Clear Sky Science · en
Consensus for the most suitable trial design to assess therapy for rare vascular malformations: a Delphi study
Why this research matters for families with rare conditions
For families facing rare blood vessel malformations in their children, finding reliable information about which treatments really work can be frustratingly difficult. Classic medical studies usually need large numbers of patients, but when a disease is rare and highly variable from one person to another, those big trials are hard to run. This article explores how doctors and patient representatives from around the world tried to agree on smarter, more realistic ways to test treatments for rare vascular malformations, so that children and adults can benefit from solid evidence even when patient numbers are small.
Rare vessel problems and the evidence gap
Vascular anomalies are disorders of blood or lymphatic vessels that can cause pain, swelling, bleeding, and visible marks on the skin. Some, such as infantile hemangiomas, are relatively common, but many others are rare and complex. They often appear in childhood and may affect a child’s face or limbs, creating both medical and emotional challenges. Standard randomized controlled trials—where patients are randomly assigned to one of two groups and followed over time—work well for common diseases, but in rare vascular conditions there may be only a handful of eligible patients worldwide. On top of that, each person’s malformation can look and behave differently, which makes straightforward comparisons between groups less reliable.
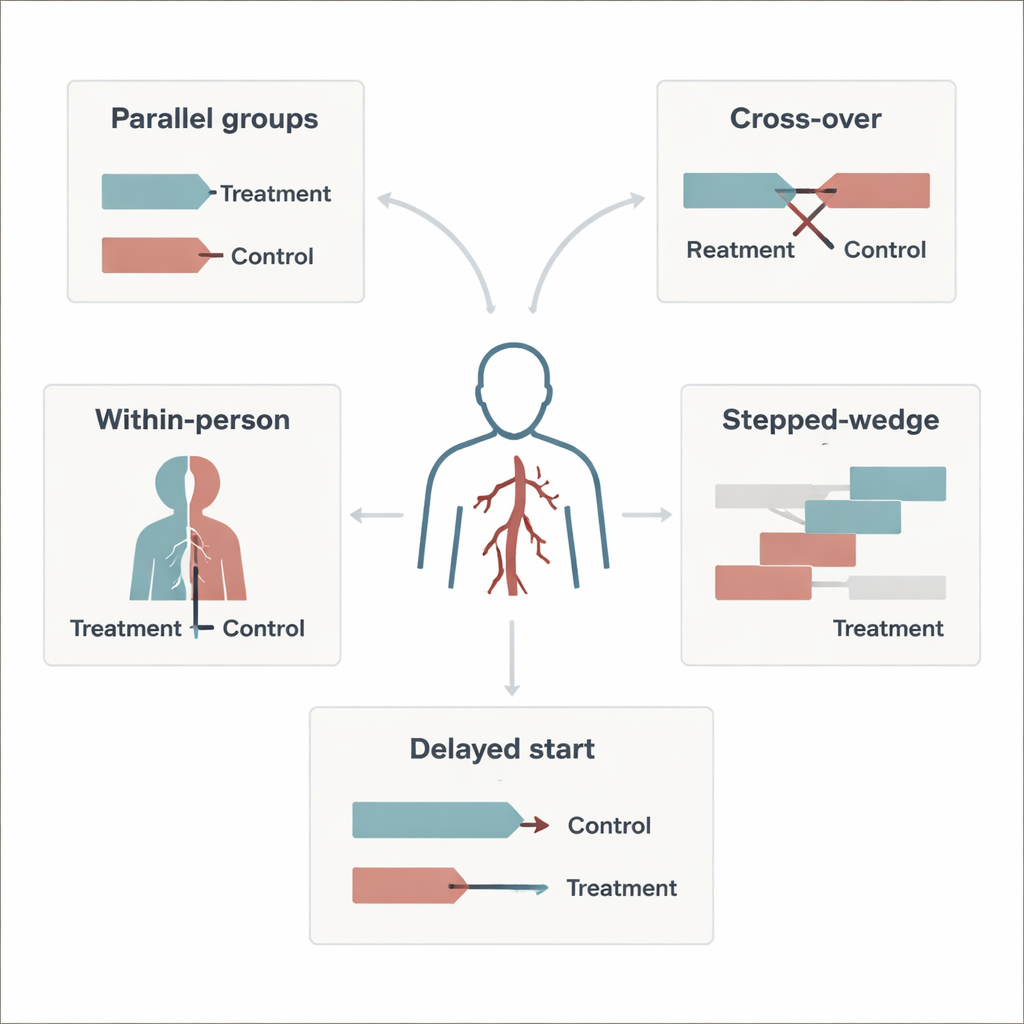
Looking for better ways to test treatments
To tackle this problem, researchers turned to a set of alternative trial designs that are especially suited to rare diseases. These include “cross-over” studies, where each patient receives both the new treatment and the comparison treatment at different times, and “within-person” or “split-body” studies, where different parts of the same person’s body receive different treatments. Other options are “delayed-start,” where everyone eventually gets the new treatment but some start later, and “stepped-wedge” designs, where treatment is rolled out in stages across patients or centers. All of these approaches try to make the most of small patient numbers, often by allowing each person to serve as their own control, which reduces the influence of natural differences between individuals.
Asking experts and families to weigh in
The team used a structured survey technique called the Delphi method to gather opinions from international experts and from patients and caregivers. This method involves several anonymous rounds of questionnaires, with feedback between rounds, to see whether a group can move toward agreement. More than 200 experts were invited; 29 took part in the first round and 26 in the second. Most were experienced physicians from Europe, North America, and Asia, and a smaller group were patients or caregivers who live with these conditions. The survey presented four realistic clinical stories involving children with different types of vascular malformations—treated with oral drugs, creams, injections, or laser therapy—and asked which trial designs would be feasible, acceptable, and scientifically appropriate in each situation.
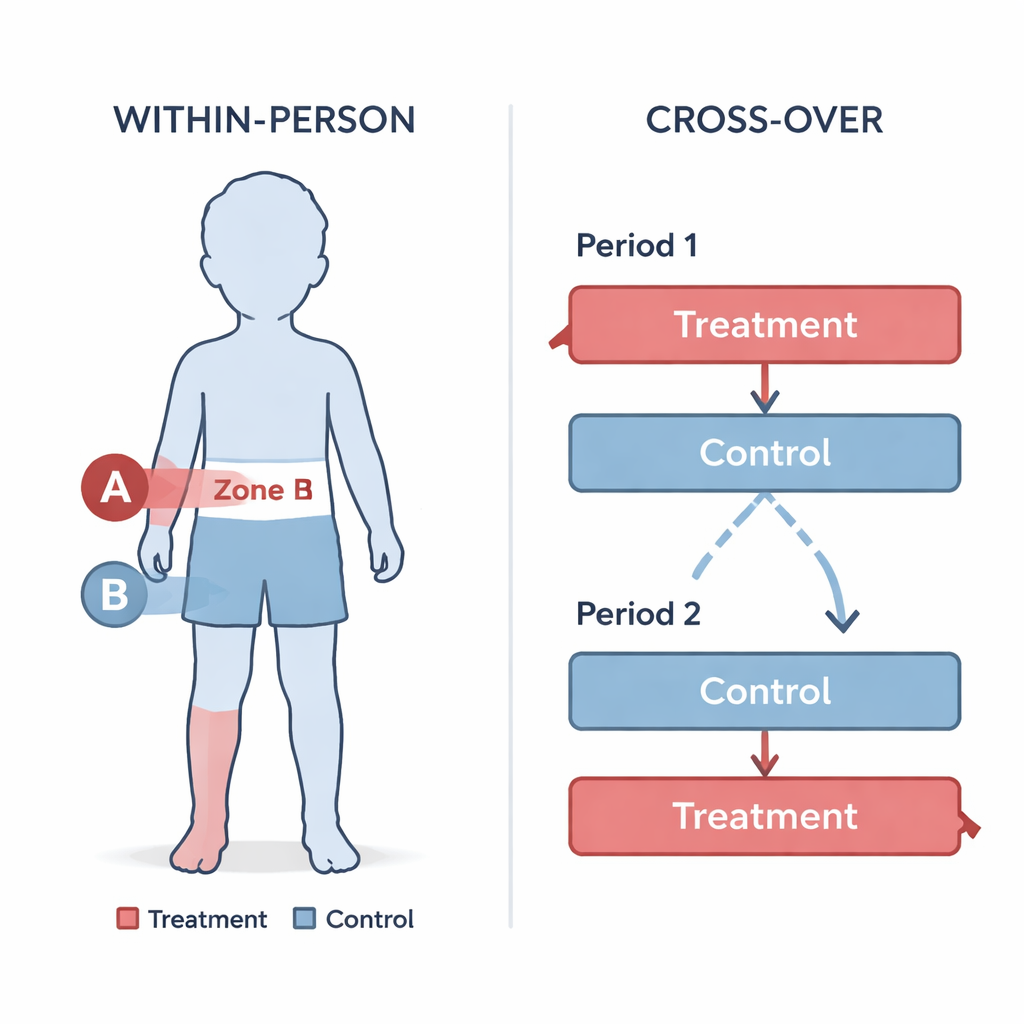
Where agreement was found—and where it was not
Across ten specific treatment situations, the panel reached clear consensus in only two, both of which favored designs that compare treatments within the same person. For example, for an oral drug in a nine-year-old girl and for an injectable drug in a teenager, many experts favored cross-over designs that let every participant try both options while still allowing blinded assessment. For laser treatment of a port-wine stain on the face, experts preferred a split-body approach, where one side receives active laser and the other a sham procedure, again with masked evaluation. These designs were appreciated because they use fewer patients, make it easier to hide which treatment is being given to avoid bias, and ensure that participants have access to the experimental option. However, in eight out of ten situations, opinions remained scattered. This reflected real uncertainties and the fact that no single design works best for every drug, lesion type, or ethical context—especially when there is no clear comparison treatment.
What this means for future studies and patients
For non-specialists, the main message is that clever study designs can help overcome the "too few patients" barrier in rare vascular malformations, but choices must be tailored to each clinical question. The experts generally agreed that trials which compare treatments within the same person—such as cross-over and split-body designs—are particularly promising when patient numbers are small and when it is safe and ethical to switch or divide treatment. At the same time, this Delphi study provides guidance rather than hard proof: it reflects expert preferences, not direct head-to-head testing of designs. The authors call for future work, including simulations and real trials, to test how well these designs perform in practice. Still, their findings offer a practical starting map for researchers planning studies, and a hopeful sign for patients and families who want better, faster answers about which treatments truly help.
Citation: Allemang-Trivalle, A., Giraudeau, B. & Maruani, A. Consensus for the most suitable trial design to assess therapy for rare vascular malformations: a Delphi study. Sci Rep 16, 5638 (2026). https://doi.org/10.1038/s41598-026-36295-7
Keywords: rare diseases, vascular malformations, clinical trial design, Delphi study, cross-over trials