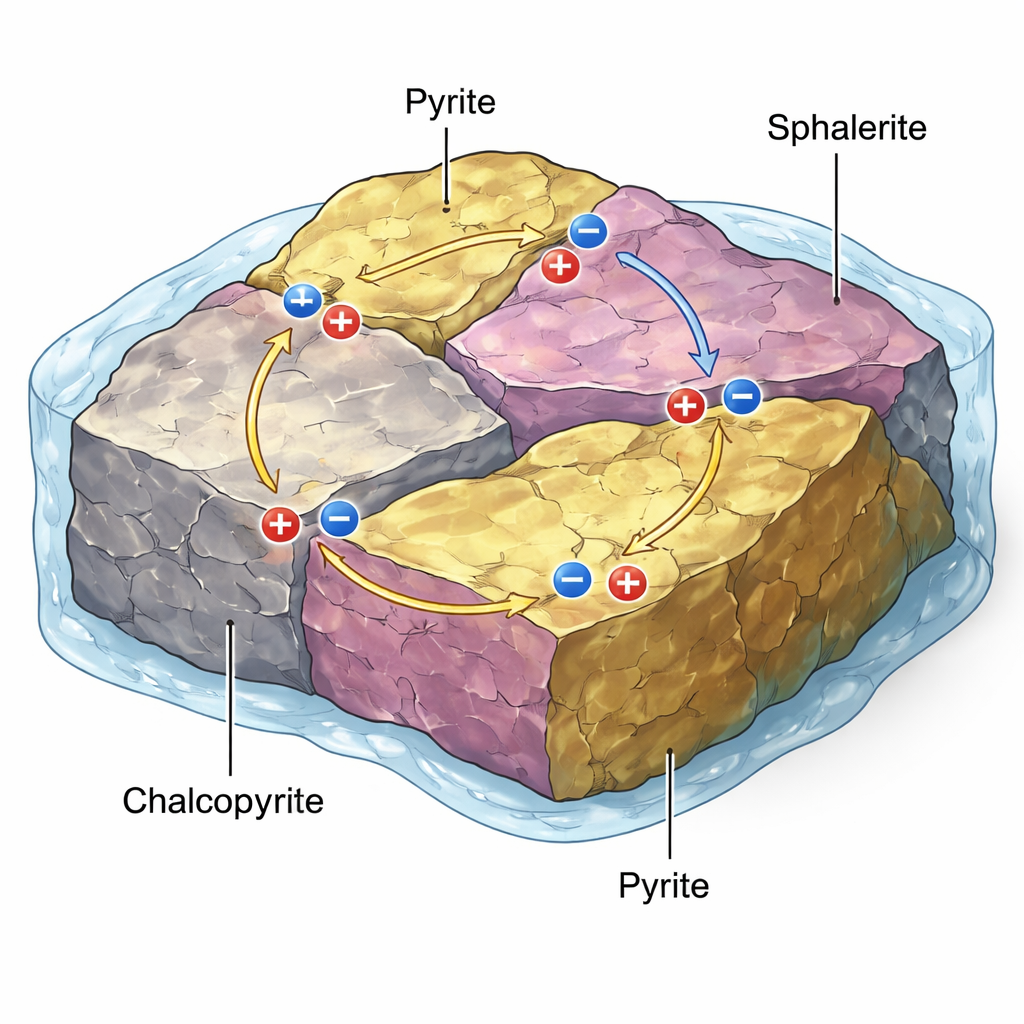
Why tiny electrical pairs in ore matter
In many metal deposits, minerals like pyrite (“fool’s gold”), sphalerite (a zinc sulphide), and chalcopyrite (a copper‑iron sulphide) touch each other along intricate grain boundaries. Because these minerals are natural semiconductors with different internal energy levels, their contacts can behave like miniature galvanic cells—essentially micro‑batteries. When these rocks meet acidic or oxygen‑rich fluids, the electrical potential between mineral pairs can drive one mineral to dissolve faster (acting as the anode), while another is protected (acting as the cathode). This galvanic action can speed up the release of metals during leaching and flotation, or drive the formation of acid mine drainage when waste rocks weather at the surface.
Seeing chemistry and electricity at once
To study these effects, the authors examined a pyrite‑rich rock from the Otago Schist in New Zealand, packed with tiny inclusions of sphalerite and other sulphides. First, they used electron microprobe analysis and a nuclear microprobe to create detailed elemental maps, showing where iron, zinc, arsenic, cobalt and other trace elements are concentrated. These impurities are crucial because they subtly tune the semiconducting properties of each grain, shifting whether a region behaves more like a positive (p‑type) or negative (n‑type) side of a junction. The maps revealed strongly zoned pyrite—bands richer in arsenic or cobalt—and numerous iron‑rich sphalerite grains, suggesting many potential electrical junctions at the micrometre scale.
Lighting up active micro‑batteries
The central tool in this work is laser beam induced current (LBIC) microscopy. A violet laser (405 nm) is scanned across the polished rock surface, while two tiny probes, placed some distance away, measure the photocurrent generated inside the minerals. Where a strong internal electric field exists—such as at a galvanic contact between pyrite and sphalerite—the light frees charge carriers that are swept along that field, producing a measurable current signal. By modulating the laser and using lock‑in detection, the researchers can pick out extremely weak signals from noise. When these photocurrent maps are overlaid on the elemental images, bright hotspots line up with specific sphalerite–pyrite contacts, confirming they behave as active micro‑batteries in three dimensions.
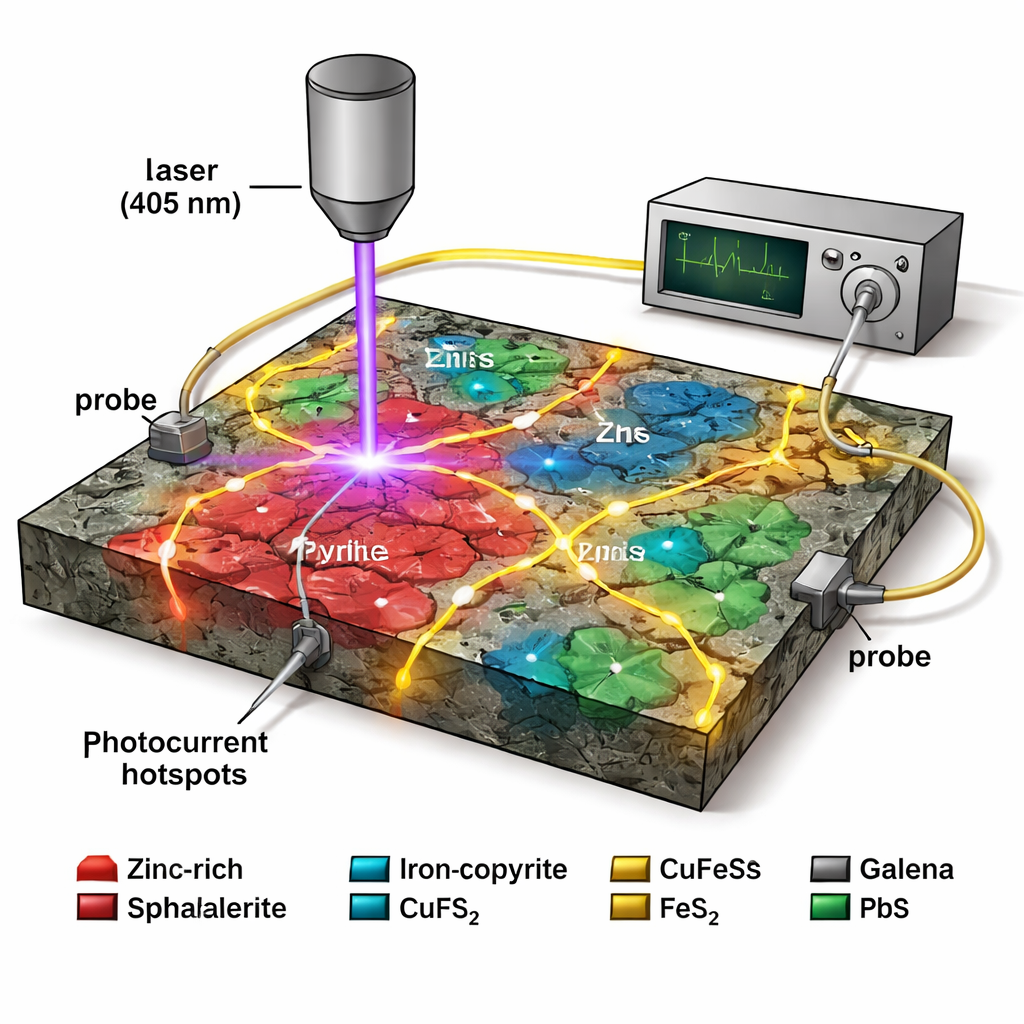
Not all grains behave the same
Intriguingly, the study found that not every sphalerite grain lit up under LBIC, even when its chemistry looked similar to neighbouring grains that did. Several small iron‑rich sphalerite inclusions beside a pyrite grain boundary produced strong photocurrents, while a much larger sphalerite grain nearby was almost silent. The authors explore several explanations: the large grain may be thicker than the depth reached by the laser, so only weak lateral junctions at its rim contribute; thin films of sulphur or oxidation products may partially insulate it; or local variations in impurity content may create weaker junctions, or a less favourable type of contact, reducing the driving voltage. This grain‑to‑grain variability highlights how texture and microstructure, not just bulk composition, control electrochemical behaviour.
What this means for mines and the environment
For non‑specialists, the broader message is that the way minerals are mixed and wired together inside a rock can be as important as the overall chemistry. The multi‑modal approach demonstrated here—combining chemical imaging with photocurrent mapping—offers a way to screen ore samples for where galvanic couples are active, over areas large enough to be representative of “real rock.” In practical terms, that could help engineers fine‑tune leaching and flotation strategies for complex, low‑grade ores, and improve predictions of which waste rocks are most likely to generate acid drainage. Although some details, such as why certain large grains stay electrically “quiet,” remain to be fully resolved, this work shows that hidden electrical landscapes inside rocks can now be imaged directly, opening new paths to cleaner and more efficient mineral processing.
Citation: Laird, J.S., Macrae, C.M. & Ryan, C. Imaging galvanic couples in complex sulphide assemblages using multi-modal elemental and photocurrent microscopy. Sci Rep 16, 6442 (2026). https://doi.org/10.1038/s41598-026-36337-0
Keywords: galvanic corrosion, sulphide minerals, geometallurgy, acid mine drainage, photocurrent microscopy