Clear Sky Science · en
Mechanical stability and thermodynamic properties of GeP and $$\hbox {GeP}_{3}$$ as battery anode materials from first principles
Why new battery materials matter
From smartphones to electric cars, modern life leans heavily on rechargeable batteries. Most of today’s lithium-ion batteries still rely on graphite, a decades‑old material, to store and release charge. But graphite is nearing its performance limits, especially for applications that demand fast charging, high capacity, and long lifetimes. This study explores germanium phosphides—compounds made of germanium and phosphorus—as possible replacements for graphite in battery anodes, asking a simple but crucial question: which version of these materials can store a lot of energy while also surviving years of swelling and shrinking inside a working battery?
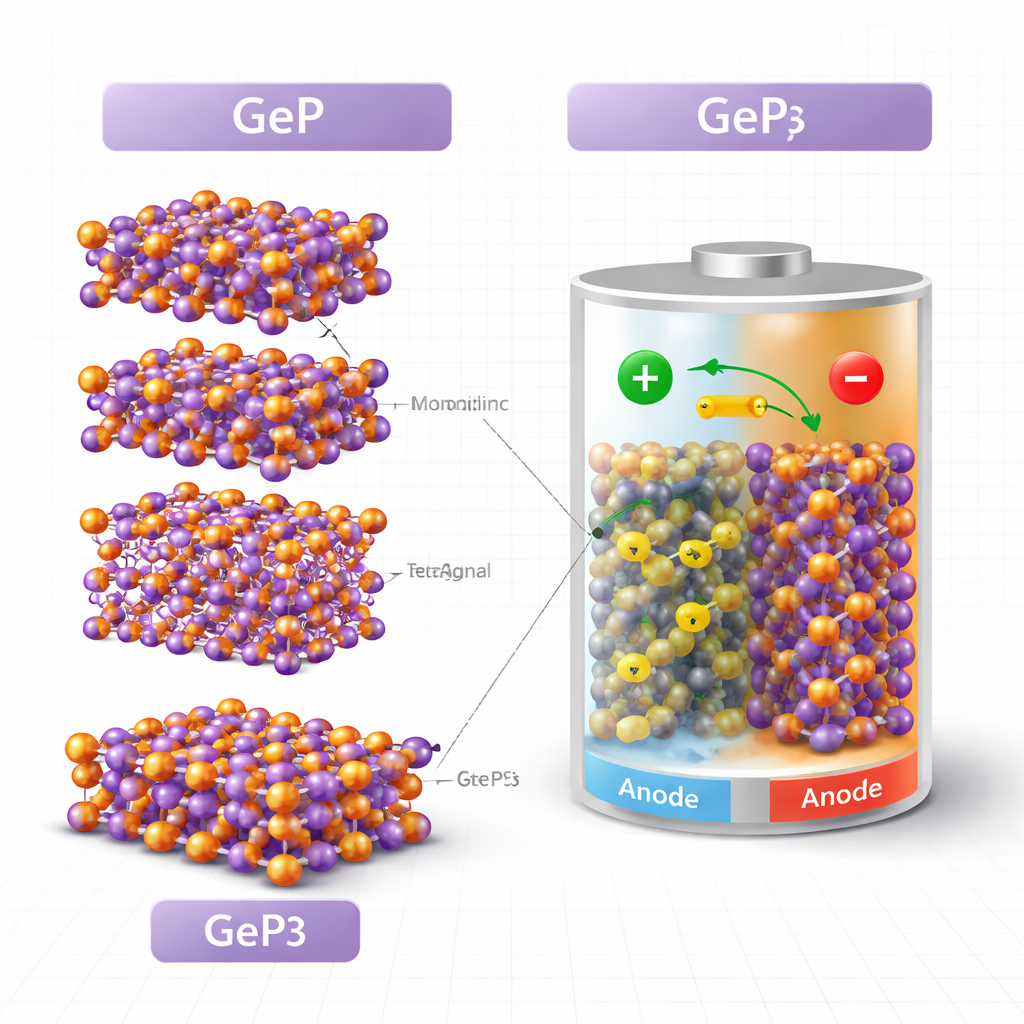
Meet the family of germanium phosphides
The researchers focus on four related crystals: three forms (or polymorphs) of GeP and one phosphorus‑richer compound, GeP3. Although these materials share the same elements, their atoms are arranged differently, giving each a distinct personality. Using quantum‑mechanical calculations, the team first recreated the crystal structures and checked them against known experiments, confirming that their models closely match reality. The monoclinic form of GeP (GeP‑mono) is layered and relatively open, which may help it host lithium ions. The tetragonal form (GeP‑tetra) is more tightly packed and symmetric, while the cubic form (GeP‑cubic) has the highest symmetry on paper but, as the study shows, turns out to be mechanically unsound. GeP3, with three times as much phosphorus, adopts a robust layered network where germanium and phosphorus atoms form a strong, three‑dimensional framework.
How these crystals handle stress
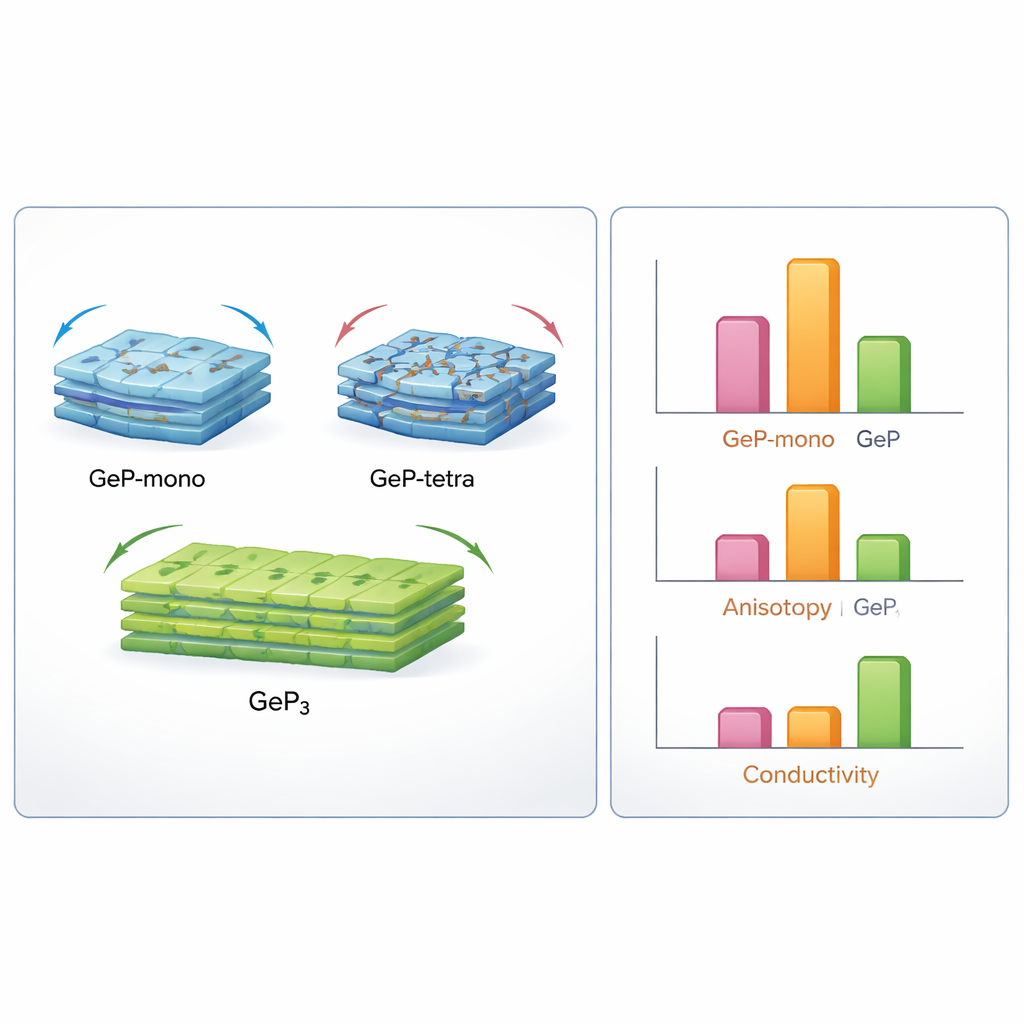
Inside a battery, anode materials must endure repeated volume changes as lithium or sodium ions move in and out. If the material is too rigid or uneven in how it stretches, it can crack, crumble, and lose capacity. By virtually squeezing, shearing, and bending the crystals, the authors calculate key measures of stiffness and flexibility, such as bulk and shear moduli. GeP‑tetra comes out extremely stiff and brittle: it resists deformation but is likely to fracture under the large volume swings typical of high‑capacity anodes. GeP‑mono is much softer and more flexible overall but behaves very differently along different directions in the crystal, which can funnel stress into weak planes. GeP‑cubic fails even basic stability tests, suggesting it would collapse rather than hold together in a real electrode. GeP3 lands in the middle ground—stiffer than GeP‑mono but less rigid than GeP‑tetra, and, importantly, with far more uniform behavior in different directions.
Electricity flow and heat management
For an anode to work well, it must not only survive mechanical stress but also conduct electrons efficiently. The team calculates the electronic band structures and densities of states for each material, which reveal whether they behave like semiconductors or metals. GeP‑mono is a semiconductor with a modest energy gap, meaning its natural conductivity is limited and would need help from additives such as carbon. In contrast, GeP‑tetra and GeP3 show metallic character: electrons can move freely, which is ideal for fast charging and discharging. Beyond electricity, the authors also estimate how these crystals store and conduct heat. GeP3 stands out again, with a higher heat capacity and stronger bonding than the GeP forms. That means it can buffer temperature spikes better and remain stable across a wider temperature range, both important for safety and performance in demanding uses like electric vehicles.
Balancing capacity with durability
High‑capacity anode materials often swell by 100–300 percent as they absorb ions, a punishing test for any solid. The study shows that all mechanically stable germanium phosphides are inherently brittle, but the way they distribute stress differs. GeP‑mono’s softness may help it accommodate volume changes, yet its extreme direction‑dependent behavior could trigger cracking along specific planes unless engineers carefully control particle size and orientation. GeP‑tetra’s great stiffness offers strength but leaves little room for safe swelling, making fracture a serious concern unless the material is used as very small particles or in reinforced composites. GeP3, with its moderate stiffness and low directional bias, promises more even expansion and contraction, reducing stress hotspots and improving long‑term cycling stability.
What this means for future batteries
By combining structural, mechanical, electronic, and thermodynamic calculations in one framework, the authors conclude that GeP3 is the most promising candidate among the phases they studied. It may not offer the absolute highest theoretical capacity, but it strikes a desirable balance: good mechanical resilience, metallic conductivity, and robust thermal behavior. GeP‑mono and GeP‑tetra could still play roles in specialized designs, provided their weaknesses are managed through nano‑engineering and composite architectures. Overall, the work offers a roadmap for choosing and designing germanium phosphide anodes that do not just store more energy than graphite, but also stand up to the mechanical and thermal realities inside next‑generation lithium‑ and sodium‑ion batteries.
Citation: Truong, D.T., Hoang, NH., Phan, C.M. et al. Mechanical stability and thermodynamic properties of GeP and \(\hbox {GeP}_{3}\) as battery anode materials from first principles. Sci Rep 16, 6058 (2026). https://doi.org/10.1038/s41598-026-36336-1
Keywords: battery anodes, germanium phosphide, lithium-ion batteries, mechanical stability, GeP3