Clear Sky Science · en
Virtual screening of novel alkaloids as potent inhibitors for G2032R-mutant ROS1 kinase in non-small-cell lung cancer
Why this matters for future lung cancer treatments
Many people with a certain form of lung cancer initially respond well to modern targeted drugs, only to see the cancer return when the tumor evolves resistance. This study explores whether nature-made chemicals called alkaloids might offer a new way to block a stubborn, drug-resistant version of a cancer-driving protein, using advanced computer simulations instead of years of early lab work.
A cancer signal that learns to dodge today’s drugs
Non–small-cell lung cancer (NSCLC) is the most common type of lung cancer, and in a small but important fraction of patients the tumor is driven by a faulty protein called ROS1. Normally, ROS1 helps control cell growth, but when its gene is rearranged, the protein sends constant “grow and divide” signals, fueling cancer. Drugs such as crizotinib and later lorlatinib were designed to fit into ROS1 and switch it off. Over time, however, many tumors acquire a tiny change—one amino acid in the protein, called G2032R—that acts like a bump at the doorway of the drug’s docking site. This alteration makes it much harder for existing medicines to fit, and patients lose the benefit of treatment.
Looking to natural molecules for fresh ideas
Alkaloids are a huge family of nitrogen-containing compounds produced by plants and other organisms. They include famous medicines such as quinine for malaria and vinblastine for cancer. Because of their rich and varied shapes, alkaloids are a favored hunting ground for new drugs. In this work, the researchers assembled a digital library of 447 different alkaloids and asked a series of computer programs to predict which ones might best stick to the altered G2032R version of ROS1, while avoiding some of the weaknesses of current drugs.
Screening, stress-testing, and safety checks on a computer
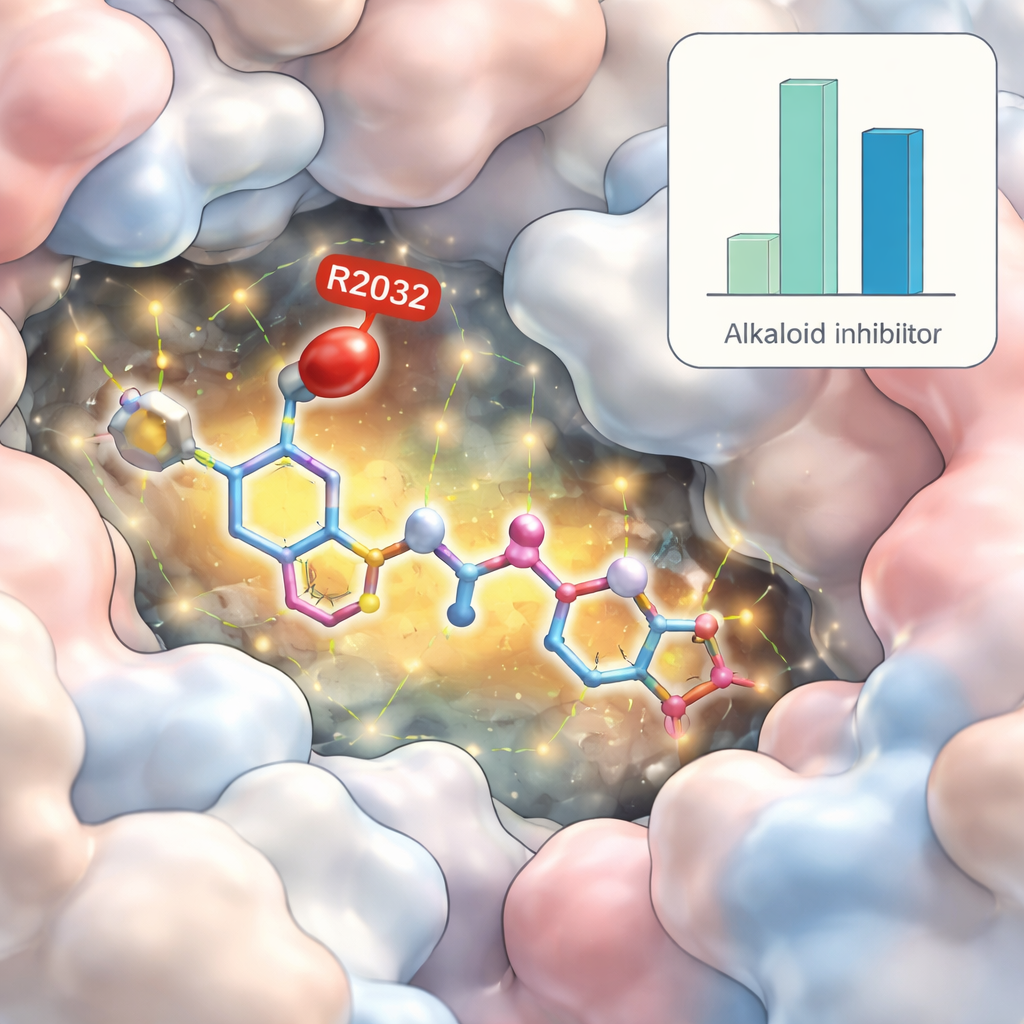
The team first used a technique called virtual screening, in which each alkaloid was computationally “docked” into the 3D structure of the mutant ROS1 protein to estimate how tightly it might bind. From this large set, two molecules stood out with especially strong predicted fit: yibeinoside A, a steroid-like alkaloid from the bulb of a Fritillaria plant, and vomicine, an indole alkaloid from the Strychnos nux-vomica tree. Their scores were as good as or better than lorlatinib, a current ROS1-blocking drug. The researchers then examined how these compounds were predicted to sit in the protein’s pocket and what kinds of chemical contacts they formed, including whether they could take advantage of the very mutation that defeats existing drugs.
Putting the best candidates through virtual motion
To go beyond static snapshots, the scientists ran molecular dynamics simulations, which model how the protein and drug move and flex in water over time. These “stress tests” suggested that complexes containing yibeinoside A stayed particularly stable, with only modest motion of the protein backbone and a tight, compact shape similar to that seen with lorlatinib. Vomicine also bound strongly but caused slightly more movement and loosening of the protein surface. Importantly, yibeinoside A was predicted to form a rich network of hydrogen bonds and hydrophobic contacts, including a direct interaction with the altered residue at position 2032, potentially turning the resistance-causing bump into an anchor point rather than an obstacle.
Early clues about benefits and risks
Because a strong binder can still make a poor medicine if it behaves badly in the body, the team used additional software to estimate absorption, distribution, metabolism, and toxicity. Both alkaloids were predicted to be absorbed in the gut, but vomicine showed warning signs: likely to damage DNA and cause cancer in animal models, and to be generally more toxic. Yibeinoside A, in contrast, showed a more favorable predicted safety profile, with the main concern being possible liver stress—a common issue that would need careful follow-up in the lab. When the researchers combined all their energy calculations, yibeinoside A and vomicine both appeared to bind the mutant ROS1 more strongly than lorlatinib, but only yibeinoside A balanced this with more reassuring safety predictions.
What this means and what comes next
This study does not deliver a ready-to-use drug, but it offers a strong computer-based case that yibeinoside A could be a promising starting point for medicines that tackle G2032R-mutant ROS1 in resistant lung cancer. By showing that a natural product can both fit the altered protein snugly and pass basic virtual safety checks, the work narrows the field to a realistic lead for chemists and biologists to test in real cells and animals. If future experiments confirm these predictions, yibeinoside A—or improved versions inspired by it—might one day help patients whose tumors have outsmarted current ROS1-targeting therapies.
Citation: Cho, SC., Wang, YW., Chu, CA. et al. Virtual screening of novel alkaloids as potent inhibitors for G2032R-mutant ROS1 kinase in non-small-cell lung cancer. Sci Rep 16, 5342 (2026). https://doi.org/10.1038/s41598-026-36317-4
Keywords: non-small-cell lung cancer, ROS1 mutation, drug resistance, alkaloids, virtual screening