Clear Sky Science · en
Fecal microbiome predicts treatment response after the initiation of semaglutide or empagliflozin uptake
Why your gut bacteria may shape diabetes treatment
People with type 2 diabetes increasingly receive newer drugs such as semaglutide (often known from weight‑loss ads) and empagliflozin, which help lower blood sugar and body weight. Yet not everyone responds equally well. This study asks a simple but powerful question: can the trillions of bacteria living in our gut help explain who benefits most from these medications, and could they someday guide more personalized diabetes care?
Inside the study and what was measured
Researchers followed 20 adults with type 2 diabetes who were already taking the standard drug metformin and had obesity. Their doctors added either semaglutide or empagliflozin based on medical need. Over a full year, participants provided stool samples four times and blood and urine samples three times. The stool samples were used to profile gut bacteria, while the blood and urine tests tracked key health markers such as long‑term blood sugar (HbA1c), body weight, kidney function, and signs of inflammation. This design allowed the team to see both how the drugs affected health and whether the starting gut microbiome could forecast those changes. 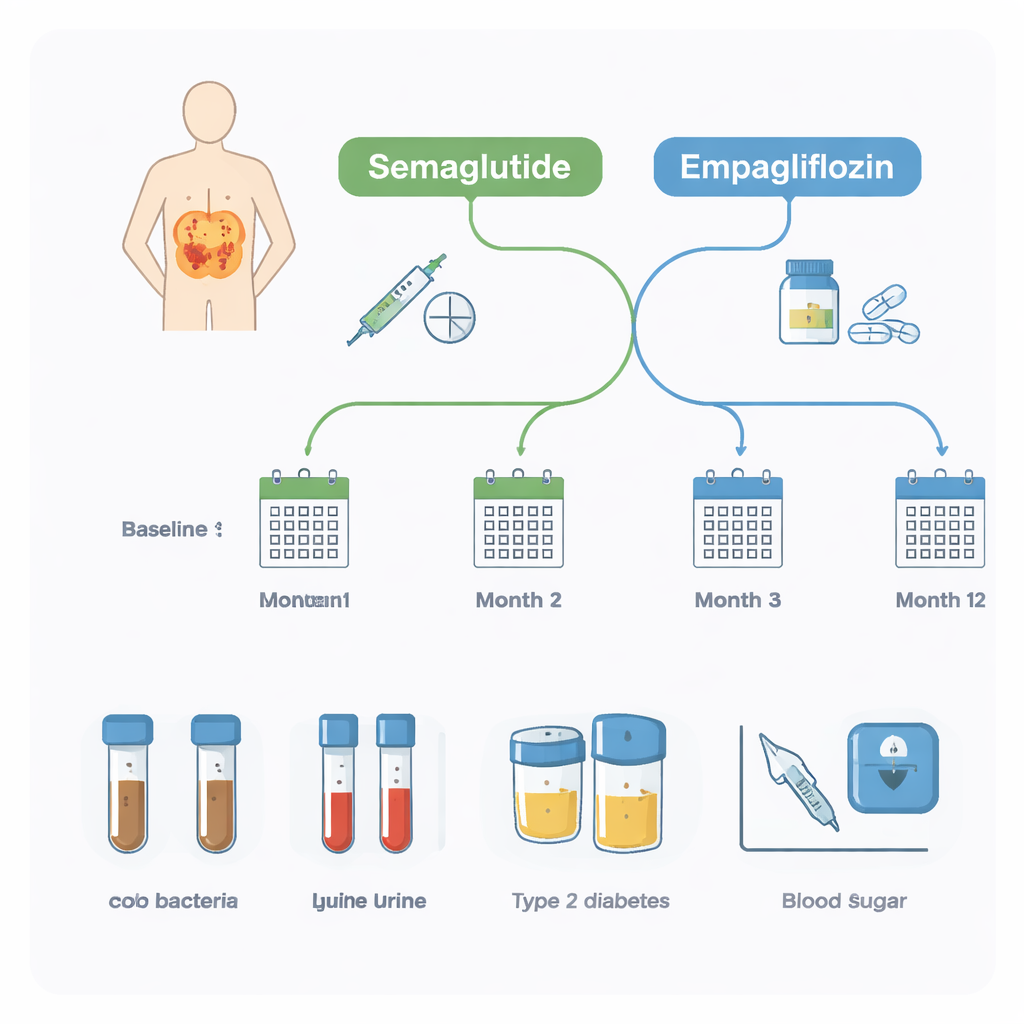
How the drugs affected health and gut communities
Clinically, both drugs improved blood sugar control by the third month of treatment, and semaglutide in particular produced marked and sustained weight loss over 12 months. When the team examined gut bacterial diversity – how many different types were present and how evenly they were distributed – they saw no clear overall shift after starting either drug. Likewise, when they looked at broad patterns in which bacterial groups were present, most apparent changes did not remain statistically solid once strict corrections for multiple testing were applied. Some bacterial groups and predicted metabolic pathways seemed to change between months 3 and 12, but these patterns were modest and likely reflected indirect effects of better metabolism or altered lifestyle rather than a direct impact of the drugs themselves.
Gut bacteria as potential predictors of success
The most intriguing findings came from looking at the microbiome before treatment started and asking whether it could predict who would improve the most. For people taking semaglutide, hundreds of baseline microbial features showed nominal links with later changes in clinical markers, especially white blood cell counts and a measure of inflammation known as the neutrophil‑to‑lymphocyte ratio. After strict statistical filtering, one bacterial group called Alistipes remained significantly associated: individuals with more of this bacterium at baseline tended to have a greater drop in inflammatory cells. Several bacterial genera and microbial pathways at baseline also tracked with how much HbA1c fell by the third month, hinting that the initial gut community may influence how strongly semaglutide lowers blood sugar. In the empagliflozin group, numerous bacteria and pathways were also tied to changes in HbA1c and kidney function measures, but none survived the most conservative statistical tests, in part because the study was small.
What the findings mean – and what they do not
Importantly, this work does not claim that semaglutide or empagliflozin dramatically remodel the gut microbiome on their own, nor that any specific bacterium guarantees success or failure. Rather, it suggests that subtle features of a person’s existing gut community may be linked to how their body responds – not only in terms of blood sugar but also inflammation and other health markers. Because all participants were already taking metformin, which itself affects gut bacteria, any added influence of the newer drugs may have been harder to detect. The modest sample size and some missing samples further limit how confidently individual bacterial groups can be singled out as true drivers of drug response.
Looking ahead to tailored diabetes care
For a layperson, the takeaway is that your gut bacteria might one day help doctors choose the diabetes drug that works best for you. In this early study, the mix of microbes in stool samples taken before treatment was linked to how strongly semaglutide and empagliflozin improved blood sugar and inflammation, while the drugs themselves produced only subtle, slow shifts in gut communities. Larger, longer studies will be needed to confirm which microbes truly matter and whether changing them can boost treatment effects. If those efforts succeed, a simple stool test could become part of personalized diabetes care, helping match the right medicine to the right microbiome. 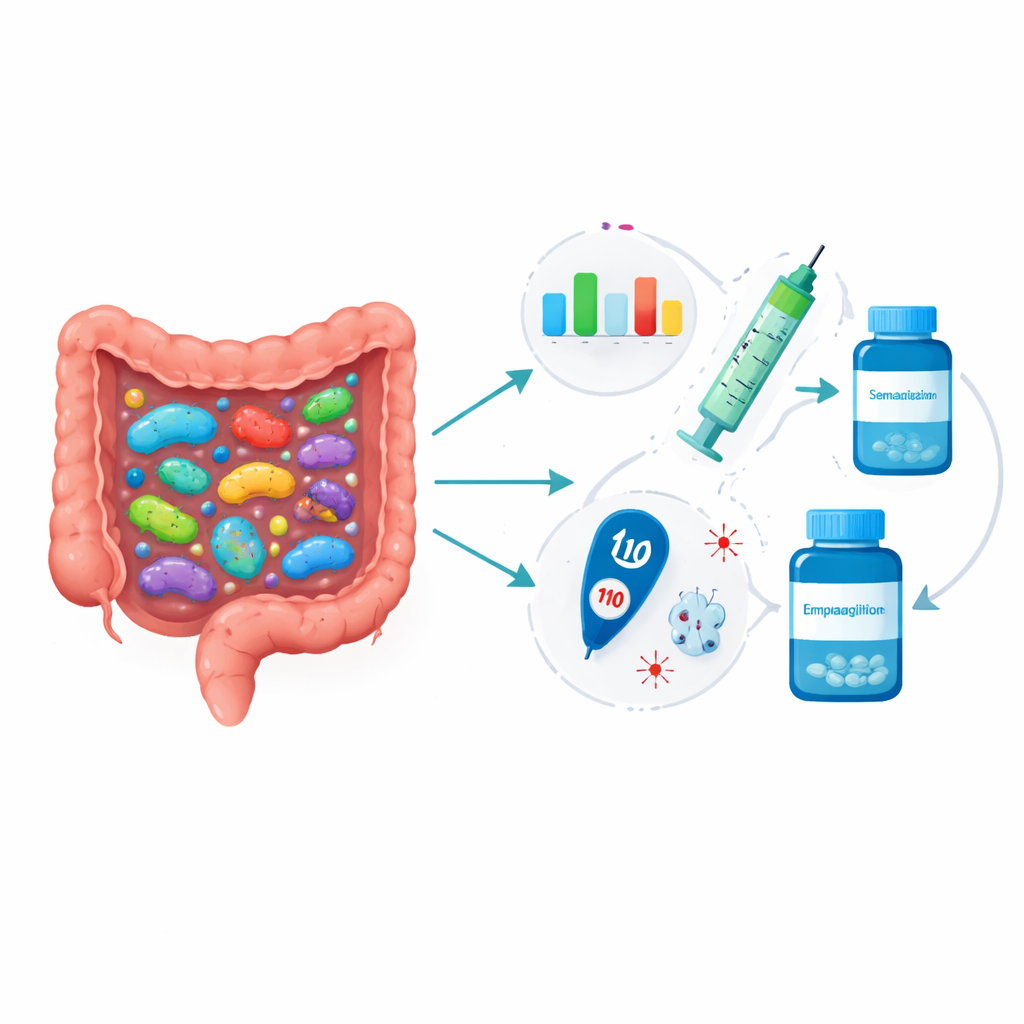
Citation: Klemets, A., Reppo, I., Krigul, K.L. et al. Fecal microbiome predicts treatment response after the initiation of semaglutide or empagliflozin uptake. Sci Rep 16, 6126 (2026). https://doi.org/10.1038/s41598-026-36318-3
Keywords: gut microbiome, type 2 diabetes, semaglutide, empagliflozin, personalized medicine