Clear Sky Science · en
TET1 suppresses hepatocellular carcinoma progression by modulating the PI3K/Akt signaling pathways
Why a Liver Enzyme Matters for Cancer
Primary liver cancer, especially a type called hepatocellular carcinoma, is one of the deadliest cancers worldwide. Many patients are diagnosed too late for curative surgery, and existing drugs only help a fraction of them. This study explores a molecule called TET1, an enzyme that edits chemical tags on DNA, to ask a simple but vital question: does TET1 help liver tumors grow, and could it become a new target for treatment?
A Hidden Editor in Our DNA
Our genes are controlled not only by the DNA code itself but also by chemical marks that turn genes on or off. One common mark is DNA methylation, which acts like a dimmer switch on gene activity. TET1 is part of a family of enzymes that erase or reshape these marks, helping cells respond to their environment and maintain normal function. When this system goes wrong, cells can slip into disease states, including cancer. Earlier research showed that TET1 can either slow or speed up tumor growth depending on the tissue, but its exact role in human liver cancer remained unclear and hotly debated.
TET1 Levels Spike in Liver Tumors
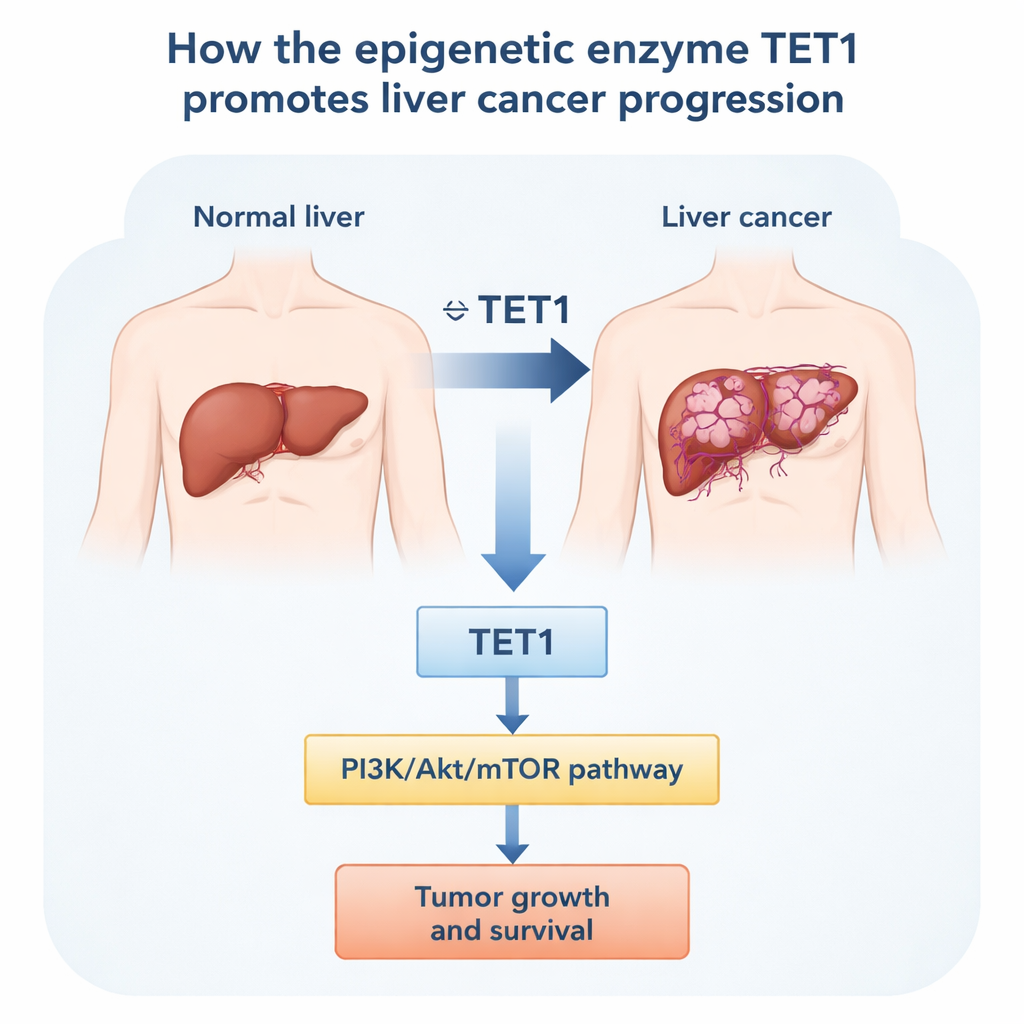
To find out what TET1 is doing in liver cancer, the researchers first turned to large public gene databases and then to patient samples. Across many cancer types, TET1 levels were unusually high in several tumors, including liver cancer. Focusing on liver tumors, they compared tumor tissue with nearby noncancerous liver from the same patients. In eight matched pairs, TET1 protein was consistently higher in the tumor. Public datasets from hundreds of patients told the same story at the RNA level: liver cancers had more TET1 than healthy livers or livers with scarring alone. Patients whose tumors showed higher TET1 tended to have more advanced disease and shorter overall and disease‑free survival, suggesting that TET1 is linked to both tumor aggressiveness and poor prognosis.
Turning TET1 Down Slows Tumor Cells
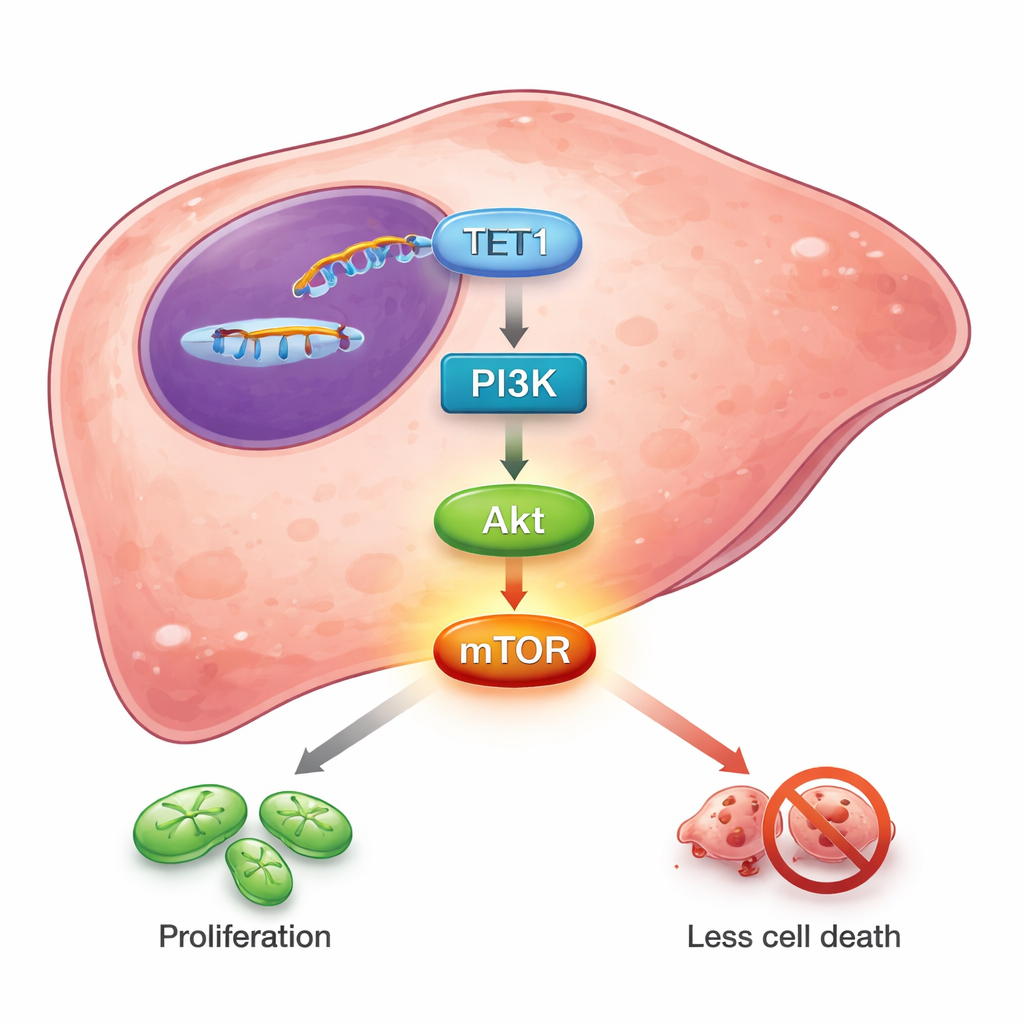
Next, the team asked what happens when TET1 is dialed down inside liver cancer cells grown in the lab. Using small pieces of RNA to silence the TET1 gene, they dramatically reduced TET1 levels in several human liver cancer cell lines, including those that carry hepatitis B virus–related changes. Once TET1 was suppressed, the cells divided more slowly, formed fewer colonies, and showed clear signs of stress. Flow‑cytometry experiments revealed that more cells were stalled in the early “G1” phase of the cell cycle, rather than pushing forward to divide. At the same time, the fraction of cells undergoing programmed cell death, or apoptosis, rose sharply. These effects appeared in both typical liver cancer cells and an HBV‑positive line, indicating that TET1 helps sustain survival and continual growth across different liver cancer backgrounds.
A Growth Pathway Under TET1’s Control
To uncover how TET1 exerts this influence, the researchers profiled which genes changed between normal liver cells and cancer cells. Thousands of genes were different, and many of them clustered into well‑known growth and survival circuits. One pathway stood out: PI3K/Akt/mTOR, a central signaling chain that many cancers use to boost metabolism, resist cell death, and shrug off therapies. Database analyses showed that TET1 levels rose in step with core components of this pathway, and fell in opposition to PTEN, a natural brake on PI3K/Akt activity. When TET1 was silenced in liver cancer cells, the activated, “phosphorylated” forms of PI3K, Akt, and especially mTOR dropped. Treating cells with a chemical inhibitor that blocks TET‑family enzyme activity likewise reduced phosphorylation of Akt and mTOR. These results suggest that TET1’s enzymatic activity—its ability to reshape DNA marks—feeds into the PI3K/Akt/mTOR cascade, tipping the balance toward growth and survival rather than restraint.
From Molecular Insight to Possible Treatments
Taken together, this work paints TET1 as a key ally of liver tumors rather than a guardian against them. High TET1 helps liver cancer cells proliferate and avoid death, in part by keeping the PI3K/Akt/mTOR pathway switched on. In patients, more TET1 in tumors goes hand in hand with more advanced disease and worse outcomes. For a lay reader, the message is that an enzyme quietly editing chemical tags on DNA can strongly influence whether liver cancer cells live or die. By designing drugs that inhibit TET1, or by pairing such drugs with existing PI3K/Akt‑targeted therapies, future treatments may be able to cut off a crucial support line for liver tumors and improve survival for people with this challenging disease.
Citation: Qi, S., Chen, M., Ding, Z. et al. TET1 suppresses hepatocellular carcinoma progression by modulating the PI3K/Akt signaling pathways. Sci Rep 16, 5791 (2026). https://doi.org/10.1038/s41598-026-36334-3
Keywords: liver cancer, TET1, epigenetics, PI3K Akt mTOR, hepatocellular carcinoma