Clear Sky Science · en
Overcoming Candida albicans biofilm drug resistance via azole-sophorolipid synergy
Why stubborn fungal infections matter
Anyone who has had a persistent yeast infection, a central line in the hospital, or a medical implant is potentially at risk from Candida albicans, a common fungus that can turn dangerous when it forms hard-to-treat communities called biofilms. These slimy layers protect the microbes from many of our best antifungal drugs, allowing infections to linger and sometimes turn life-threatening. The study described here looks for a new way to crack open those defenses by pairing existing drugs with a natural soap-like molecule made by yeast.
A hidden shield on medical surfaces
Candida albicans can stick to tissues and medical devices and then build up into structured biofilms—dense layers of cells held together by a self-made matrix of sugars, proteins, and DNA. Inside this fortress, the fungus slows its metabolism and pumps drugs back out, so medications that easily kill free-swimming cells barely touch biofilm-dwelling ones. Azole drugs such as fluconazole and itraconazole are mainstays of antifungal therapy, but against mature biofilms they often require much higher doses, which can cause side effects and still fail to fully clear the infection. Clinicians increasingly face infections that are both biofilm-based and resistant, underscoring the need for helper molecules that can weaken the biofilm itself.
Soap-like helpers from a friendly yeast
The researchers turned to sophorolipids, natural, biodegradable surfactants—molecules that behave a bit like very mild detergents—produced by certain non-pathogenic yeasts. These molecules have a water-loving sugar head and an oily tail, allowing them to sit at surfaces and interfaces. In this study, a yeast called Starmerella riodocensis was used to produce a mixture of sophorolipids, which was then purified and analyzed. On their own, these sophorolipids did not kill free-floating Candida cells even at high doses, but they did interfere strongly with the fungus’s ability to form and maintain biofilms. Young, just-forming biofilms were especially sensitive, and even established 24‑hour biofilms could be thinned and weakened when exposed to purified sophorolipids.
Teaming up with existing antifungal drugs
The central question was whether sophorolipids could make standard azole drugs work better. The team tested combinations of sophorolipids with three widely used azoles: fluconazole, itraconazole, and ketoconazole. When they exposed biofilms to these mixtures, they found that some pairings did more than simply add their effects—they acted synergistically, meaning the combo was more powerful than expected from each component alone. The standout partnership was itraconazole plus sophorolipids. Against tough, preformed biofilms, the amount of itraconazole needed to cut the biofilm in half dropped up to sixteenfold, while the required sophorolipid dose also fell several-fold compared with using sophorolipids alone. Fluconazole and ketoconazole combinations showed more modest gains, suggesting that the chemical nature of each drug influences how well it benefits from the surfactant "assist."
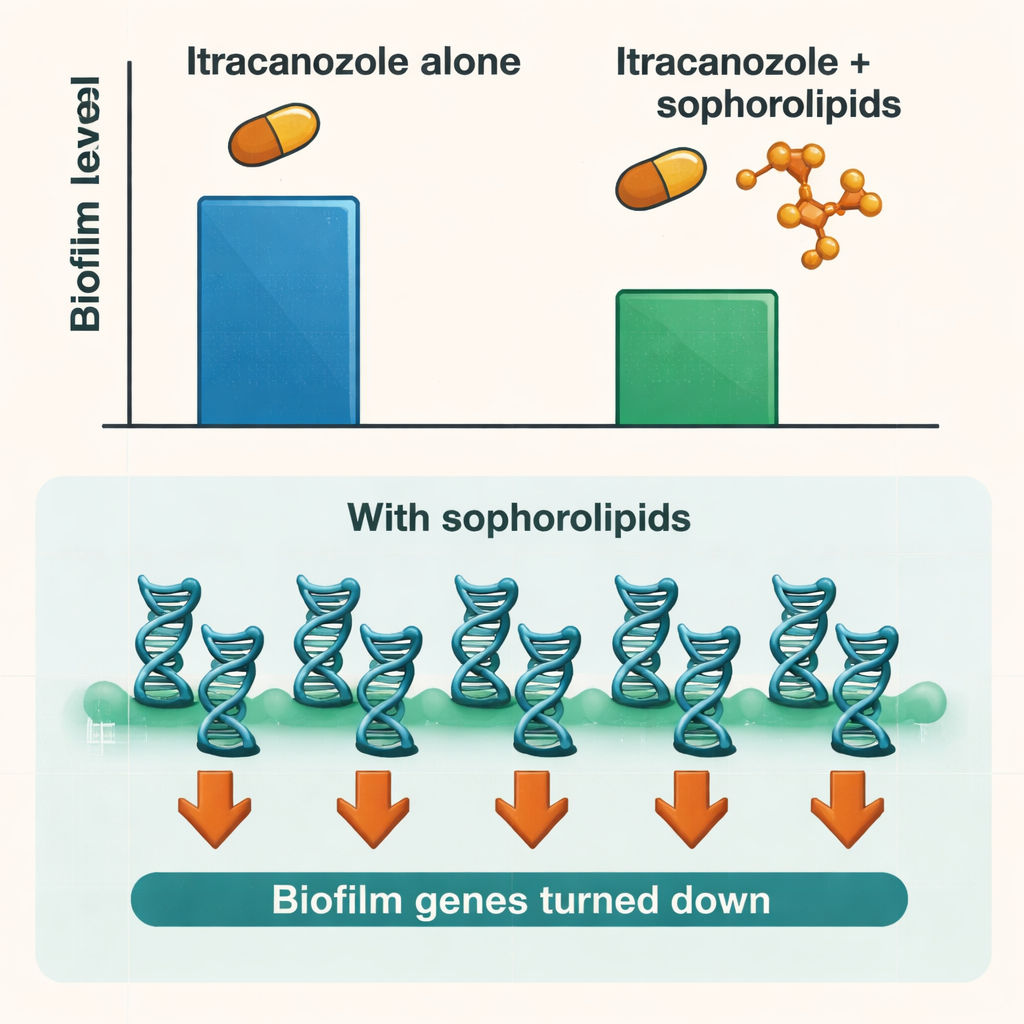
Silencing the fungus’s biofilm program
To understand what was happening inside the fungal cells, the researchers examined the activity of key Candida genes involved in sticking to surfaces, growing as filaments, and building the biofilm matrix. Treatment with sophorolipids alone dialed down many of these genes, especially those controlling the earliest steps of adhesion and the shift from yeast-like to filamentous forms. When cells were synchronized at a specific cell-cycle stage that favors biofilm formation, the gene-silencing effects were even stronger. This suggests that sophorolipids do not simply wash away the biofilm; they also disrupt the fungus’s own biological program for constructing and maintaining its protective community.
Promise for gentler, more effective treatments
The study concludes that sophorolipids from Starmerella riodocensis can act as safe, biofilm-targeting partners for azole drugs, with the itraconazole–sophorolipid combination being especially potent. Because sophorolipids show low toxicity in mammalian cells and mainly weaken the biofilm structure and gene networks rather than directly poisoning the fungus, they may allow doctors to use lower doses of conventional drugs while still clearing stubborn infections on devices and tissues. For patients, this kind of smart combination therapy could someday translate into treatments that are both more effective against resistant Candida biofilms and kinder to the rest of the body.
Citation: Jayasekara, L.C.B., Watchaputi, K., Butkinaree, C. et al. Overcoming Candida albicans biofilm drug resistance via azole-sophorolipid synergy. Sci Rep 16, 5963 (2026). https://doi.org/10.1038/s41598-026-36278-8
Keywords: Candida biofilm, azole antifungals, sophorolipids, drug synergy, antimicrobial resistance